
ISO 13485:2016 Certified Product Development

PORTFOLIO OF ENGINEERING CASE STUDIES AND MARKETS
Up-close, in-depth, and detailed.
Check out our Engineering Case Studies for an in-depth look at how our multi-disciplinary team innovates and delivers industry-leading projects.
Beyond the myriad technical challenges to be solved when creating any new product, designing novel diagnostic instruments requires a unique blend of systems design expertise and biochemical process understanding.
Connected Consumer
Wearable technology has seen an explosion in popularity in recent years, with consumers rushing to purchase devices to monitor their heart rate, blood pressure, activity, sleep, and much more.
Medical Devices
Medical devices are subject to regulatory requirements that ensure a well-documented and reviewable design process — and ultimately, a safe product.
Artificial Intelligence (AI)
Commercial equipment.
Simplexity defines commercial products as equipment that our clients sell to other businesses or use themselves as a part of their core technology.
Test Fixtures
“One of the great things about Simplexity is that their expertise is fairly wide ranging throughout the product development process. It's great to have a partner like Simplexity who has so much expertise in electro-mechanical systems as well as embedded software because it brings so much capability to my engineering team in those really critical technology areas.”
-Steve Schell CEO and Co-Founder, New Matter
IUNU ROBOTIC VINE PLANT MONITOR
Viewplus color inkconnect, automated dna sample extraction robot, reperio health, food automation vending machine, anavasi diagnostics molecular detector, nailbot by preemadonna, case study: oregon health & science university (ohsu) vascular monitoring, bio1 systems, catheter design applications, custom syringe and air-displacement pump, non-electric oscillating toothbrush.
A company based in providing environmentally conscious dental care products created a toothbrush with improved cleaning performance of an electric brush without the environmental impact associated with battery-powered electronics. Complex mechanical design considerations associated with gear trains, dynamic mechanisms, and stored energy were an important part of this final product design.
OPULUS LUXURY SKINCARE DEVICE
Ossia cota forever battery.
Ossia has a revolutionary, proprietary technology which eliminates the need for power cords or inductive charging pads by charging devices over the air at distances of multiple meters.
STERIFRE AURA
Sterifre Medical chose Simplexity to improve AURA, their automated point-of-care disinfection device. Noise reduction, weight reduction, and cost reduction were key in the improvement of this device while maintaining functionality and quality.
Valve / HTC VIVE VR
The Valve/HTC Vive Virtual Reality system is recognized as offering the best in class VR experience. A complex head mounted display worn by the user uses state of the art technology to achieve the highest in performance while maintaining minimum weight and a comfortable user experience.
Valve / VALVE INDEX VR
The Valve Index Virtual Reality system is recognized as offering the best in class VR experience. A complex head mounted display worn by the user uses state of the art technology to achieve the highest in performance while maintaining minimum weight and a comfortable user experience.
HP ENVY Zero-Gravity Printer
Simplexity helped solve the challenges posed by dedicating one of the most printer-experienced teams, with cumulative combined printer-experience exceeding 50 years.
Senaptec Strobe Eyewear
Simplification goes a long way when it leads to reducing manufacturing costs by 35%
New Matter 3D Printer
Before New Matter the company, there was just a guy with the notion that 3D printing was too complicated.
Bowflex SelectTech 560
Nautilus asked Simplexity to assist with sensor architecture, signal processing, and firmware development. Our engineers worked closely with Nautilus to develop an embedded instrumentation PCA based upon low cost mems technology.
NeuraLabel 300x On-Demand Label Printer
The quality and speed of HP’s record-setting page-wide array technology transforms the label market.
Light Sensor Calibration System
Microsoft band.
When Microsoft kicked off their project to get into the booming “wearables” marketplace, they had some big challenges and little time to conquer them. After assessing their hurdles, they asked Simplexity to step in and help.
Computer aided CPR Training
Working in close collaboration with HSI, Simplexity developed a microprocessor based LOOP CPR Controller that is used with a standard CPR manikin during training.
Autofocus Module
Simplexity developed a prototype focusing module that replaced traditional bearing rails with a flexure (aka a compliant mechanism). The flexure consists of parallel plates that act like a four-bar-linkage over a limited motion range.
Library Preparation Device
A fully functional production prototype of a complex instrument with a proprietary fluidic transport system.
Molecular Diagnostic Testbed
A testbed that enables end-to-end assay workflow development on a prototype cartridge for a point-of-care molecular diagnostic device.
Embedded 2-Axis Stage for Scientific Imaging
When converting from off-the-shelf motion technology to a customized embedded solution, a major life sciences company chose Simplexity to minimize development risk.
HP Officejet Pro X
Helping HP develop a page wide office printer that is twice the speed and half the price of the color laser.
Increasing patient compliance via an in-home, easy-to-use dried blood spot collection device
Biomaterial Analyzer
Simplexity engineers helped optimize the design of a biomaterial analyzer that integrates biochemistry, fluidics, electronics, and mechanics.
Bullseye Custom Golf Club Bender
Simplexity built a unique manufacturing tool that brings custom precision to the manufacturing floor that adjusts the loft and lie angles of golf clubs with unprecedented speed and precision.
High Accuracy GPS System
When a low cost and high accuracy GPS solution was needed for an industrial application, Simplexity engineers collaborated with the client to complete the mechanical and firmware design.
Library Preparation System Breadboard
Learn more about the Library Preparation System Breadboard that Simplexity helped to design in our Case Study!
HORIBA Surface Analyzer
Designing a quality product that can meet the business and customer needs can be a risky proposition, even when a company has the engineering talent to do the work.
Reagent Dispensing Robot
Next-generation sequencing (NGS) library preparation involves fragmenting a genomic DNA sample and ligating specialized adapters to both fragment ends. This high-quality sample serves as the input for a sequencing instrument to perform the gene sequencing process.
END-OF-LINE TESTER FOR DIAGNOSTIC INSTRUMENT
Sample prep testbed, benchtop end-of-line tester for handheld device , hardware based firmware test rig, preemadonna nailbot printer pca tester, test fixture, flex cable life testing, wireless pressure sensing, led life testing, automatic sheet feeder, benchtop tester for mobile device main pca, benchtop tester for mobile device modem pca , drop test fixtures, life tester for cpr training device, uv and epoxy bonding characterization, sensor calibration system, end of line test tool for x-y stages, drop detect, flex heater tester, thermal testing, would you like to discuss your project, phase 3: design verification and design transfer, design & engineering, software: design complete, hardware: pre-production units for design verification.
Test: Design verification test
Mfg. readiness: cm schedule and budget, unit build tracking.
Quality: Quality metrics verification process, Process validation support
The design team works closely with the manufacturing team to enable a smooth transfer, often with Simplexity engineers traveling to the contract manufacturer sites to ensure product quality. The design is transferred to the client based upon specific needs, most often after all tests are complete and the design is verified.
Typical deliverables:
- Pre-production units
- Formal verification test reports
- Design transfer package, including Design History File (DHF) if needed for FDA submittal
- Process validation support
- Unit build tracking
- Contract manufacturing schedule and budget
- Quality metrics verification
Gate definition:
- Design verification complete
Phase 2C: Detailed Design Prototype 2
Software: full feature implementation, hardware: prototype 2 units with production-representative materials and processes, test: engineering confidence test, integration test.
MFG. Readiness: CM onboarding Design transfer prep
Quality: build quality plan, 2c. prototype 2 design, build and test.
Phase 2C iterates on the learnings of Phase 2B and involves a refined prototype build of a fully integrated system. Some projects also benefit from additional iterations of the product based on prior learnings through additional phases (2D, 2E, etc), which are not represented in this graphic. All requirements are intended to be tested, and at the end of Phase 2 there will be confidence that the units will pass verification in Phase 3. The Bill of Materials is further refined, and the team updates estimates for the per unit cost of the product by receiving pricing from vendors and suppliers.
- Updated prototypes
- Software and/or firmware binaries and source code
- Updated schematics and layout
- Updated 3D CAD files and 2D drawings
- Verification/test plans and reports
- Updated Bill of Materials (BOM) and Cost of Goods Sold (COGS)
- Build Quality Plan development
- Design transfer preparation
- Contract Manufacturer onboarding
- Engineering confidence test reviews (integration tests)
Phase 2B: Detailed Design
Software: core functionality implementation, hardware: prototype 1 units with rapid prototyped components, test: engineering confidence test, unit test, mfg: readiness: project build plan cm selection, quality: critical manufacturing process identification, 2b. prototype 1 design, build and test.
The detailed design phase usually has multiple, iterative sub-phases as the design progresses and representative prototypes are built. Phases 2B and 2C are typically the largest efforts in the product development process, where the specific implementation for all disciplines occurs (mechanical, industrial design, electrical, firmware, systems, software, manufacturing, and quality).
Simplexity typically engages with production component suppliers and contract manufacturing groups early in this phase to provide additional manufacturing input on the design. If the product has stringent testing or certification requirements, pre-screens are performed in this phase prior to formal regulatory agency testing.
- Prototypes (3D printed or other rapid prototypes, electrical PCAs, and/or preliminary code)
- Software and hardware design documentation
- Initial product firmware or software binaries and source code
- Electrical schematics and layout
- 3D CAD files
- Design failure mode and effect analysis
- Test plans and reports
- Project build plan – from prototype to pre-production
- Initial Bill of Materials (BOM) and Cost of Goods Sold (COGS)
- Manufacturing process identification
- Contract Manufacturer (CM) selection
- Engineering confidence test reviews
Phase 1: Requirements & Planing
Project plan requirements, id/ux concepts, risk analysis, manufacturing strategy identification.
- Product requirements document
- Project development plan (including plans for software/firmware electrical, quality, systems, and mechanical)
- Risk analysis
- Industrial Design (ID) and User Interface (UI) concepts
- Product requirements document complete
- Client approval of project development plan
Manufacturing design guidance and ongoing engineering support
Ongoing quality metrics monitoring & optimization.
Simplexity has a dedicated New Product Introduction (NPI) team that can guide the transition from design into production. The NPI team presents multiple options for manufacturing to the client, allowing clients to choose the solution that best suits their needs. This can involve Simplexity performing initial builds in-house prior to full handoff to a contract manufacturer or building the product via established relationships with contract manufacturing partners either domestically or overseas early in the process.
- Manufacturing guidance and ongoing engineering support
- Ongoing quality metrics monitoring and optimization
Phase 2: Detailed Design
Software: architecture design: block, sequence and state diagrams, hardware: major component definition & proof of concept subsystems build, test: characterization and qualification of high risk subsystems & components, quality: design for manufacturing tradeoffs evaluation, 2a. architecture and technology feasability.
The detailed design phase starts with defining options for the product architecture, with the goal of having the greatest chance of successfully meeting product requirements while best mitigating risk. Engineering activities in this phase include presenting options for hardware components, outlining the system block, sequence, and state diagrams, creating rough CAD, and breadboarding of high-risk subsystems. Results are presented with a description of the pros, cons, and key tradeoffs for each scenario.
- System architecture design (including mechanical, electrical and software/firmware)
- Initial product risk analysis
- Breadboards or proof-of-concept prototypes of high-risk technologies or subsystems.
- ID concept models
- Client approval following hardware and software architecture reviews
Phase 0: Exploration
Exploration, concept work, architecture explorations, feasibility study.
- Exploration report
- Client approval on feasibility of idea
What is Product Engineering
What is Product Engineering? Key Insights and Examples
Turn your ideas into reality with this detailed guide on what is product engineering. Get everything from design to development and reliable examples.
Case studies
Get an estimation
Summary by AI ✦
It is a comprehensive guide covering key insights and examples, aimed at helping users understand the process of turning ideas into real-world products. The article covers everything from design, development, and prototyping to tools and technologies, highlighting successful product engineering examples such as Apple, Tesla, and Amazon Web Services.

Product engineering is where creativity meets technology, driving the development of innovative products that we rely on every day. It's the process of turning ideas into reality, from brainstorming and design to testing and bringing products to market. And with the global product engineering market projected to hit $1,042.6 billion by 2027, growing at a rapid 10.7% annually, it’s clear that more and more businesses are focusing on building products that can truly make a difference.
In our fast-moving world, customers expect quick, reliable, and user-friendly products. Product engineering helps companies meet those expectations by creating solutions that are not only cutting-edge but also built to scale. Whether it’s software, smart devices, or AI-powered technology, product engineering is at the heart of innovation, helping companies launch successful products that keep pace with changing demands.
Product Engineering Statistics
70% of digital transformation initiatives fail due to lack of user adoption, highlighting the importance of customer-focused product engineering.
60% of a product's development cost typically comes from fixing defects, stressing the need for quality assurance during the engineering process.
Only 35% of companies successfully launch products on time, reflecting the challenges in project management and product engineering efficiency.
87% of businesses consider speed to market as a key advantage of agile product engineering practices.
95% of product engineers report that collaboration tools significantly improve their productivity and output quality.
Companies investing in design see a 228% higher return on investment , underlining the crucial role of product engineering in creating user-centric designs.
44% of engineers believe that integrating AI into product development enhances innovation and streamlines the process.
78% of top-performing companies prioritize continuous integration and delivery in their product engineering cycles.
65% of organizations using cloud platforms report improved product development agility and reduced time-to-market.
85% of product failures are attributed to ineffective engineering and design processes, emphasizing the need for robust methodologies and tools.
The Role of Product Engineering
You have a brilliant idea for a product—something that could change lives, solve a problem, or make everyday tasks easier. But turning that idea into something real and tangible isn't just about creativity; it's about engineering. That's where product engineering steps in, bridging the gap between imagination and reality.
Product engineering transforms your big idea into a functional, real-world product. It's like having a blueprint for success. Whether it's a sleek gadget or a cutting-edge app, product engineers take your concept and develop it into something that's not only practical but also market-ready. They ensure that every detail, from design to functionality, works best together so that your vision becomes something people can actually use and love.
Integration of Various Disciplines
But here’s the exciting part: product engineering isn’t just about one thing—it’s about bringing together different areas of expertise to create something awesome.
Design: First, it starts with design. This isn’t just about making something look cool (though that’s important too). Product engineers and designers work hand in hand to make sure the product not only looks good but also works beautifully in the real world. It’s about merging aesthetics with usability.
Development: Then comes the development phase. Now, we’re talking about the technical side—where the design becomes a working product. Whether it’s coding for software or creating prototypes for physical products, engineers ensure that what was once a concept can actually be built and function properly.
Manufacturing: Finally, there’s manufacturing. You’ve got the design, and it works—great! But how do you produce it at scale without losing quality? That’s where product engineering shines. It ensures that your product can be efficiently manufactured, using the right materials and methods, while maintaining top-notch quality. It’s the difference between a cool prototype and a successful product on store shelves.
By bringing these disciplines together, product engineering ensures that your idea doesn’t just stay in your head—it becomes a real product that people can buy.
Key Phases of Product Engineering

1. Concept Development
This initial phase focuses on brainstorming ideas, understanding market needs, and defining the product's purpose. It involves research, feasibility analysis, and creating a product vision.
Ensures the product meets market demand and customer needs.
Provides a clear direction for the entire project.
Establishes a strong foundation for future product development .
The design phase translates the concept into a tangible form through detailed product specifications, including aesthetics, functionality, and user experience.
Enhances usability and functionality for a better user experience.
Aligns the product with technical requirements and user expectations.
Creates a visually appealing and marketable product.
3. Prototyping
Prototyping involves creating a working model of the product. It allows engineers to test design ideas and functionalities on a smaller scale before full-scale production.
Identifies and resolves design flaws early on, saving time and resources.
Offers a tangible model for stakeholder review and approval.
Enables experimentation with different ideas and functionalities.
Testing ensures that the product meets safety, quality, and performance standards. It involves rigorous assessments like functionality tests, stress tests, and user trials.
Minimizes risks of defects, ensuring a reliable and safe product.
Verifies that the product meets industry standards and customer expectations.
Enhances overall product quality and performance through rigorous assessments.
5. Production
Once testing is complete, the product moves to mass production. This phase involves scaling up manufacturing, quality control, and distribution planning.
Ensures timely and scalable delivery while maintaining high quality.
Efficiently brings the product to market on a larger scale.
Maintains consistency and control over the final product output.
Each phase is interconnected and essential for the overall success of the product. Skipping or underestimating any of these stages can lead to increased costs, delays, and ultimately, product failure.
Tools and Technologies in Product Engineering
Product engineering involves a wide range of tools and technologies that help streamline the development process, improve accuracy, and enhance collaboration among teams. Here are some of the most common tools and technologies used:
1. CAD Software (Computer-Aided Design)
CAD software is one of the foundational tools in product engineering. It allows engineers to create detailed 2D and 3D models of products, facilitating precise design and testing before any physical prototypes are made. Popular CAD software includes AutoCAD, SolidWorks, and Fusion 360.
CAD software streamlines the engineering process by enabling easy modifications, simulations, and analysis of designs, reducing errors and speeding up the development timeline.
2. PLM Software (Product Lifecycle Management)
PLM software helps manage the entire lifecycle of a product, from concept and design to manufacturing and disposal. It centralizes engineering data management , documentation, and processes, ensuring that all teams are aligned. Examples include Siemens Teamcenter and PTC Windchill.
PLM tools streamline workflows by providing a single source of truth for product data, improving collaboration, and reducing miscommunications across departments.
3. CAE Software (Computer-Aided Engineering)
CAE tools allow engineers to simulate and analyze how a product will perform under various conditions, such as stress, heat, or fluid dynamics. ANSYS and Abaqus are common CAE tools.
These simulations help engineers identify potential issues early in the design process, reducing the need for physical prototypes and speeding up the time to market.
4. Version Control Systems
Tools like Git and SVN allow engineers to manage changes to design files, code, and documentation over time. They track revisions and enable multiple team members to collaborate without overwriting each other’s work.
Version control ensures that engineers can work together efficiently and maintain a history of changes, reducing the risk of data loss or conflict.
5. 3D Printing Technologies
3D printing, or additive manufacturing, is used to create physical prototypes quickly from digital models. This technology is often integrated with CAD software and is widely used for testing and refining designs.
3D printing accelerates the prototyping phase, allowing engineers to test physical models and make iterative improvements without the time and cost of traditional manufacturing methods.
6. Project Management Tools
Tools like Jira, Trello, and Microsoft Project are essential for managing timelines, tasks, and team collaboration in product engineering. They help teams stay on track, prioritize tasks, and communicate effectively.
By organizing tasks and providing visibility into project progress, these tools help engineers and project managers meet deadlines and stay within budget.
Digital Product Engineering Tools
Challenges in Product Engineering

1. Budget Constraints : Limited financial resources can impact the scope, quality, and speed of product development. Budget constraints may lead to compromises in features or quality.
Prioritize Features: Focus on core functionalities that deliver the most value and can be developed within the budget.
Lean Development: Use agile methodologies to iteratively develop and test features, allowing for adjustments based on budget and feedback.
Cost-effective Tools: Utilize open-source tools or platforms that can reduce development costs.
2. Time Limitations :Tight deadlines can pressure teams to deliver quickly, potentially compromising quality or thorough testing.
Project Management : Implement strong project management practices, such as detailed planning and scheduling, to manage time effectively.
Agile Methodologies: Use agile practices like sprints to break the project into manageable chunks, allowing for faster iterations and adjustments.
Resource Allocation: Ensure that the team has the right mix of skills and resources to meet deadlines efficiently.
3. Technical Debt : Accumulation of shortcuts or quick fixes made during development that may lead to future issues or inefficiencies.
Regular Refactoring : Allocate time for regular code reviews and refactoring to address technical debt proactively.
Documentation : Maintain thorough documentation to avoid misunderstandings and ensure that quick fixes are addressed in future updates.
4. Integration Issues : Difficulties in integrating new products with existing systems or third-party services can lead to delays and additional costs.
API-first Approach : Design products with robust APIs to facilitate easier integration with other systems.
Early Testing : Test integrations early and often to identify and address issues before they escalate.
5. Changing Requirements : Shifts in project scope or requirements can disrupt development and lead to rework.
Change Management : Implement a change management process to evaluate and control changes in requirements.
Stakeholder Communication : Maintain clear and continuous communication with stakeholders to manage expectations and incorporate changes effectively.
6. Quality Assurance : Ensuring that the product meets quality standards and is free of defects can be challenging, especially under tight deadlines.
Automated Testing : Use automated testing tools to increase efficiency and coverage in quality assurance.
Continuous Integration : Implement continuous integration practices to detect issues early and ensure that the product remains stable throughout development.
7. Team Collaboration : Coordination among team members can be challenging, especially in remote or cross-functional teams.
Collaborative Tools : Use collaboration tools and platforms to facilitate communication and coordination among team members.
Regular Meetings : Hold regular meetings to align the team, address issues, and ensure that everyone is on the same page.
Addressing these challenges effectively requires a combination of strategic planning, adaptive methodologies, and clear communication. By implementing these strategies, product engineering teams can navigate obstacles and deliver successful products.
The Impact of Emerging Technologies
Emerging technologies such as Artificial Intelligence (AI), the Internet of Things (IoT), and 3D printing are revolutionizing the field of product engineering. These advancements are not only enhancing design and manufacturing processes but also enabling the creation of innovative products that were previously unimaginable.
1. Artificial Intelligence (AI)
Design Optimization : AI algorithms analyze vast amounts of data to optimize product designs, improving efficiency and performance. Machine learning can predict potential design flaws or improvements.
Predictive Maintenance : AI helps in predicting equipment failures before they occur, reducing downtime and maintenance costs.
Enhanced User Experience : AI-driven features, such as personalized recommendations and intelligent interfaces, enhance the overall user experience.
Autonomous Vehicles : Companies like Tesla use AI for developing self-driving cars, employing machine learning to improve safety and navigation systems.
Smart Home Devices : AI powers voice assistants like Amazon Alexa and Google Home, enabling voice-controlled interactions and smart home automation.
2. Internet of Things (IoT)
Connected Products : IoT enables products to connect and communicate with each other, providing real-time data and insights. This connectivity enhances functionality and user interaction.
Data-Driven Decisions : IoT devices collect and analyze data, helping manufacturers and users make informed decisions about product usage and performance.
Remote Monitoring and Control : IoT allows for remote monitoring and control of devices, improving convenience and efficiency.
Smart Thermostats : Products like the Nest Learning Thermostat use IoT to adjust home temperatures based on user behavior and preferences.
Wearable Health Devices : Smartwatches and fitness trackers, such as the Apple Watch, monitor health metrics and sync data with health apps for personalized insights.
3. 3D Printing (Additive Manufacturing)
Rapid Prototyping : 3D printing accelerates the prototyping process, allowing engineers to quickly create and test physical models of their designs.
Customization : It enables the production of highly customized and complex parts that traditional manufacturing methods cannot achieve.
Reduced Waste : Additive manufacturing produces parts layer by layer, minimizing material waste compared to subtractive manufacturing methods.
Medical Implants : Companies like Organovo use 3D printing to create custom medical implants and prosthetics tailored to individual patient needs.
Aerospace Components : NASA and companies like Boeing use 3D printing to produce lightweight, high-strength aerospace components, reducing costs and improving performance.
Have you learn this product usability vs ux with examples for creating functional product.
Successful Product Engineering Examples
Here’s a brief overview of three companies that excelled in product engineering, along with the strategies they employed and the lessons learned:
1. Apple Inc.
Case Study: Apple’s Product Engineering Excellence
Strategies Employed:
Integrated Ecosystem: Apple designs its products, software, and services to work seamlessly together. This integration ensures a smooth user experience and encourages customer loyalty.
Design Thinking: Apple places a strong emphasis on design, focusing on both aesthetics and functionality. This approach is evident in their minimalist design and user-friendly interfaces.
Quality Control: Apple’s rigorous quality control processes help maintain high standards across all products.
Lessons Learned:
User-Centric Design: Prioritizing the user experience can lead to highly successful products that resonate with customers.
Ecosystem Integration: Creating an interconnected ecosystem enhances product value and customer retention.
Continuous Innovation: Regularly updating and innovating products can maintain a competitive edge.
2. Tesla, Inc.
Case Study: Tesla’s Approach to Product Engineering
Vertical Integration: Tesla controls much of its supply chain, from manufacturing to software development. This control allows for rapid innovation and improvement.
Focus on Sustainability: Tesla’s commitment to electric vehicles (EVs) and renewable energy has positioned it as a leader in sustainable technology.
Rapid Prototyping: Tesla uses rapid prototyping to quickly iterate on designs and test new ideas, which accelerates development cycles.
Innovation through Integration: Vertical integration can provide more control over product development and quality.
Market Disruption: Focusing on emerging trends like sustainability can differentiate a company in a crowded market.
Agile Development: Rapid prototyping and iteration can lead to faster development and more innovative solutions.
3. Amazon Web Services (AWS)
Case Study: AWS’s Product Engineering Strategies
Scalable Infrastructure: AWS focuses on providing scalable cloud computing services that can grow with customer needs. This approach allows customers to pay only for what they use.
Customer Feedback: AWS uses customer feedback to drive product development and prioritize features that meet market demands.
Innovation and Diversity: AWS regularly introduces new services and features, maintaining a diverse product portfolio.
Scalability: Building scalable solutions can address a wide range of customer needs and support growth.
Customer-Centric Development: Using customer feedback to guide product development ensures that offerings remain relevant and valuable.
Continuous Improvement: Regularly updating and expanding product offerings can keep a company at the forefront of technology.
Conclusion
Effective product engineering is crucial for creating products that meet customer expectations, drive innovation, and maintain competitive advantage. By focusing on user-centric design, integrating systems, controlling quality, and staying agile, companies can achieve remarkable success and push the boundaries of technology.
Product Engineering is key to making ideas come to life and meeting customer needs. It’s a crucial part of creating products that are both groundbreaking and reliable, driving success and satisfaction in the long run.
FAQs on Product Engineering
1. what is product engineering, and why is it important for my business.
Product Engineering involves the process of designing, developing, and delivering a product from concept to completion. It's crucial for businesses as it ensures that products are innovative, cost-effective, and market-ready, ultimately helping companies stay competitive and meet customer demands.
2. How does Product Engineering differ from traditional product development?
Unlike traditional product development, which focuses on creating a product based on specifications, Product Engineering encompasses the entire lifecycle, including design, development, testing, and scaling. It also involves continuous improvement based on market feedback and technological advancements.
3. What industries benefit most from Product Engineering services?
Product Engineering services benefit a wide range of industries, including technology, healthcare, manufacturing, automotive, and consumer electronics. Any industry that requires innovative and scalable products can leverage Product Engineering to enhance their offerings.
4. How can Product Engineering help in reducing time-to-market?
Product Engineering optimizes the product development process by using agile methodologies, rapid prototyping, and continuous testing. This approach helps in identifying and resolving issues early, reducing the overall time-to-market while ensuring high-quality products.
5. Can Product Engineering services help with legacy product upgrades?
Yes, Product Engineering services can assist with upgrading legacy products by modernizing them with the latest technologies, enhancing performance, and adding new features. This helps extend the product’s lifecycle and keeps it competitive in the market.
Published on:
September 3, 2024
Featured Resources
App design agency - an ultimate guide for everyone.
Let's explore the world of app design agencies—what they do, why their work is so vital, and how you can choose the right one for your project.
Small Scale Production During Product Prototype Phase
Small scale production refers to manufacturing of a limited quantity of products, generally during the prototype phase of product engineering and development.
Best Cost-Effective UI/UX Design Tips
Discover budget-friendly UI/UX design tips to create stunning digital experiences without breaking the bank. Perfect for beginners and pros alike!
Sketchish is a digital product design and engineering firm committed to addressing intricate software challenges.
Established in 2017, our team has proven record in assisting entities spanning from startups that went public offerings to esteemed Fortune 500 and progressive enterprises.
Plans & Packages
Digital product design
Web application design
Mobile application design
Website design
Landing page design
Digital Marketing
Social media
Core Business
Digital Product Design
Software Engineering
Quality Engineering
About Sketchish
Our clients
Arizona - United States
1730 S EI Camino Dr, Tempe,
Ahmedabad - India (HQ)
401-402, Palak Prime,
Ambli, A’bad - 380054
Copyright © 2024 Sketchish design llp. All rights reserved.
Reviews us on:
Privacy Policy
Terms of use
- Digital Marketing
- Facebook Marketing
- Instagram Marketing
- Ecommerce Marketing
- Content Marketing
- Data Science Certification
- Machine Learning
- Artificial Intelligence
- Data Analytics
- Graphic Design
- Adobe Illustrator
- Web Designing
- UX UI Design
- Interior Design
- Front End Development
- Back End Development Courses
- Business Analytics
- Entrepreneurship
- Supply Chain
- Financial Modeling
- Corporate Finance
- Project Finance
- Harvard University
- Stanford University
- Yale University
- Princeton University
- Duke University
- UC Berkeley
- Harvard University Executive Programs
- MIT Executive Programs
- Stanford University Executive Programs
- Oxford University Executive Programs
- Cambridge University Executive Programs
- Yale University Executive Programs
- Kellog Executive Programs
- CMU Executive Programs
- 45000+ Free Courses
- Free Certification Courses
- Free DigitalDefynd Certificate
- Free Harvard University Courses
- Free MIT Courses
- Free Excel Courses
- Free Google Courses
- Free Finance Courses
- Free Coding Courses
- Free Digital Marketing Courses
20 Product Management Case Studies [Detailed Analysis][2024]
In today’s fast-paced and highly competitive business environment, effective product management has never been more crucial. It is a strategic catalyst that drives innovation and shapes how companies respond to evolving market demands and consumer preferences. This article delves into product management by examining 20 diverse global case studies, each showcasing the profound impact and key learnings derived from some of the world’s most influential companies. From Apple’s groundbreaking entry into the smartphone market to Spotify’s transformation of music consumption, and Toyota’s efficiency-driven Lean Production Model, these case studies offer a panoramic view of how strategic product management can lead to revolutionary changes in various industries. The article aims to provide valuable insights into the challenges faced, solutions implemented, and the overarching effects of these strategies, revealing how companies like Airbnb, Tesla, Zoom, Slack, Samsung, Netflix, and Patagonia have not only achieved market success but also set new benchmarks and trends in their respective domains. Through this exploration, we aim to equip current and aspiring product managers and business leaders with practical knowledge and inspiration to navigate the complex landscape of product management, driving innovation and success in their ventures.
Related: How to Build a Career in Product Management?
1. Apple Inc. – Reinventing the Smartphone
Task/Conflict:
Apple’s entry into the already crowded mobile phone market was a bold move, particularly with the objective of introducing a product that wasn’t just another addition but a complete redefinition of what a mobile phone could be. The challenge was to innovate in a way that would not only capture the market’s attention but also set a new standard for user interaction, functionality, and design in the smartphone industry.
The solution lay in the development of the iPhone, a device that combined a phone, an iPod, and an internet communicator. This integration, coupled with a pioneering touchscreen interface and a focus on user experience, positioned the iPhone not just as a product but as an ecosystem. Apple’s emphasis on design, functionality, and user interface created a product that stood out from its competitors.
Overall Impact:
- Revolutionized the smartphone industry.
- Set new standards for technology and user experience.
Key Learnings:
- Innovation can disrupt established markets.
- User-centric design is crucial in technology products.
2. Spotify – Transforming Music Consumption
In an era dominated by music piracy and declining physical album sales, Spotify faced the daunting task of reshaping how people accessed and paid for music. The challenge was not only technological but also cultural, requiring a shift in consumer habits and a rethinking of the existing music industry’s business model.
Spotify’s approach was to introduce a user-friendly music streaming service, offering a vast library of tracks with both a free, ad-supported model and a premium subscription option. This strategy addressed the issues of accessibility and affordability while respecting the rights of artists and producers, thus presenting an attractive alternative to illegal downloads.
- Influenced the revenue model of the entire music industry.
- Became a leader in music streaming.
- Innovative business models can redefine industries.
- Addressing consumer pain points is key to success.
3. Toyota – The Lean Production Model
Toyota was confronted with the challenge of enhancing efficiency and reducing waste in their production processes. The automotive industry, characterized by intense competition and high operational costs, demanded a strategy that not only improved production efficiency but also maintained high quality.
Toyota implemented the Lean Production Model, a revolutionary approach focusing on ‘Kaizen’ or continuous improvement. This methodology involved streamlining the manufacturing process, reducing waste, and empowering workers to contribute to ongoing improvements. The Lean Model emphasized efficiency, flexibility, and a relentless pursuit of quality in production.
- Enhanced operational efficiency and profitability.
- Established as a benchmark for manufacturing excellence.
- Efficiency and quality are pillars of manufacturing success.
- Continuous improvement drives operational excellence.
4. Airbnb – Revolutionizing Hospitality
Airbnb aimed to carve out a new niche in the hospitality industry, which was traditionally dominated by hotels. The challenge was multifaceted, involving regulatory hurdles, building trust among users, and creating a reliable and scalable platform that connected homeowners with travelers seeking unique lodging experiences.
The solution was the creation of a user-friendly online platform that enabled homeowners to list their properties for short-term rental. This platform not only provided an alternative to traditional hotels but also fostered a sense of community and unique travel experiences. Airbnb focused on building a robust review system and transparent policies to overcome trust and safety concerns.
- Disrupted the traditional hotel industry.
- Became a leading figure in the sharing economy.
- Innovative platforms can create new market segments.
- Trust and transparency are crucial in community-driven businesses.
Related: History & Origin of Product Management
5. Tesla – Electrifying the Auto Industry
Tesla embarked on the ambitious goal of popularizing electric vehicles (EVs) as a sustainable and viable alternative to gasoline-powered cars. This task involved overcoming preconceptions about the performance, range, and practicality of EVs, as well as establishing the necessary infrastructure for their adoption.
Tesla’s approach was to develop high-performance, luxury electric vehicles that combined environmental friendliness with cutting-edge technology and stylish design. This strategy helped to change the perception of EVs from being seen as inferior alternatives to gasoline cars to desirable, high-tech vehicles. Tesla also invested in building a network of charging stations, further facilitating the practicality of EV ownership.
- Led the transition towards electric vehicle adoption.
- Influenced the auto industry’s direction towards sustainability.
- Sustainable technology can be aligned with luxury and performance.
- Changing consumer perceptions is key to introducing new technology.
6. Zoom – Simplifying Remote Communication
In a market crowded with various communication tools, Zoom faced the challenge of differentiating itself and proving its value. The goal was to provide a solution that was not only reliable and easy to use but also superior in terms of video and audio quality compared to existing offerings.
Zoom focused on creating a user-friendly platform that offered high-definition video and clear audio, even in low-bandwidth situations. This commitment to quality and reliability, combined with features like screen sharing, virtual backgrounds, and easy integration with other tools, made Zoom a preferred choice for businesses and individuals alike, especially during the COVID-19 pandemic.
- Became a staple tool for remote communication.
- Highlighted during the global shift to remote work due to the pandemic.
- Reliability and user experience are critical in technology solutions.
- Agility in adapting to market changes is vital.
7. Slack – Redefining Workplace Collaboration
Slack was developed with the vision of transforming the cluttered and inefficient landscape of workplace communication, dominated by email. The challenge was to create a platform that not only streamlined communication but also integrated various work tools to enhance productivity and collaboration.
The solution was an intuitive, chat-based platform that allowed for real-time messaging, file sharing, and integration with a wide range of work tools and applications. Slack’s focus on reducing the reliance on emails and consolidating communication into a single, searchable platform revolutionized team collaboration and internal communication in businesses.
- Changed the dynamics of team communication and collaboration.
- Became a central tool in many organizations for internal communication.
- Streamlining common practices can create significant market opportunities.
- Integration and user-friendliness are key in collaborative tools.
8. Samsung – Innovation in Electronics
Samsung’s challenge was to establish itself as a leader in the highly competitive and rapidly evolving consumer electronics market. This required keeping up with technological advancements and differentiating its products in terms of quality, innovation, and user experience.
Samsung’s strategy involved substantial investment in research and development, focusing on bringing innovative and high-quality products to the market. Their innovation commitment spanned various product categories, including smartphones, televisions, and home appliances. This focus on quality and technological advancement helped Samsung achieve a leading position in the global electronics market.
- Achieved a leading position in the consumer electronics market.
- Known for innovation and quality in product offerings.
- Innovation is crucial in technology sectors.
- Quality and continuous improvement attract consumer loyalty.
Related: Top Product Management Tools
9. Netflix – Pioneering Streaming Services
Netflix’s journey began with the goal of transforming the traditional movie rental business. The challenge was to transition from a DVD rental service to an online streaming platform, requiring a technological shift and a change in consumer viewing habits and content distribution models.
The solution was a gradual but determined shift to an online streaming model, offering customers an extensive and ever-growing library of movies and TV shows. Netflix’s investment in original content and exclusive deals with production studios further enhanced their appeal. This strategic pivot catered to the growing demand for on-demand entertainment, free from physical media and broadcast schedules constraints.
- Redefined media consumption habits.
- Led the rise of online streaming services.
- Adaptability to technology and market trends is critical.
- Investing in original content can differentiate streaming services.
10. Patagonia – Ethical Product Management
In a clothing industry often criticized for environmental and ethical issues, Patagonia aimed to differentiate itself by committing to sustainability and ethical practices. The challenge was not only to maintain profitability but also to influence consumer behavior and industry standards towards more responsible practices.
Patagonia’s approach included using sustainable materials, ensuring transparency in their supply chain, and advocating for environmental causes. Their commitment extended to initiatives like repairing products to extend their lifespan and encouraging responsible consumption. This strategy appealed to environmentally conscious consumers and set a new standard for corporate responsibility in the clothing industry.
- Became a model for sustainability in the clothing industry.
- Influenced both consumer and industry practices towards eco-friendliness.
- Sustainability can be a unique selling proposition.
- Ethical practices enhance brand loyalty and reputation.
11. Microsoft – Shifting to Cloud Computing
Microsoft faced significant challenges in adapting to the rapidly evolving technology landscape. The traditional software model of boxed products had grown increasingly obsolete due to a surge in cloud computing. Emerging competitors like Amazon Web Services and Google’s cloud platform gained momentum, providing flexible, scalable solutions that shifted the market’s preference away from on-premise software to on-demand, subscription-based models. Microsoft needed to transform its business approach and product portfolio to align with these market trends
Under CEO Satya Nadella’s leadership, Microsoft shifted focus to cloud computing, developing Azure as an end-to-end platform providing comprehensive infrastructure and software services. The company also transitioned its flagship Office suite to a cloud-based subscription model with Office 365. They emphasized flexibility, scalability, and security while ensuring seamless integration with existing Microsoft products. Investments in data centers globally and new pricing models enabled Microsoft to compete directly with other leading cloud providers.
- Transformed Microsoft into a leader in cloud computing.
- Significantly increased recurring revenue through subscription-based services.
- Implementation of emerging technologies is vital for staying ahead of market trends.
- Subscription models can create predictable and sustainable revenue streams.
12. Lego – Rebuilding a Toy Empire
Lego was at a crossroads in the early 2000s. The company had overextended its product lines, ventured into unrelated business areas, and faced fierce competition from digital entertainment sources like video games. The result was a decline in sales and profitability, jeopardizing the company’s future and threatening the iconic brand with irrelevance.
To rebuild its brand, Lego implemented a back-to-basics approach, refocusing on its core product, the Lego brick. It also streamlined its product lines and improved internal operations. Partnering with entertainment franchises such as Star Wars and Harry Potter, they launched themed Lego sets that resonated with younger generations. Lego expanded its reach into digital media with video games and movies like The Lego Movie, engaging customers through multiple channels and breathing new life into the brand.
- Restored profitability and renewed consumer interest in Lego products.
- Expanded their presence into digital media and entertainment.
- Diversification and partnerships can revitalize traditional products.
- Engaging customers across multiple channels strengthens brand loyalty.
Related: Inspirational Product Management Quotes
13. Dropbox – User-Friendly Cloud Storage
Dropbox faced the challenge of competing with tech giants including Google and Microsoft in the nascent cloud storage market. While these companies offered vast storage solutions integrated with their productivity suites, Dropbox needed to carve out a niche by appealing to users with an easy-to-use, reliable platform. They aimed to provide seamless file synchronization, security, and accessibility across devices.
Dropbox placed simplicity at the forefront, developing a cross-platform application that allowed users to sync files effortlessly across multiple devices. The system’s seamless synchronization and ease of use differentiated it from other cloud storage providers. They employed a freemium model that offered free storage with the option to upgrade for more capacity and features, attracting millions of users globally and enabling them to monetize their growing user base.
- Became a trusted name in cloud storage, with millions of users worldwide.
- Pioneered the freemium model, offering free and paid plans.
- User experience is a differentiator in competitive tech markets.
- Freemium models can attract users and convert them to paid subscriptions.
14. Nike – Personalizing Athletic Wear
Nike, already a leader in sports apparel, faced stiff competition from rivals like Adidas and Under Armour. The company needed a unique strategy to differentiate its products and capture the loyalty of a diverse, increasingly demanding customer base. Customers wanted personalized experiences, and Nike aimed to address this by providing a solution that matched their specific preferences in athletic wear.
Nike launched the NikeID program, which allowed customers to personalize their athletic gear online, choosing colors, patterns, and custom text. This innovation expanded the company’s appeal to athletes and fashion-conscious consumers alike, helping them express their individuality while boosting engagement. By streamlining the customization process and leveraging digital technology, NikeID created an experience that could be replicated globally, resulting in increased brand loyalty and revenues.
- Elevated customer engagement through personalized experiences.
- Expanded customization to a broad range of products, increasing brand loyalty.
- Personalization can differentiate brands in competitive markets.
- Engaging customers in the design process enhances brand value.
15. Procter & Gamble – Open Innovation with Connect + Develop
Procter & Gamble (P&G), known for a vast portfolio of consumer goods, recognized that the traditional R&D process was becoming slower and costlier, hampering the company’s ability to innovate. With the proliferation of specialized knowledge worldwide, P&G realized that internal expertise alone wouldn’t suffice fulfill the increasing demand for new products across its various brands. They needed to find a way to tap into external innovation to stay ahead of the competition.
P&G launched the Connect + Develop platform, an open innovation initiative that invited inventors, academics, and other companies to submit ideas and collaborate on new products. This platform enabled P&G to access global expertise and accelerate the product development process by integrating external solutions with their own internal capabilities. The platform generated new partnerships that broadened P&G’s R&D reach and enhanced the product pipelines for various brands, significantly improving efficiency and innovation.
- Increased innovation by sourcing solutions from a global network.
- Enhanced product pipelines across multiple categories.
- Open innovation can tap into global expertise for improved R&D.
- Collaborating beyond company boundaries accelerates product development.
16. Adobe – Transforming into a Subscription Model
Adobe faced challenges with its traditional perpetual software licensing model, which was becoming outdated due to issues like piracy and inconsistent revenue streams. As competitors moved towards more dynamic, subscription-based models, Adobe needed to reinvent its business strategy to stay competitive and relevant in the digital content creation industry.
With the introduction of Adobe Creative Cloud, Adobe shifted from selling boxed software to a subscription-based model. This move provided customers with constant updates, cloud storage, and access to a suite of creative tools for a monthly fee. The transition addressed piracy issues and allowed Adobe to offer a scalable and continually improving product experience, leading to a more predictable and stable revenue stream.
- Stabilized Adobe’s revenue with a predictable subscription-based income.
- Increased customer retention and satisfaction due to continuous updates and enhancements.
- Fostered a broader adoption of Adobe’s software suite among freelancers and small businesses due to more accessible pricing.
- Transitioning to a subscription model can provide stable revenue and reduce piracy.
- Offering continual improvements and added value can enhance customer loyalty.
Related: Reasons to Study Product Management
17. GoPro – Innovating in a Niche Market
GoPro aimed to dominate the action camera market but faced the challenge of distinguishing itself from larger electronics manufacturers with broader product lines. The company needed to innovate continuously while fostering a strong brand identity that resonated with extreme sports enthusiasts and casual users alike.
GoPro focused on developing durable, high-quality cameras with unique features such as waterproofing and compact design tailored to capture extreme sports and adventure. They also built a robust community by leveraging user-generated content and social media, turning their customers into brand ambassadors. This strategy solidified their market position and expanded their customer base.
- Established GoPro as the leading brand in action cameras with a significant market share.
- Expanded the brand’s appeal beyond extreme sports to general consumers.
- Fostered a new market for accessory and lifestyle products related to action cameras.
- Leveraging user-generated content can effectively enhance community engagement and marketing.
- Creating an ecosystem around a product can extend its market reach and usability.
18. IBM – Pioneering Artificial Intelligence with Watson
IBM recognized the potential of artificial intelligence early on but faced the dual challenge of developing cutting-edge technology and finding practical applications for AI in business. They needed to create a platform that could demonstrate AI’s capabilities and be applicable and beneficial across various industries.
IBM developed Watson, an AI system capable of understanding natural language and generating data-based hypotheses. Watson was first introduced to the public by participating in the quiz show Jeopardy!, where it challenged humans. Following this, IBM expanded Watson’s capabilities to serve industries like healthcare, finance, and customer service, showcasing its versatility and practical utility.
- Expanded Watson’s applications into healthcare, finance, and beyond, proving AI’s versatility in solving complex problems.
- Strengthened IBM’s brand as an innovator and thought leader in the technological space.
- Demonstrating technology through high-visibility challenges (like Jeopardy!) can effectively capture public and commercial interest.
- Strategic partnerships in diverse industries can enhance the practical applications and market acceptance of new technologies.
19. Unilever – Sustainability as a Business Strategy
Facing increasing consumer awareness and demand for sustainable and ethical products, Unilever needed to integrate sustainability deeply into its business model without compromising on profitability and market competitiveness.
Unilever launched the Sustainable Living Plan, committing to halve its environmental footprint, improve health and well-being for more than a billion people, and sustainably sourcing 100% of its agricultural raw materials. This comprehensive strategy helped Unilever strengthen its brand loyalty among conscious consumers and drove long-term growth by reducing costs and innovating in product development.
- Achieved cost reductions and efficiency improvements through sustainable practices.
- Set industry standards for sustainability, influencing other companies to adopt similar practices.
- Sustainability can drive business growth and consumer loyalty when integrated into core business strategies.
- Ethical practices can be a competitive advantage, attracting both consumers and investors.
- Transparency in sustainability efforts can enhance corporate reputation and build stronger relationships with stakeholders.
20. Zara – Revolutionizing Fashion with Fast Fashion
Zara, part of the Inditex group, needed to maintain its edge in the highly competitive and fast-paced fashion industry. The challenge was to continually offer the latest fashion trends faster than traditional retailers, addressing the consumers’ desire for immediate gratification.
Zara implemented a unique business model, fast fashion, which involves rapid prototyping, small batch production, and an extremely efficient supply chain that can bring designs from the runway to store shelves in weeks. This approach kept inventory costs low and ensured that Zara’s offerings were always fresh, appealing, and aligned with current trends.
- Enabled Zara to become a global leader in the fashion industry, significantly outpacing competitors in responsiveness to fashion trends.
- Reduced unsold inventory and increased profitability through efficient supply chain management.
- Catalyzed shifts in consumer buying behavior, with more frequent purchases and higher expectations for rapid trend availability.
- Speed and agility in product development and supply chain can significantly enhance market responsiveness.
- Continuous market research and rapid response to consumer trends are crucial for maintaining competitive advantage in fast-paced industries.
Related: Product Management Failure Examples
Closing Thoughts
In conclusion, these case studies exemplify the transformative power of effective product management. They highlight the importance of understanding market needs, embracing innovation, focusing on user experience, and the value of ethical practices. Aspiring business leaders can draw valuable lessons from these examples to navigate challenges and drive success in their endeavors.
- How Much Equity Should a CFO Get? [2024]
- Top 20 ESG and Sustainability Skills to Add to Your Resume [2024]
Team DigitalDefynd
We help you find the best courses, certifications, and tutorials online. Hundreds of experts come together to handpick these recommendations based on decades of collective experience. So far we have served 4 Million+ satisfied learners and counting.
Career in Product Marketing vs. Product Management: Pros and Cons [2024]

Why are AMPs (Advanced Management Programs) Expensive? [2024]

20 Pros & Cons of being a Cybersecurity Manager [2024]

10 Tips on How to Dress Well as a Manager [2024]

Are certificate programs helpful for top management? [2024]

Is Supply Chain Management a Good Career Option? [2024]
Journey Mapping: A Product Development Process Case Study
Product teams should regularly assess the product development process itself. Sebastian Gherman, a Toptal senior product manager, outlines his approach.
Underpinning every successful product is a successful product development process. As a senior product manager at Toptal, I’ve found that treating that process as a product in its own right results in measurable improvements that touch every facet of our work.
The team I lead comprises an engineering manager, nine software engineers , and one quality assurance engineer. Our work covers a wide spectrum of products and features that facilitate a healthy supply-and-demand balance for our talent network. In the sections that follow, I share how we evolved our product development process using customer journey mapping and arrived at greater efficiency, communication, and collaboration.
When the Process Is the Product, the Team Is the User
Products that are unsuccessful or ineffective often result from what a team, or even a single person, thinks users want and need, not what they actually want and need. A good product, however, is built on the qualitative and quantitative data derived from extensive user research sessions. Likewise, the product development process itself can be unsuccessful or ineffective when it is designed by a leader who assumes they know what their teams need.
As product manager or team lead, you should engage in the same kind of user research that you would carry out for a product—shadowing, interviewing, and surveying—with your team to make sure your process is similarly successful. The goal is to understand how your team is using the product development process and address any pain points they encounter along the way.
Build a Customer Journey Map
There are various ways to collect feedback from users, but customer journey mapping is the product discovery technique I use with my engineers. The result is a diagram that illustrates the steps users go through when engaging with your company, whether that be through a product, online experience, retail experience, service, or any combination of these. The more touchpoints your users have, the more complicated—and therefore necessary—a map becomes.
This technique explores users’ actions and emotions around that engagement to reveal pain points and opportunities. It’s an excellent way to uncover problems in your process.
Within the diagram, users are depicted as hypothetical personas. Each persona should have a short bio, including a description of their inner motivations and responsibilities, as this helps to humanize them. Each persona should represent a key type of user to offer a sense of the diverse wants and needs the solution must address.
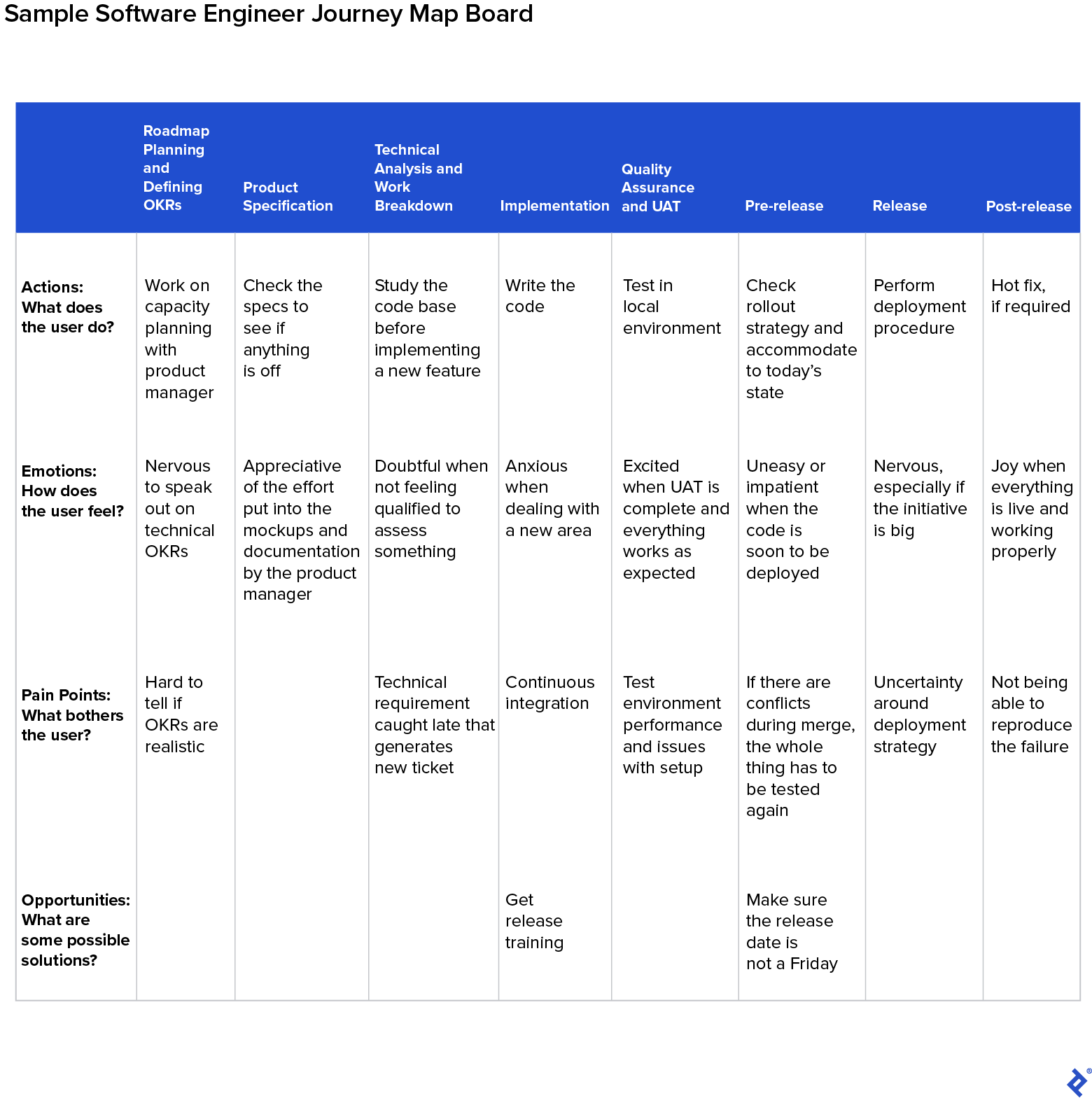
Journey maps are organized by user stages. Each stage represents a major goal the user is trying to achieve in their overall journey. For each stage, and for each persona, ask your team to consider:
- Actions: What does the user do?
- Emotions: How does the user feel?
- Pain Points: What bothers the user?
- Opportunities: What are some possible solutions?
Asking the team “What bothers you about this product and how can we fix it?” would not be a useful way to gather information because, at the time the question is asked, they may not recall use cases or how they felt when they experienced an issue. Asking them to split the interaction into steps and asking them how users encounter each step helps the team surface the emotions associated with each stage of the journey.
Applying This Theory to Our Toptal Team
To understand how this theory applies in action, consider the journey map for the product development process that I created with my engineering team.
Using Miro , I created the journey map board, splitting the product development process into eight major stages:
- Roadmap Planning, and Defining Objectives and Key Results (OKRs)
- Product Specification
- Technical Analysis and Work Breakdown
- Implementation
- Quality Assurance and User Acceptance Testing (UAT)
- Pre-release
- Post-release
I chose two personas—software engineer and product manager—as these are the main users who engage with the process.
- Sergey, the software engineer: Sergey ensures the initiatives are delivered on time and to a high standard, while maintaining a robust code base and understanding of the latest technologies and tools.
- Matt, the product manager: Matt ensures the team prioritizes its efforts by working on the most impactful initiatives first. He also listens to stakeholder needs and communicates updates to the team regularly.
Prior to the session, I filled out the journey map for Matt, the persona in my role, in order to get an idea of how much time was required to complete the exercise, as well as to set the team’s expectations of the format. Next, I scheduled two 90-minute sessions across two consecutive days to ensure my team had enough time to complete the exercise without losing focus or energy. Because most engineers are unfamiliar with the journey mapping process, I shared links to the Miro board and a YouTube tutorial to help them prepare. Before the beginning of the first session, I confirmed that everyone understood the concepts.
As facilitator, I asked the team to suggest the actions, emotions, pain points, and opportunities for Sergey’s persona. Some team members were shy at first, but once a few people shared their thoughts, the session started to flow. I filled out cards on the Miro board based on their input.
Key Learnings From the Journey Mapping Process
The journey mapping process yielded five main takeaways:
- Keep the sessions short and focused. If there are more than a few stages within the journey map, I advise splitting the effort into two or three sessions to maximize productivity and to prevent team members from losing focus.
- Be a role model. Filling out the Product Manager swimlanes before the session sets a tone of honesty and openness, and demonstrates how to express these issues, encouraging team members to share their own emotions and pain points more readily.
- Create emotional safety. Team members may find it intimidating to share their struggles—most likely from a fear of being judged—but try your best not to intervene. Sooner or later, a more courageous team member will break the ice and things will start moving. When that happens, show empathy and appreciation. This will reassure other members that they are in a safe environment and they will feel more comfortable sharing their thoughts.
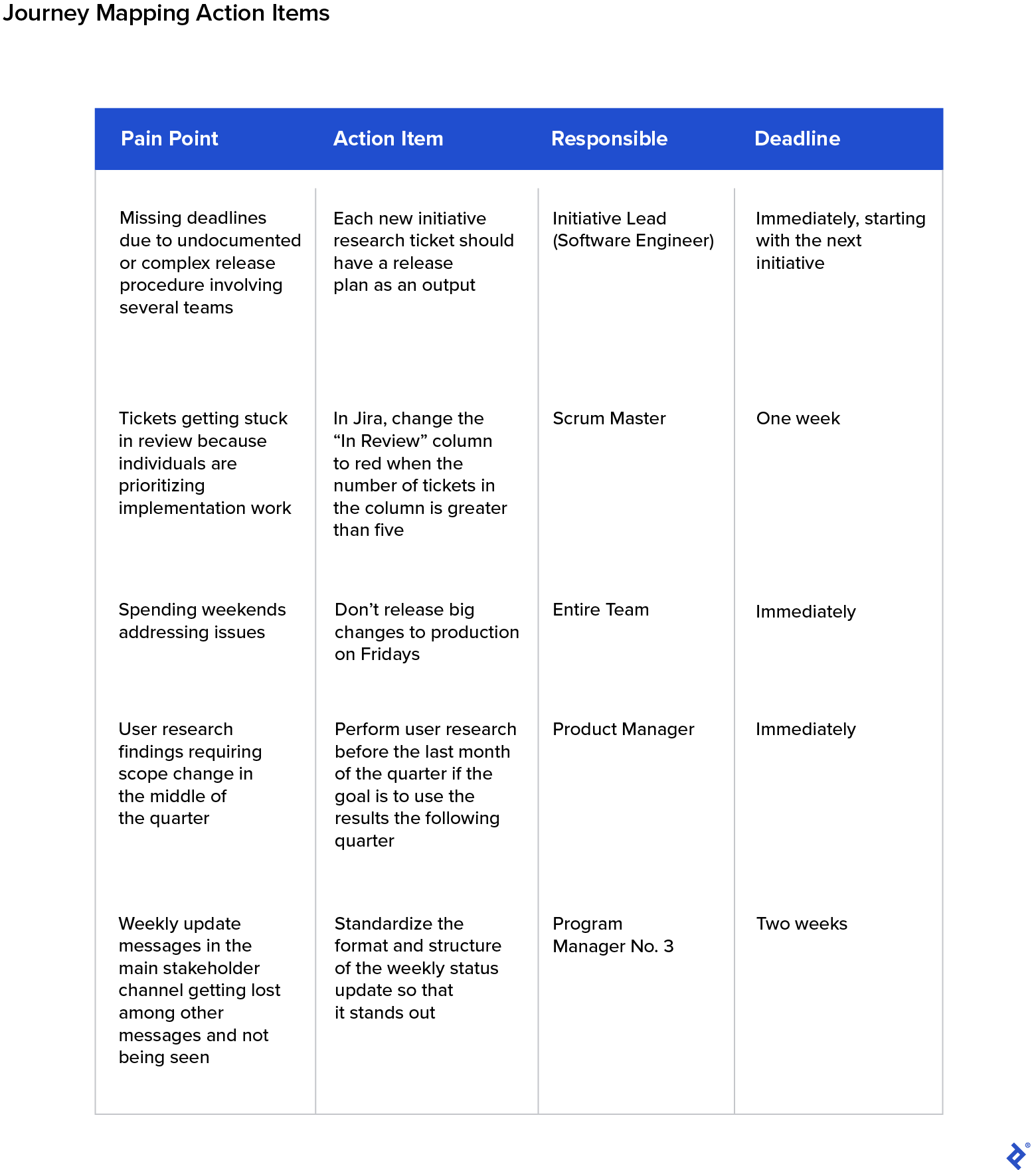
- Create a follow-up plan with your team. Some problems may be hard to solve, especially if the solution involves other teams or departments, but plan to keep your team updated about any relevant communication with, or changes from, those responsible parties who may impact the results of the journey mapping process.
- End with action steps. Create a list of action items, and assign an owner and deadline to each, which will help you realize tangible results from the session. Some examples that resulted in our case are depicted in the following table:
Why Was the Journey Mapping Exercise Effective?
The journey mapping exercise was extremely successful in presenting potential opportunities for improvement and fostering team spirit. It helped us in the following ways:
- It uncovered issues where I believed things were running smoothly and reinforced the importance of not making assumptions. For example, I assumed that everyone had sufficient training on Jira , which was not the case. On the other end of the spectrum, I thought asking the engineering team to record demo videos for new pieces of functionality burdened them, when in fact they valued the exercise because it helped them improve their presentation techniques and lessened their anxiety around being in front of a camera.
- It illuminated some improvements I could make, such as restructuring initiative cover pages to make them more accessible for engineers.
- It empowered the engineering team to take responsibility for the outcomes within their control because they were the ones proposing changes that they could test and further iterate. It was primarily a bottom-up process.
- It revealed that the pain point hot spots were predominantly around roadmap planning and implementation.
- It forged stronger working relationships among the team by acknowledging shared challenges. For example, a number of individuals on our team thought they were the only ones struggling with the CI/CD pipeline for a particular subsystem when, in fact, most of the team was struggling.
Scaling Considerations
If every product manager or team lead for engineering goes through this process with their team, a common set of problems will likely arise, indicating which issues should be addressed first. Teams should follow the updated process for a few months, then the feedback loop must be revisited again. This cycle should continue until the product development process is natural and easy, and supports the needs of the users in building top-quality software products.
In the case of my team, our new process has delivered tangible improvements on several fronts:
- The average time for tickets in review has been reduced by 22%.
- The product OKR completion rate has risen above 90% over the course of the last three quarters.
- The service-level agreement time for high-priority bugs has been met in 100% of cases.
- There have been no failed releases due to deployment problems.
- The average number of post-release reported bugs has decreased by 37%.
If your team is involved in building products , then your process should be subject to continuous scrutiny and improvement. If one function is not performing well, or if its product development process is weaker, that will impact the end result. While I used this practice for an engineering team, it can easily translate to user research , design, UI/UX , and content teams.
Your product development process is your most important product. Use this exercise to help perfect it, and see how much it elevates every product your team makes.
World-class articles, delivered weekly.
Understanding the Basics
What is a product development plan, what is a journey map used for, what are the elements of a journey map.

Join the Core Team
We’re growing quickly and always on the lookout for the best people to join our team.
We work hard to review every application, and we’ll reach out to you if you’re a great match.

Start Reading
Inside Toptal’s Product Team

- Product Strategy
- Performance Engineering and Testing
- Product Engineering and Development
- Application Modernization
- QA Engineering
- Cloud Advisory Services
- Architecture Reviews
- Migration and Modernization
- DevOps Engineering
- Data Engineering
- Big Data Analytics
- Data Science
- Machine Learning
- Custom Software Development
- Application Development
- Financial Services
- Healthcare and Life Sciences
- Retail and E-commerce
- Supply Chain & Logistics
- Case Studies
- AI & ML Insights
- Product Engineering Insights
- Cloud Engineering Insights
- News and Tech Insights
A Deep Dive into Product Engineering: Process, Roles, and Best Practices
Transform your business ideas into functional products with efficient product engineering
Table of Contents
What is product engineering, product engineering process, roles and responsibilities of a product engineer, product engineering techniques and tools.
Every business needs a product; without it, it’s difficult to sustain.
Product engineering helps develop those functional products from ideas. It provides a framework for software development, and offers a competitive edge to businesses through innovations like building a chatbot like “ ChatGPT ” for your business and integrating it into existing applications.
Product engineering helps define the development process, key roles, and responsibilities and thus understanding it is crucial.
Let’s dive in and explore the fascinating world of product engineering!
Simform is a leading software product engineering service that can help you enhance user experience and improve business agility. Our team of experts leverages product engineering best practices to enhance performance for your operations and improve ROI. Contact us now for imformation.
Product engineering is a process of applying engineering principles, design thinking, and project management to improve user experience, software quality and enable businesses to deploy high-performance systems.
Product engineering provides key benefits of cost-effectiveness, quality improvement, and enhanced security. It includes several processes designed to offer a granular insight into product development.
Additionally, product engineering provides a roadmap for an efficient software development lifecycle (SDLC).
The product engineering process has several stages, from conceptualization to launch and deployment. Each stage differs from organization to organization based on specific business requirements.
#1. Product discovery
Product discovery is a phase where you research different aspects of software, understand the gaps in the market, and discover the need for a product. You can conduct surveys to understand market demands.
Such surveys also allow you to analyze whether the product is a market fit. You must segment the market per product niche and research specific customer demands. Here are some of the approaches you can take for market research,
- One-on-one interviews- Conduct interviews with potential customers and industry experts to understand the pain points and challenges faced by customers.
- Online surveys- Use the internet to conduct surveys and ask customers about their expectations of your product. This will give you a broader perspective on the market and help gauge the level of interest in your product.
- Focus groups- Build focused groups that test your product and provide feedback on the gaps in user experience.
- Competitor analysis- Observe competitors and analyze their products to identify the gaps in the market and understand what makes your product unique.
- Price-to-value comparison- Compare products in the market, their pricing, and yours to understand how much value your product offers.
Based on your analysis, you will be able to determine the feasibility of the product. Once you have checked the feasibility of the product, it’s time to plan the development. This will include design requirements, production roadmap, and resource and process planning.
#2. Ideation and planning
Product ideation uses insights from the discovery stage to create a roadmap outlining the features, functionalities, and processes for product development. For example, if you are developing an eCommerce application, you must ideate features that improve the shopping experience.
The ideation and planning phase also includes consideration of factors like,
- Scalability- Plan for accommodating growing user base during development
- Security- Ideate security policies and ensure better compliance
- Target audience- Identify the target customer and design features that address their pain points.
- Cost – Consider the budget for developing the product and optimizing it.
Moreover, to build the product, you must plan for development resources, design requirements, tools, and infrastructure.
Based on the product development plan, create a roadmap that will include,
- What resources will you need to develop the product?
- How will you develop the product? (Processes required)
- What will be the product design and architecture?
- What type of tests will you need for the product testing?
- What are the environments in which we will deploy the product?
Once the roadmap is ready, the next stage is to design the product.
#3. Product design
Designing your product requires profoundly understanding your target customers and carefully considering feasibility, functionality, and performance requirements. An effective way to product design is the ”design thinking” approach.
The design thinking approach has five stages,
- Empathize with your target customers and understand their needs, pain points, and goals.
- Define the core problem your product will solve based on customer pain points.
- Ideate solutions that can solve the defined problem
- Prototype, create low-fidelity prototypes to test and refine your idea
- Test your prototypes to gain insights into design requirements.
Design thinking helps you create a comprehensive product design and test it for feasibility. However, design architecture is another crucial aspect of product design that determines how different elements of your software will interact.
It provides structure to different elements and processes that form the core of your software. For example, the microservice architecture allows you to leverage independent services communicating through APIs.
Once your design architecture is in place, you can develop your software product.
#4. Product development
Product development is a broader term encompassing everything from ideation to the product’s final launch.
This section focuses on the development stage of product engineering, which starts with creating a design wireframe of the product. Stakeholders and users provide feedback on the wireframe, and based on the design architecture and feedback, you can create a minimum viable product (MVP) with minimal required features..
MVP offers several benefits, particularly when using an iterative development approach. These include,
- Increased business focus and clarity
- Customer-centric product design
- Improved feedback integration
- Reduced errors and performance bottlenecks
- Ability to refine products with each iteration
MVP testing and feedback integration are critical to enhancing product development, and testing mechanisms should be in place before deployment to ensure a bug-free experience.
#5. Product testing
Product testing is a stage where your software is tested, and results are shared with the developers to remove errors. The testing phase requires planning to identify the types of tests to be conducted, resources needed, and quality assurance requirements.
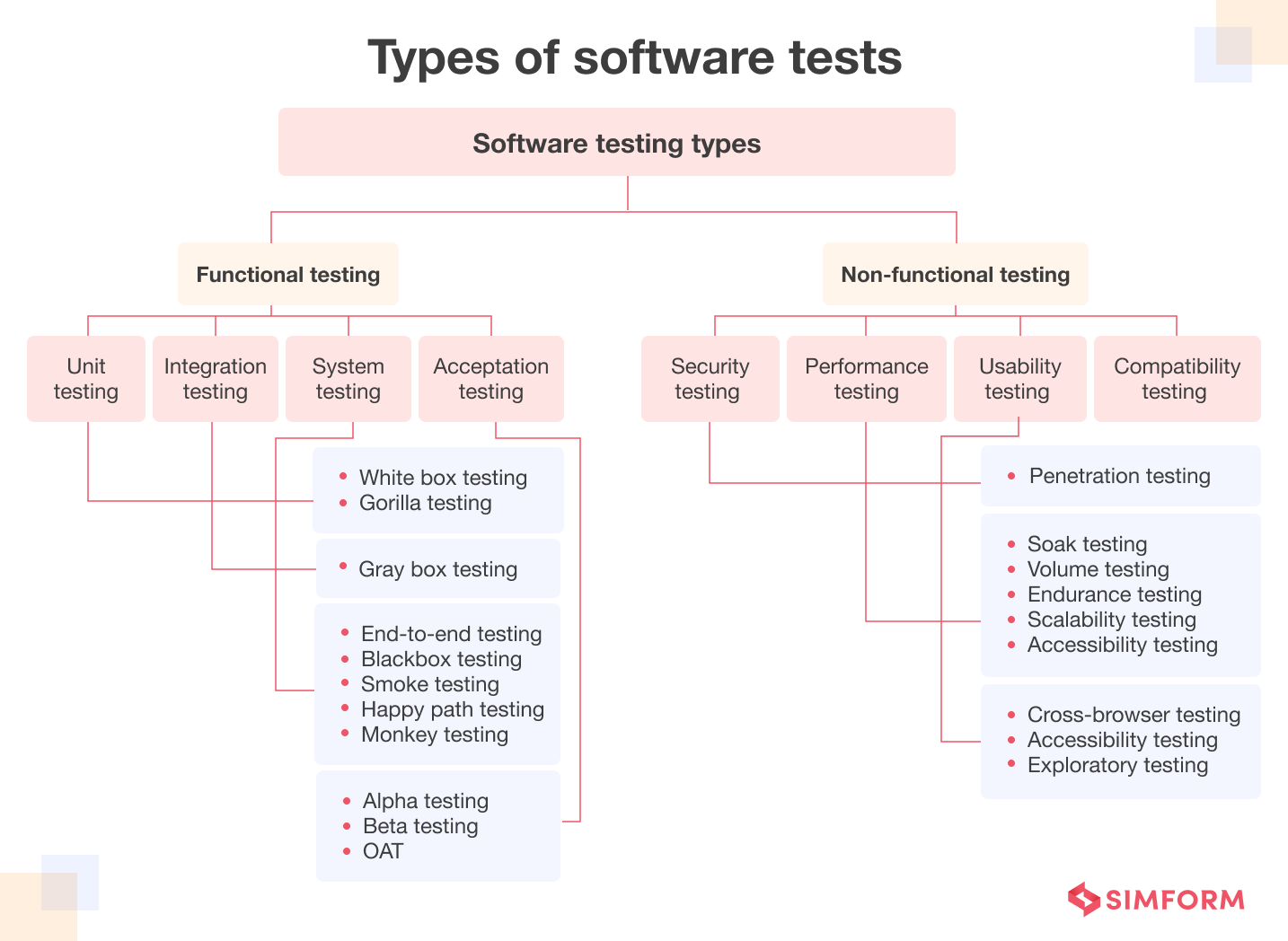
Software product testing begins by identifying multiple test types:
- Unit testing tests each component of the products as an individual unit in two ways: “White box testing,” where the source code or internal structure code is visible to the tester, and “Black box testing,” where the source code is hidden from the testers.
- Integration testing determines how different software modules work together and whether they communicate as expected.
- System testing verifies whether the application meets all specified requirements in different scenarios.
- Acceptance testing is a test to check whether the software meets customer expectations.
- Regression testing ensures that changes to the software do not break the application due to backward compatibility issues by performing functional and non-functional tests across previous software versions.
- Performance testing evaluates the software’s performance under different conditions, such as higher loads, limited system resources, etc.
- Security testing ensures that the software is secure from unauthorized access and that sensitive data is protected. It can include vulnerability assessments, penetration testing, and other security-related tests.
- Usability testing ensures that software is user-friendly and meets the end user’s needs. It can involve user surveys, user interface testing, and feedback sessions.
- Exploratory testing tests the software without predefined test cases or scenarios by exploring the system and attempting to find bugs or issues that other types of testing may have missed.
#6. Deployment and maintenance
After product testing, the deployment stage becomes crucial. Configuring the deployment platforms and ensuring the product meets customer expectations is necessary.
Higher deployment efficiency is one of the key benefits of product engineering, as it provides a framework for deploying products across environments and ensuring high performance.
The product engineering process completes its cycle with the maintenance stage, where you must monitor the performance and pre-defined metrics.
Monitoring and logging key events while the user interacts with the product helps enhance observability, which is crucial for better maintenance. Defining roles and responsibilities at each product engineering stage is also essential.
Product engineering is a process, and the product engineer is the processor! A product engineer connects the production and development teams to build efficient operations. They help organizations develop reliable and resilient products with process efficiency at each stage of software development.
Who is a product engineer?
A product engineer takes ownership of designing, developing, and implementing engineering principles to deliver products that meet customer demands with a high focus on user experience and customer satisfaction. A clearly defined role for a product engineer ensures efficient product delivery.
What do product engineers do?
A product engineer has multiple responsibilities, from creating a design roadmap to development planning and execution.
Product engineers’ role in designing
Product engineers create a design roadmap that includes
- Design architecture,
- Process of designing
- Validation hierarchy,
- Approval standards,
- Post-approval execution plan
Due to their customer-centric approach, product engineers ensure that design aligns with user expectations and the organization’s goals.
Product engineers’ role in software development
Product engineers stress the importance of quality and functionality through an efficient process. They are involved in development planning, process workflow creation, and project execution.
So, a product engineer,
- Defines key processes in software development
- Applies optimal frameworks for enhanced product performance
- Adopts an iterative approach with agile methodologies
- Develops MVP and test prototypes to improve deliverable quality
- Standardizes the development process across teams when implementing microservices
- Implements software engineering principles for highly reliable product development
The software development phase needs the implementation of different best practices and approaches. An experienced product engineer enables organizations to deliver highly resilient products by implementing suitable engineering approaches.
Product engineers’ role in software delivery
Software delivery involves defining requirements, designing to specifications, and developing and deploying the product. One of the critical aspects of software delivery is implementing continuous improvement in product development and delivery.
Product engineers ensure enhanced software delivery by,
- Gathering customer requirements and feedback, conducting market research and data analysis
- Creating wireframes, mockups, and prototypes of product design as per requirements
- Collaborating with cross-functional teams embracing DevOps culture
- Implementing CI/CD pipeline for continuous improvement
- Ensuring error-free performance through optimized monitoring and logging
Skillset of product engineers
To be an effective communicator and collaborator, a product engineer needs to possess a specific set of skills that enable them to,
- Communicate effectively
- Build relationships
- Work collaboratively
These skills include,
- Ability to explain complex technical concepts in simple language.
- Capability to build relationships, manage conflict, and facilitate effective communication among team members.
- Think creatively, and develop innovative solutions to complex problems.
- Manage projects effectively, including developing project plans, setting milestones, and tracking progress.
- Have a strong background in engineering, mathematics, and science.
- Know the latest market trends, processes, new approaches and design techniques.
Product engineering is essential to ensure software quality, and implementing its principles can be far more efficient with specific techniques and tools.
Several techniques can help in this process, such as prototyping, user experience design, software development life cycle (SDLC) optimization, and agile methodology.
Product engineering best practices
1. leverage agile methodologies for rapid product development ..
In the IT market, the timing of the product is crucial. Agile is an excellent product engineering technique if you want to get your product in the market fast without compromising quality.
The agile methodology enables organizations to make quick changes during product development and use an iterative process to enhance software delivery.
Implementing product engineering principles and leveraging essential techniques like agile methodology or optimizing SDLC requires reliable tools.
2. Use prototyping to ensure reduced cost and higher quality.
Prototyping involves creating smaller versions of products to ensure that the final product meets customers’ expectations.
Benefits of prototyping include
- Improving the understanding of product design across teams
- Ensuring the development of optimized products
- Reducing the errors and time needed to rectify them
3. Design the product for enhanced UX.
User experience (UX) design aims to create an easy-to-use, functional, and interactive product. This technique involves gathering data from customer-facing teams to create a design that addresses user problems and pain points.
Steps to follow for enhancing user experience,
- Conduct thorough research on target users’ needs, preferences, and pain points.
- Keep your design consistent and in alignment with the user’s preferences
- Add more whitespace for visual hierarchy, contrast, and balance to the overall UI look
- Use coherent color scheme, typography, iconography, and layout for the unity of elements
- Optimize micro-interactions, animations, and loading indicators for a better experience
- Integrate feedback from users on the design aspects
4. Optimize stages of SDLC.
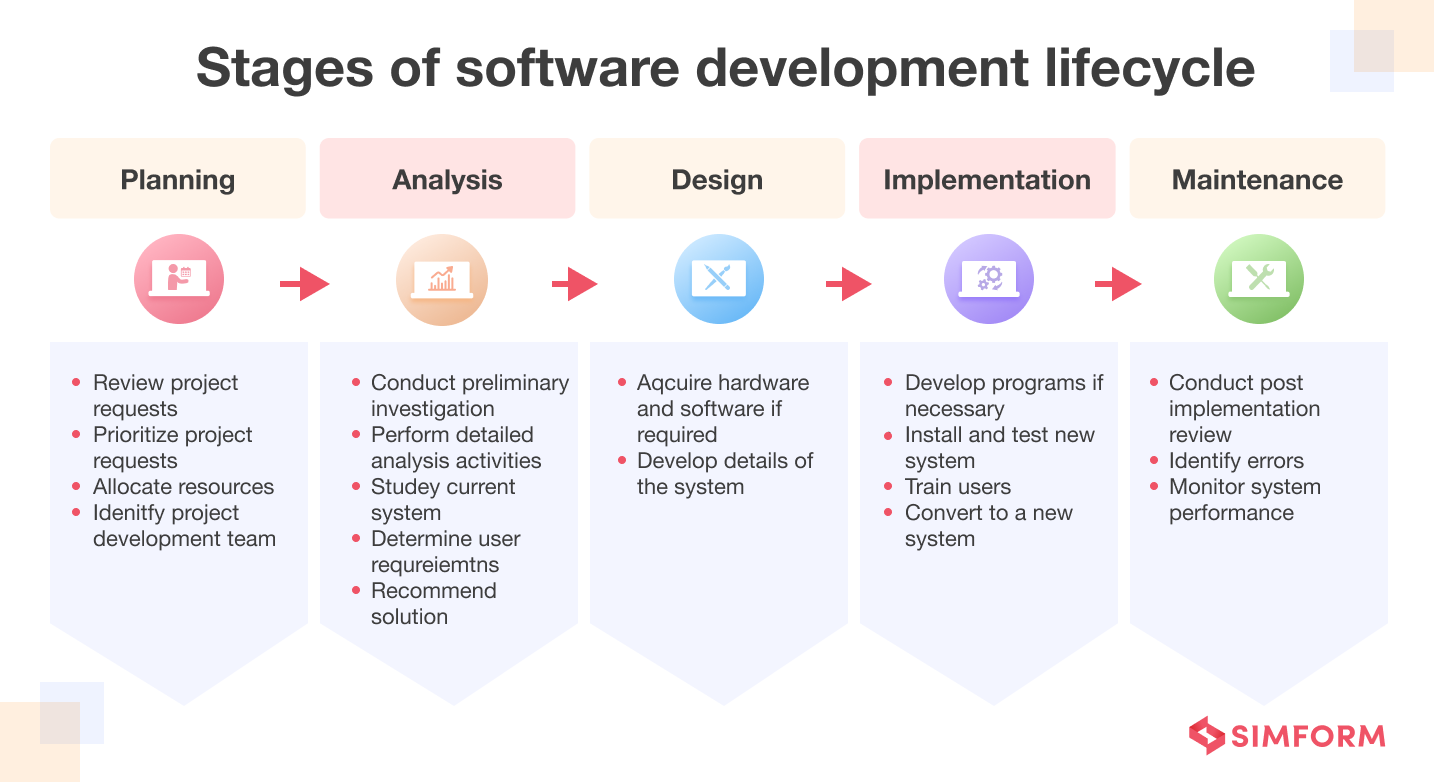
Software development life cycles (SDLCs) have several stages planning, design, development, testing, and maintenance. Optimizing SDLC stages requires implementing product engineering principles. Here’s how to implement product engineering at each phase of SDLC,
- Planning stage- Optimize planning by gathering data through enhanced data analytics from the market and customers to understand the product requirements.
- Analysis stage- Analyze product requirements and create a wireframe design.
- Design stage- Use the wireframe and information from the planning stage to design your product.
- Implementation phase – Implement the design using optimal development approaches such as agile methodologies, embracing DevOps, and implementing CI/CD.
- Maintenance stage- Monitor system performance and fine-tune software delivery
Top product engineering tools
GitHub is a web-based repository and hosting service. You can use it to share, collaborate, and contribute to open-source projects. It offers several features and tools for version control, code review, and issue tracking.
- Git-based version control system
- Excellent code collaboration and sharing features
- Robust issue tracking and project management tools
- Integration with various third-party tools and services
- Limited free features compared to other similar platforms
- Steep learning curve for beginners
- It can be expensive for enterprise organizations
Jira is a product engineering tool that you can use for project management, root cause analysis, and bug tracking. It enables cross-functional teams to collaborate and share critical project information easily.
Jira provides three indicators for monitoring software issues: status, transition, and resolution. A status indicates where the issue is within a workflow, the transition showcases the action taken to resolve it, and finally, the resolution tag indicates the completion of the task.
- Flexible and customizable workflow management
- Excellent issue-tracking and bug-reporting features
- Integration with other development tools and services
- Agile methodology support with features such as Scrum and Kanban boards
- It can be complex and overwhelming for small teams or beginners
- Limited customization options for non-technical users
- It can be expensive for larger teams or enterprise organizations
Git solves the problem of maintaining product versions. You can use a distributed version control system to manage versions in product development projects. It helps manage and track changes to source code.
- Fast and efficient version control system
- Supports distributed development workflows
- Excellent branching and merging capabilities
- Command-line interface and integration with other tools
- Requires technical knowledge to use effectively
- Limited support for large binary files
#4. IntelliJIDEA
IntelliJ IDEA is an integrated development environment (IDE) for Java, Kotlin, and other programming languages. It provides features such as code navigation, debugging, and refactoring. You can use it to create code for your software products and debug and refactor applications.
- Excellent code completion and error detection
- Robust debugging and testing tools
- Integration with various frameworks and tools
- Supports multiple programming languages
- Expensive compared to other IDEs
- It can be resource-intensive and slow on older hardware
- The steep learning curve for beginners
#5. Jenkins
Jenkins is an open-source automation server that enables organizations to implement CI/CD pipelines. It provides features such as build automation, testing, and deployment for your software projects.
This tool is beneficial for microservices -based projects, where updating services individually is necessary, as Jenkins offers better isolation and automated CI/CD pipeline implementation.
- Flexible and customizable automation workflows
- Integration with various development tools and services
- Open-source with a large community for support
- Resource-intensive and slow on older hardware
#6. SonarQube
SonarQube is an open-source code quality assurance tool that helps you find errors and bugs automatically before deploying them. It combines dynamic and static analysis tools to provide a comprehensive report on code quality.
- Optimizes application performance through reduced bugs, and errors
- Helps detect vulnerabilities, initially securing the applications
- Improves the code quality of apps
- Reduces cost of maintenance
- Does not provide automatic pull requests for fixes needed
- Does not offer enough insights into issues with project libraries
- Can create false positives, making maintenance tedious
ESLint is a linter of ECMAScript and a static code analysis tool that helps analyze the code quality to find bugs. This tool allows you to find problems with Javascript code through static analysis. ESLint is designed to be extensible and has several custom rules. These rules make it easy to understand the root cause of any error message.
- Highly flexible with the facility of changing rules as per specific requirements
- Provides tons of plugins, making it extensible
- Efficient in finding errors due to a set of several rules
- Supports ES6, and custom reporters
- Configuration requirements make it a little tricky
- Takes more time to find the issue and fix i
JUnit is a unit testing tool that you can use to test Java code. It enables you to find errors and ensure the expected output from the code execution. JUnit also provides a test runner to find methods marked as tests in the source code and execute those code blocks
- Provides text-based command lines or AWT-based or Swing-based graphical test mechanisms
- Offers annotations for testing purposes
- Allows you to write test code
- Pre-built test runners to test running application
- Not suitable for higher-level testing
- Group testing is not possible with JUnit
- Can’t create HTML test reports for different use cases
#8. Selenium
Selenium is an open-source automation testing framework that helps validate web apps across different browsers. You can use programming languages like Python, C#, Java, and others to write custom Selenium tests. However, it is essential to understand that Selenium is not just a tool but a suite of tools that help automate testing. Selenium is a suite of testing tools such as,
- Selenium Integrated Development Environment (IDE)
- Selenium Remote Control (RC)
- Selenium Grid
- Allows modification of the base framework due to open-source origins
- Cross-programming language compatibility makes it easy to use
- Easier integration of third-party tools and services
- Supports all the web browsers which are popular like Chrome, Firefox, Opera, etc.
- Higher initial cost as you need to make one-time payments
- Requires integrations for advanced functionality
Parallel testing with Selenium can get tricky based on project requirements
Product engineering principles are essential to businesses looking for optimal user experience. It helps streamline processes and define roles and responsibilities to make customer-centric products.
This, in turn, requires elaborate planning, analysis, monitoring, and applying several best practices. Partnering with product engineers can help organizations employ the best techniques and tools.
Just like this guide on AWS data engineering tools that help enhance product development. Reach out to discuss your needs!
Get in touch
Hiren Dhaduk
Hiren is CTO at Simform with an extensive experience in helping enterprises and startups streamline their business performance through data-driven innovation.
Cancel reply
Your email address will not be published. Required fields are marked *
Your comment here*
Sign up for the free Newsletter
For exclusive strategies not found on the blog
Sign up today!
Related Posts

8 Reasons Why Software Product Engineering Projects Fail and How to Avoid Them
CTO’s Guide to the Total Cost of Ownership (TCO) of a Digital Product

How to Scale Your Engineering Team to Accommodate Growth
Get insights on Gen AI adoption and implementation in 2024. Download the survey report now!

Agile Case Studies: Examples Across Various Industires
Home Blog Agile Agile Case Studies: Examples Across Various Industires
Agile methodologies have gained significant popularity in project management and product development. Various industries have successfully applied Agile principles, showcasing experiences, challenges, and benefits. Case studies demonstrate Agile's versatility in software development, manufacturing, and service sectors. These real-world examples offer practical insights into Agile implementation, challenges faced, and strategies to overcome them. Agile case studies provide valuable inspiration for implementing these methodologies in any project, regardless of the organization's size or industry.
Who Uses Agile Methodology?
Agile methodology is used by a wide variety of organizations, including:
- Software development companies use Agile to improve collaboration, increase flexibility, and deliver high-quality software incrementally.
- IT departments use agile to manage and execute projects efficiently, respond to changing requirements, and deliver value to stakeholders in a timely manner.
- Startups use agile to quickly adapt to market changes and iterate on product development based on customer feedback.
- Marketing and advertising agencies use agile to enhance campaign management, creative development, and customer engagement strategies.
- Product development teams use agile to iterate, test, and refine their designs and manufacturing processes.
- Project management teams use agile to enhance project execution, facilitate collaboration, and manage complex projects with changing requirements.
- Retail companies use agile to develop new marketing campaigns and improve their website and e-commerce platform.
Agile Case Study Examples
1. moving towards agile: managing loxon solutions.
Following is an Agile case study in banking:
Loxon Solutions, a Hungarian technology startup in the banking software industry, faced several challenges in its journey towards becoming an agile organization. As the company experienced rapid growth, it struggled with its hiring strategy, organizational development, and successful implementation of agile practices.
How was it solved:
Loxon Solutions implemented a structured recruitment process with targeted job postings and rigorous interviews to attract skilled candidates. They restructured the company into cross-functional teams, promoting better collaboration. Agile management training and coaching were provided to all employees, with online courses playing a crucial role. Agile teams with trained Scrum Masters and Product Owners were established, and agile ceremonies like daily stand-ups were introduced to enhance collaboration and transparency.
2. Contributions of Entrepreneurial Orientation in the Use of Agile Methods in Project Management
This Agile project management case study aims to analyze the degree of contribution of entrepreneurial orientation (EO) in the use of agile methods (AM) in project management. The study focuses on understanding how EO influences the adoption and effectiveness of agile methods within organizations. Through a detailed case study, we explore the relationship between entrepreneurial orientation and Agile methods, shedding light on the impact of entrepreneurial behaviors on project management practices.
A technology consulting firm faced multiple challenges in project management efficiency and responsiveness to changing client requirements. This specific problem was identified because of the limited use of Agile methods in project management, which hindered the company's ability to adapt quickly and deliver optimal outcomes.
Entrepreneurial orientation (EO) is a multidimensional construct that describes the extent to which an organization engages in entrepreneurial behaviors. The technology firm acknowledged the significance of entrepreneurial orientation in promoting agility and innovation in project management.
The five dimensions of Entreprenurial orientation were applied across the organization.
- Cultivating Innovativeness: The technology consulting firm encouraged a culture of innovativeness and proactiveness, urging project teams to think creatively, identify opportunities, and take proactive measures.
- Proactiveness: Employees were empowered to generate new ideas, challenge traditional approaches, and explore alternative solutions to project challenges. This helped them to stay ahead of the competition and to deliver the best possible results for their customers.
- Encouraging Risk-Taking: The organization promoted a supportive environment that encouraged calculated risk-taking and autonomy among project teams. Employees were given the freedom to make decisions and take ownership of their projects, fostering a sense of responsibility and accountability.
- Autonomy: Agile teams were given the autonomy to make decisions and take risks. This helped them to be more innovative and to deliver better results.
- Nurturing Competitive Aggressiveness: The technology firm instilled a competitive aggressiveness in project teams, motivating them to strive for excellence and deliver superior results.
3. Improving Team Performance and Engagement
How do you ensure your team performs efficiently without compromising on quality? Agile is a way of working that focuses on value to the customer and continuous improvement. Integrating Agile in your work will not only make the team efficient but will also ensure quality work. Below is a case study that finds how agile practices can help teams perform better.
The problem addressed in this case study is the need to understand the relationship between the Agile way of working and improving team performance and engagement. We see that teams often face challenges in their daily work. It could be a slow turnover due to bad time management, compromised quality due to lack of resources, or in general lack of collaboration. In the case study below, we will understand how adopting agile practices makes teams work collaboratively, improve quality and have a customer-focused approach to work.
How it was Solved:
A number of factors mediated the relationship between agile working and team performance and engagement.
- Create a culture of trust and transparency. Agile teams need to be able to trust each other and share information openly. This will help to create a sense of collaboration and ownership. This in turn can lead to increased performance and engagement.
- Foster communication and collaboration. Effective communication within the team and with stakeholders helps everyone be on the same page.
- Empower team members. Agile teams need to be empowered to make decisions and to take risks.
- Provide regular feedback. Team members need to receive regular feedback on their performance. This helps them to identify areas where they need improvement.
- Celebrate successes. By celebrating successes, both big and small, team members are motivated. This in turn creates a positive work environment.
- Provide training and development opportunities. help the team to stay up to date on the latest trends and to improve their skills.
- Encourage continuous improvement: Promoting a culture of continuous improvement helps the team to stay ahead of the competition and to deliver better results for their customers.
It was concluded that agile ways of working can have a positive impact on employee engagement and team performance. Teams that used agile methods were more likely to report high levels of performance and engagement.
4. $65 Million Electric Utility Project Completed Ahead of Schedule and Under Budget
Xcel Energy faced a significant challenge in meeting the Reliability Need required by the Southwest Power Pool in New Mexico. The company had committed to constructing a new 34-mile, 345-kilovolt transmission line within a strict budget of $65 million and a specific timeline. Additionally, the project had to adhere to Bureau of Land Management (BLM) environmental requirements. These constraints posed a challenge to Xcel Energy in terms of project management and resource allocation.
A PM Solutions consultant with project management and utility industry experience was deployed to Xcel Energy.
The PM Solutions consultant deployed to Xcel adapted to the organization's structure and processes, integrating into the Project Management functional organization. He utilized years of project management and utility industry experience to provide valuable insights and guidance.
- Collaborative and social skills were used to address roadblocks and mitigate risks.
- Focused on identifying and addressing roadblocks and risks to ensure timely project delivery.
- Vendor, design, and construction meetings were organized to facilitate communication and collaboration.
- Monitored and expedited long-lead equipment deliveries to maintain project schedule.
- Design and Construction milestones and commitments were closely monitored through field visits.
- Actively tracked estimates, actual costs, and change orders to control project budget.
- Assisted functional areas in meeting their commitments and resolving challenges.
The project was completed eleven days ahead of schedule and approximately $4 million under budget. The management team recognized the project as a success since it went as planned, meeting all technical and quality requirements.
5. Lean product development and agile project management in the construction industry
The construction industry, specifically during the design stage, has not widely embraced Lean Project Delivery (LPD) and Agile Project Management (APM) practices. This limited adoption delays the industry's progress in enhancing efficiency, productivity, and collaboration in design.
- Integrated project delivery and collaborative contracts: Collaborative contracts were implemented to incentivize teamwork and shared project goals, effectively breaking down silos and fostering a collaborative culture within the organization.
- Lean principles in design processes: Incorporating Lean principles into design processes was encouraged to promote lean thinking and identify non-value-adding activities, bottlenecks, and process inefficiencies.
- Agile methodologies and cross-functional teams: Agile methodologies and cross-functional teams were adopted to facilitate iterative and adaptive design processes.
- Digital tools and technologies: The organization embraced digital tools and technologies, such as collaborative project management software, Building Information Modeling (BIM), and cloud-based platforms.
- A culture of innovation and learning: A culture of innovation and learning was promoted through training and workshops on Lean Project Delivery (LPD) and Agile Project Management (APM) methodologies. Incorporating Agile management training, such as KnowledgeHut Agile Training online , further enhanced the team's ability to implement LPD and APM effectively.
- Clear project goals and metrics: Clear project goals and key performance indicators (KPIs) were established, aligning with LPD and APM principles. Regular monitoring and measurement of progress against these metrics helped identify areas for improvement and drive accountability.
- Industry best practices and case studies: industry best practices and case studies were explored, and guidance was sought from experts to gain valuable insights into effective strategies and techniques for implementation.
6. Ambidexterity in Agile Software Development (ASD) Projects
An organization in the software development industry aims to enhance their understanding of the tensions between exploitation (continuity) and exploration (change) within Agile software development (ASD) project teams. They seek to identify and implement ambidextrous strategies to effectively balance these two aspects.
How it was solved:
- Recognizing tensions: Teams were encouraged to understand and acknowledge the inherent tensions between exploitation and exploration in Agile projects.
- Fostering a culture of ambidexterity: The organization created a culture that values both stability and innovation, emphasizing the importance of balancing the two.
- Balancing resource allocation: Resources were allocated between exploitation and exploration activities, ensuring a fair distribution to support both aspects effectively.
- Supporting knowledge sharing: Team members were encouraged to share their expertise and lessons learned from both exploitation and exploration, fostering a culture of continuous learning.
- Promoting cross-functional collaboration: Collaboration between team members involved in both aspects was facilitated, allowing for cross-pollination of ideas and insights.
- Establishing feedback mechanisms: Feedback loops were implemented to evaluate the impact of exploitation and exploration efforts, enabling teams to make data-driven decisions and improvements.
- Developing flexible processes: Agile practices that supported both stability and innovation, such as iterative development and adaptive planning, were adopted to ensure flexibility and responsiveness.
- Providing leadership support: Leaders promoted and provided necessary resources for the adoption of agile practices, demonstrating their commitment to ambidexterity.
- Encouraging experimentation: An environment that encouraged risk-taking and the exploration of new ideas was fostered, allowing teams to innovate and try new approaches.
- Continuous improvement: Regular assessments and adaptations of agile practices were conducted based on feedback and evolving project needs, enabling teams to continuously improve their ambidextrous strategies.
7. Problem and Solutions for PM Governance Combined with Agile Tools in Financial Services Programs
Problem: The consumer finance company faced challenges due to changing state and federal regulatory compliance requirements, resulting in the need to reinvent their custom-built storefront and home office systems. The IT and PMO teams were not equipped to handle the complexities of developing new systems, leading to schedule overruns, turnover of staff and technologies, and the need to restart projects multiple times.
How it was Solved:
To address these challenges, the company implemented several solutions with the help of PM Solutions:
- Back to Basics Approach: A senior-level program manager was brought in to conduct a full project review and establish stakeholder ownership and project governance. This helped refocus the teams on the project's objectives and establish a clear direction.
- Agile Techniques and Sprints: The company gradually introduced agile techniques, starting with a series of sprints to develop "proof of concept" components of the system. Agile methodologies allowed for more flexibility and quicker iterations, enabling faster progress.
- Expanded Use of JIRA: The company utilized Atlassian's JIRA system, which was already in place for operational maintenance, to support the new development project. PM Solutions expanded the use of JIRA by creating workflows and tools specifically tailored to the agile approach, improving timeliness and success rates for delivered work.
- Kanban Approach: A Kanban approach was introduced to help pace the work and track deliveries. This visual management technique enabled project management to monitor progress, manage workloads effectively, and report updates to stakeholders.
- Organizational Change Management: PM Solutions assisted the company in developing an organizational change management system. This system emphasized early management review of requirements and authorizations before work was assigned. By involving company leadership in prioritization and resource utilization decisions, the workload for the IT department was reduced, and focus was placed on essential tasks and priorities.
8. Insurance Company Cuts Cycle Time by 20% and Saves Nearly $5 Million Using Agile Project Management Practices
In this Agile Scrum case study, the insurance company successfully implemented Agile Scrum methodology for their software development projects, resulting in significant improvements in project delivery and overall team performance.
The insurance company faced challenges with long project cycles, slow decision-making processes, and lack of flexibility in adapting to changing customer demands. These issues resulted in higher costs, delayed project deliveries, and lower customer satisfaction levels.
- Implementation of Agile Practices: To address these challenges, the company decided to transition from traditional project management approaches to Agile methodologies. The key steps in implementing Agile practices were as follows:
- Executive Sponsorship: The company's leadership recognized the need for change and provided full support for the Agile transformation initiative. They appointed Agile champions and empowered them to drive the adoption of Agile practices across the organization.
- Training and Skill Development: Agile training programs were conducted to equip employees with the necessary knowledge and skills. Training covered various Agile frameworks, such as Scrum and Kanban, and focused on enhancing collaboration, adaptive planning, and iterative development.
- Agile Team Formation: Cross-functional Agile teams were formed, consisting of individuals with diverse skill sets necessary to deliver projects end-to-end. These teams were self-organizing and empowered to make decisions, fostering a sense of ownership and accountability.
- Agile Project Management Tools: The company implemented Agile project management tools and platforms to facilitate communication, collaboration, and transparency. These tools enabled real-time tracking of project progress, backlog management, and seamless coordination among team members.
9. Agile and Generic Work Values of British vs Indian IT Workers
Problem:
In this Agile transformation case study, the problem identified is the lack of effective communication and alignment within an IT firm unit during the transformation towards an agile work culture. The employees from different cultural backgrounds had different perceptions and understanding of what it means to be agile, leading to clashes in behaviors and limited team communication. This situation undermined morale, trust, and the sense of working well together.
The study suggests that the cultural background of IT employees and managers, influenced by different national values and norms, can impact the adoption and interpretation of agile work values.
- Leadership: Leaders role-modeled the full agile mindset, along with cross-cultural skills. They demonstrated teamwork, justice, equality, transparency, end-user orientation, helpful leadership, and effective communication.
- Culture: Managers recognized and appreciated the cultural diversity within the organization. Cultural awareness and sensitivity training were provided to help employees and managers understand and appreciate the diverse cultural backgrounds within the organization.
- Agile values: The importance of agile work values was emphasized, including shared responsibility, continuous learning and improvement, self-organizing teamwork, fast fact-based decision-making, empowered employees, and embracing change. Managers actively promoted and reinforced these values in their leading and coaching efforts to cultivate an agile mindset among employees.
- Transformation: A shift was made from a centralized accountability model to a culture of shared responsibility. Participation in planning work projects was encouraged, and employees were empowered to choose their own tasks within the context of the team's objectives.
- Roadmap: An agile transformation roadmap was developed and implemented, covering specific actions and milestones to accelerate the adoption of agile ways of working.
- Senior management received necessary support, training, and additional management consultancy to drive the agile transformation effectively.
Benefits of Case Studies for Professionals
Case studies provide several benefits for professionals in various fields:
- Real-world Application: Agile methodology examples and case studies offer insights into real-life situations, allowing professionals to see how theoretical concepts and principles are applied in practice.
- Learning from Success and Failure: Agile transformation case studies often present both successful and failed projects or initiatives. By examining these cases, professionals can learn from the successes and avoid the mistakes made in the failures.
- Problem-solving and Decision-making Skills: Case studies present complex problems or challenges that professionals need to analyze and solve. By working through these cases, professionals develop critical thinking, problem-solving, and decision-making skills.
- Building Expertise: By studying cases that are relevant to their area of expertise, professionals can enhance their knowledge and become subject matter experts.
- Professional Development: Analyzing and discussing case studies with peers or mentors promotes professional development.
- Practical Application of Concepts: Teams can test their understanding of concepts, methodologies, and best practices by analyzing and proposing solutions for the challenges presented in the cases.
- Continuous Learning and Adaptation: By studying these cases, professionals can stay updated on industry trends, best practices, and emerging technologies.
Examine the top trending Agile Category Courses
In conclusion, agile methodology case studies are valuable tools for professionals in various fields. The real-world examples and insights into specific problems and solutions, allow professionals to learn from others' experiences and apply those learning their own work. Case studies offer a deeper understanding of complex situations, highlighting the challenges faced, the strategies employed, and the outcomes achieved.
The benefits of case studies for professionals are numerous. They offer an opportunity to analyze and evaluate different approaches, methodologies, and best practices. Case studies also help professionals develop critical thinking skills, problem-solving abilities, and decision-making capabilities through practical scenarios and dilemmas to navigate.
Overall, agile case study examples offer professionals the opportunity to gain practical wisdom and enhance their professional development. Studying real-life examples helps professionals acquire valuable insights, expand their knowledge base, and improve their problem-solving abilities.
Frequently Asked Questions (FAQs)
Three examples of Agile methodologies are:
Scrum: Scrum is one of the most widely used Agile frameworks. It emphasizes iterative and incremental development, with a focus on delivering value to the customer in short, time-boxed iterations called sprints.
Kanban: Kanban is a visual Agile framework that aims to optimize workflow efficiency and promote continuous delivery.
Lean: Lean is a philosophy and Agile approach focused on maximizing value while minimizing waste.
- People over process: Agile values the people involved in software development, and emphasizes communication and collaboration.
- Working software over documentation: Agile prioritizes delivering working software over extensive documentation.
- Customer collaboration over contract negotiation: Agile values close collaboration with customers and stakeholders throughout the development process.
- Responding to change over following a plan: Agile recognizes that change is inevitable, and encourages flexibility and adaptability.
The six phases in Agile are:
- Initiation: Define the project and assemble the team.
- Planning: Create a plan for how to achieve the project's goals.
- Development: Build the product or service in short sprints.
- Testing: Ensure the product or service meets requirements.
- Deployment: Release the product or service to the customer.
- Maintenance: Support the product or service with bug fixes, new features, and improvements.

Lindy Quick
Lindy Quick, SPCT, is a dynamic Transformation Architect and Senior Business Agility Consultant with a proven track record of success in driving agile transformations. With expertise in multiple agile frameworks, including SAFe, Scrum, and Kanban, Lindy has led impactful transformations across diverse industries such as manufacturing, defense, insurance/financial, and federal government. Lindy's exceptional communication, leadership, and problem-solving skills have earned her a reputation as a trusted advisor. Currently associated with KnowledgeHut and upGrad, Lindy fosters Lean-Agile principles and mindset through coaching, training, and successful execution of transformations. With a passion for effective value delivery, Lindy is a sought-after expert in the field.
Avail your free 1:1 mentorship session.
Something went wrong
Upcoming Agile Management Batches & Dates
| Name | Date | Fee | Know more |
|---|
Press Enter to search
50 Product Management Case Studies
We often wonder what kind of process other product teams have created, planned, and most importantly, how they have implemented it. That is why we at Producter have compiled 50 different case studies for you.
2 years ago • 4 min read
We often wonder what kind of process other product teams have created, planned, and most importantly, how they have implemented it.
That is why we at Producter have compiled 50 different case studies for you.
Brought to you by Roadmape

1- Rules of Flow for Product Management: an AirBnB Case Study
“Engagement” is a term that is so overused in product management that it has almost lost its meaning. So often I’ve heard from teams, “We’ll measure the success of this test with engagement,” which could mean anything from feature click-through to bounce to we-aren’t-really-sure-this-will-drive-conversion-so-we’re-hedging-our-bet. Underneath, the reason this term has been co-opted and jargonized is that genuine, productive engagement can be ramped toward long-term customer loyalty. And loyalty pays off: a loyalty increase of 7% can boost lifetime profits per customer by as much as 85%, and a loyalty increase of 3% can correlate to a 10% cost reduction ( Brand Keys ).

2- The Psychology of Clubhouse’s User Retention (...and churn)

3- Netflix Q1 ’21 Subscriber Growth Miss: Can We Avoid Another One?
As a data analyst supporting a mobile subscription business , Netflix’s Q1 ’21 subscriber growth miss is a classic example of when I would get called for recommendations to prevent a miss in the future. I thought this would make an interesting case study to discuss my approach to finding insights to drive subscriber growth. Sadly I’m not a Netflix employee and will be limited to publicly available data but the wealth of information on the Internet about Netflix is sufficient to generate insights for this case study.

4- Amazon Go Green
As part of the Design Challenge from productdesign.tips, our team came together to find ways for Amazon to encourage more sustainability on their e-commerce platform. As with any unsolicited design project, the challenge comes with a lack of access to application analytics and technical feasibilities. Nonetheless, the question remains: How might we design checkout screens for an e-commerce app to help people recycle the goods they buy?

5- Quora Case Study – The Wonderful World of Quora
Quora has become a substantive resource for millions of entrepreneurs and one of the best sources for Business to Business market. Majorly used by writers, scholars, bloggers, investors, consultants, students this Q/A site has much to offer in terms of knowledge sharing, connection building and information gathering.

6- Building a product without any full-time product managers

Jambb is an emerging social platform where creators grow their communities by recognizing and rewarding fans for their support. Currently, creators monetize fan engagement through advertisements, merchandise, and subscriptions, to name a few. However, this only represents 1% of fans, leaving the other 99% (who contribute in non-monetary ways) without the same content, access, and recognition that they deserve.

8- What if you can create Listening Sessions on Spotify
Summary: The project was done as a part of a user experience design challenge given to me by a company. I was given the brief by them to work on a feature of Spotify and I spent around 25–30 hours on the challenge in which I went through the entire process, from the research to testing.

9- Redesigned Apple Maps and replicated an Apple product launch for it
Quick-fire question; what is the single most important and widely used feature in a phone — asides from texting and instant messaging friends, coworkers and family? Maybe you guessed right, perhaps this feature is so integrated into your life that you didn’t even think about it — either way, it is your phone’s GPS. It is reasonable to say that GPS technology has changed society’s lives in ways we never could’ve imagined. Gone are the days of using physically printed maps and almanacks, when we now have smartphones with navigation apps. Since the launch of the iPhone and the App Store, consumers have been able to use different apps for their personal navigation needs. Everyone has a preference, and apps have come out to try and address every need.

10- Intuitive design and product-led growth
In 2018, Miro was hardly a blip on the radar in the Design world. Fast forward two years, and suddenly Miro is solidly the number one tool for brainstorming and ideation.

Click below to see the complete list 👇

Producter is a product management tool designed to become customer-driven.
It helps you collect feedback , manage tasks , sharing product updates , creating product docs , and tracking roadmap .
Spread the word
What is customer segmentation, learnings about product development strategy in 2022, keep reading, boost product management with slack: a comprehensive guide to producter's slack integration, mastering the art of product management: 10 essential strategies for success, what is user research.

Updated: 5 June 2024 Contributors: Molly Hayes, Amanda Downie
Product engineering is a phase of product development in which engineering principles and practices are applied to a product lifecycle . The process includes design, development, testing, and optimization .
Product engineering can refer to software engineering or the design of new products like electronics, industrial equipment, or automobiles. Regardless of the end product, product engineering involves a multidisciplinary approach. This can mean combining aspects of mechanical engineering, electrical engineering, software engineering, and other relevant fields to create innovative and high-quality products.
Successful product engineering delivers products that not only meet functional and technical requirements but provide elevated user experiences and complement an organization’s overarching business strategy . It is often useful for an organization to systematize the product engineering process, outlining a detailed roadmap and designating product managers to ensure a timely and cost-effective process. This will help an organization maximize a product’s value for its entire lifecycle .
Explore how and why digital product engineering is the way companies can bridge the digital divide.
While product engineering and product development are closely related terms that are occasionally used interchangeably, they refer to distinct processes. The product development process encompasses the full, non-technical undertaking of creating a new product and involves post-launch activities like marketing and sales. Product engineering is more limited in scope. Typically, it focuses on engineering functions like technical specifications, materials, manufacturing, and the integration of technologies or software. While both processes are crucial for the successful creation and launch of a new product, product engineering is a specialized subset of the broader product development framework.
The product engineering process involves collaboration between various teams and stakeholders to ensure the successful development and delivery of the final product. The makeup of this team can vary depending on the organization and nature of the product. Commonly involved team members can include:
This could include software engineers and developers, a quality assurance team who performs testing and validation, or hardware engineers (e.g., mechanical engineers) who work on the physical aspects of product design.
Executive leadership may be involved to provide overall direction, support, and resources for product development.
Procurement specialists and the finance team may collaborate with the product engineering team. These specialists help to ensure the most high-quality materials are used in the product development process and that those materials comply with the project’s budget.
Depending on the product, legal council or compliance specialists may be required to ensure the product complies with relevant laws and regulations.
Product managers are responsible for defining the organizational roadmap and product strategy.
While there is no single product engineering roadmap that will apply to all organizations, there are some central elements that are typically included in an agile and systematic process.
These phases ensure that a product is intelligently designed and built with both business requirements and the end user in mind. Testing, iteration, validation, and continuous monitoring of outcomes ensures innovation is balanced with technical feasibility and budgetary considerations. Given the complexity of the product engineering process, some organizations may elect to contract an experienced third-party firm specializing in product engineering services or project management to streamline the undertaking.
As with product design, product engineering begins with brainstorming and the conceptualization of product ideas. While engineers typically don’t engage in market research themselves, during this development phase they might work to translate abstract concepts like customer needs into tangible product specifications, considering the role of product, technical feasibility, time-to-market, and other external demands. This phase might also include the gathering of in-depth data, identifying system requirements , and outlining functionalities.
Once an idea is finalized, engineers begin the design phase in which they create detailed wireframes, specifications, drawings, and models of the product. This phase might include the use of CAD (computer-aided design) tools to develop two-dimensional and three-dimensional models. Engineers design and build prototypes, which are built and tested to validate the product before it moves to full-scale production. Multiple prototypes might be built to assist in difficult problem-solving processes. In software engineering, engineers might define the overall system architecture of the product during this phase.
As the product is being designed, or shortly after the final prototype is approved, the design team and engineering team collaborates with procurement specialists to plan how the end product will be constructed. During this time, engineers make crucial decisions about the selection of materials and the manufacturing process. Considering factors such as cost, scalability, durability, and sustainability, engineers and their team members make a detailed plan for mass production.
As technology has advanced, it’s become more common for products to involve a combination of hardware and software components. The final product might be a piece of software itself, or require a separate software product engineering process to ensure seamless integration with a piece of hardware. Ideally, the design and prototyping phase would have included plans for the integration of various technologies to create a cohesive and functional product.
During the product testing phase, engineers stridently evaluate the product to ensure it meets the predefined specifications and requirements — this can include testing for performance, reliability, safety, scalability, and usability. As part of the testing process, quality assurance (QA) measures are implemented to ensure the product can meet or exceed industry standards and consumer expectations. Validation processes might include testing automation or performance testing, depending on the product designed.
In an ideal product engineering workflow, development and testing is an ongoing process rather than an end state. During the design phase, engineers may have planned for future updates and maintenance. Ongoing testing and monitoring key metrics is crucial to ensuring the product is performing as intended. For some organizations, investing in ongoing iteration may be the most appropriate course of action after the product launches. This could mean designing and deploying updates, collecting and filtering user feedback, or dedicating an engineering team to continuously monitor the product’s performance.
- Competitive advantage: A systematized and informed product engineering process can result in goods or services that exceed customer expectations and create a competitive edge in the market.
- Cost efficiency: Through careful selection of materials and manufacturing processes—and the optimization of system architecture and design—product engineers can achieve cost efficiency without compromising on quality. By identifying issues before they arise and working continuously to improve the product, engineers can reduce costs.
- High-quality products: Product engineers optimize the design of a product, focusing on creating efficient consumer goods with the best possible functionality and performance. This results in more durable and reliable products with a high degree of usability.
- Increased innovation: Product engineering facilitates innovation by encouraging creative solutions to design and manufacturing challenges, applying new technologies, materials, and approaches to the development of novel products.
- Regulatory compliance: The product engineering process ensures that the final product complies with all relevant regulations, standards, and safety requirements.
With a focus on business process and outcomes optimization, IBM's high-quality digital product engineering services help clients turn ideas into differentiated product portfolios that accellerate growth.
IBM Engineering Lifecycle Managment (ELM) provides end-to-end engineering solutions that stand at the forefront of the market, seamlessly guiding clients from requirements to systems design, workflow, and test management.
IBM Garage helps clients break through strategy roadblocks, integrate intelligent insights, and harness the power of data to radically improve efficiency, generating innovative and effective ideas that are rapidly converted into business value.
Discover how continuous experimentation and iteration can help companies quickly adapt to customer needs and preferences.
Discover how successful companies can fuse product development practices with overarching business strategies to ensure sustainable innovation.
Explore how a value-based mindset can triumph over traditional approaches to product development.
Discover how clear, concise, and error-free requirements help engineering teams detect errors early, reducing project cost and risk.
Discover why it's time to embrace an end-to-end management approach to your systems and software development.
Explore how IBM helped a national healthcare innovator optimize operating room efficiency with streamlined data management and intelligent product engineering.
Accelerate business outcomes and improve competitive advantage with custom built products and platforms.
Sourcing & Supply Chain Report: Data is the New Currency
Design for Manufacturing Examples: Real-Life Engineering Case Studies

Key Takeaways:
- More than 70% of a part’s cost can be locked in during the early design phase
- Adopt a robust DFM process using digital manufacturing simulation tools to address cost, sustainability, and innovative design iterations simultaneously
The Full Article:
Typically, more than 70% of a part’s cost is locked in once its design is finalized. And at that point, manufacturing and sourcing teams have limited options to optimize part costs. That’s why cost modeling is exponentially more effective during the design phase. Product engineers need the ability to identify viable, cost-effective design alternatives while a project is still on the drawing board. This approach empowers design teams to innovate without sacrificing time to market or profit margins.
We explore this topic in greater detail by examining real-life examples to illustrate a key DFM principle in action. This includes why spreadsheets and other tools that rely on historical estimates provide a static, incomplete view of costing data – and how you can overcome this challenge with solutions that link design decisions to cost outcomes. Areas addressed include:
- The Design for Manufacturing (DFM) Imperative
- Overview of Important Cost Categories
- DFM Success Stories: Identifying Cost Inefficiencies
- Hidden Material Cost Drivers
- DFM Material Conversion Cost Example
- Other Methods for Cost-Effective Design for Manufacturability
- Learn More About the Power of Digital Manufacturing Insights
1) The Design for Manufacturing (DFM) Imperative
What if engineers had precise, design-level guidance on key cost drivers for their new product designs? And what if they had the insight to see how the cost is being affected by raw materials, conversion (i.e., the cost of turning raw material into a part), routing, and other manufacturability issues?
Having access to this capability would provide design and cost engineers with guidance to revise parts for added cost efficiency during the design phase. aPriori’s Manufacturing Insights Platform offers a solution that enables organizations to achieve this objective.
Unlike traditional spreadsheets, aPriori automatically evaluates the geometry of 3D CAD models whenever they are checked into a product lifecycle management (PLM) system. Through this software functionality, engineers gain real-time cost insights for parts and sub-assemblies, improving design and sourcing decisions.
Moreover, aPriori provides teams with a deeper understanding of the complex factors influencing part costs. The software is also equipped with cost and process modeling capabilities , enabling engineers to configure and run various scenarios. As a result, teams can seamlessly compare a part’s material, supplier, regional expenses, and more to make informed decisions.
To understand the impact of advanced manufacturing cost modeling, it’s helpful to consider the factors that contribute to a part’s final cost. Below, we break down a few key categories of part cost. The specifics may vary greatly, but these basic cost categories apply whether the part in question is sheet metal or plastic, cast or machined.
2) Overview of Important Cost Categories
Direct + variable costs:.
The powerful interaction between each choice in the direct/variable cost category is significantly important. While engineering decisions may have an impact on period costs in the long run, we will focus on direct costs, as they often have the most substantial impact. The following categories describe the expenses associated with the marginal cost of producing each additional part.
Key Drivers of Material Costs
- Material type
- Material stock size (standard or non-standard)
- Material selection and utilization
- Special grain orientations (e.g., tight bends on a part may only allow manufacturing to orient the part in one direction when cutting it on the sheet)
Key Drivers of Overhead and Labor Costs
- Cycle time to make the part. Note: more than one machine may be used to make a part.
- Number of times that the part must be set up – whether in one machine or multiple machines
- Type and size of machine(s) that will be used to make the part
- Any secondary production processes such as paint, heat treatment, etc.
Indirect/Period Costs:
These costs matter for overall profitability but aren’t necessarily immediately impacted by marginal production changes. For instance, a factory will have some base level of maintenance costs regardless of the number of parts being made within a given period. These costs must be associated with specific supporting functions and spread across all parts produced.
Key Factory-Related Cost Drivers
- Energy costs
- Heating and cooling the plant
- Cleaning and maintenance
- Purchasing, manufacturing, engineering, shipping and receiving, and other supporting business functions
Key Administrative Cost Drivers
- General management costs
- Sales, marketing, and business development expenditures
- Technology support (e.g., IT staff or services)
Capital Expenditures (CapEx) and Non-Recurring Costs:
- Examples include initial investments in productive capital such as molds, stamping dies, machining fixtures, weld fixtures, and more.
- The cost impact of capital expenditures will vary depending on the complexity of the part, number of cavities, number of parts over the life of the tool, etc.
3) DFM Success Stories: Identifying Cost Inefficiencies
We developed both case studies using aPriori’s digital factory capabilities, which involve simulated production based on modeling a part’s digital twin .
During the design stage, you don’t need the absolute value estimate to be exact; a good, reliable approximation will suffice. For instance, you may determine that 20% of the part cost is material and 65% is conversion cost. While these amounts may vary during final production, they can provide a useful guidepost for prioritizing cost optimization projects. This practice can help you save time by avoiding product design changes that will have minimal impact on cost.
Manufacturing insights can help engineers minimize time-consuming activities and work faster. This automation-driven platform can determine the most efficient manufacturing methodology through near-instant cost estimates for new design alternatives.
Material Cost Example One: Truck Sheet Metal Fan Cover Redesign
The following screenshot shows that 88% of the fan cover cost is material. To reduce material costs, you can:
- Select an alternative material that is cheaper (but still reflects functional load requirements and tolerances).
- Use less material by making the part thinner, adding ribbed forms to strengthen it, or improving material utilization to reduce waste.
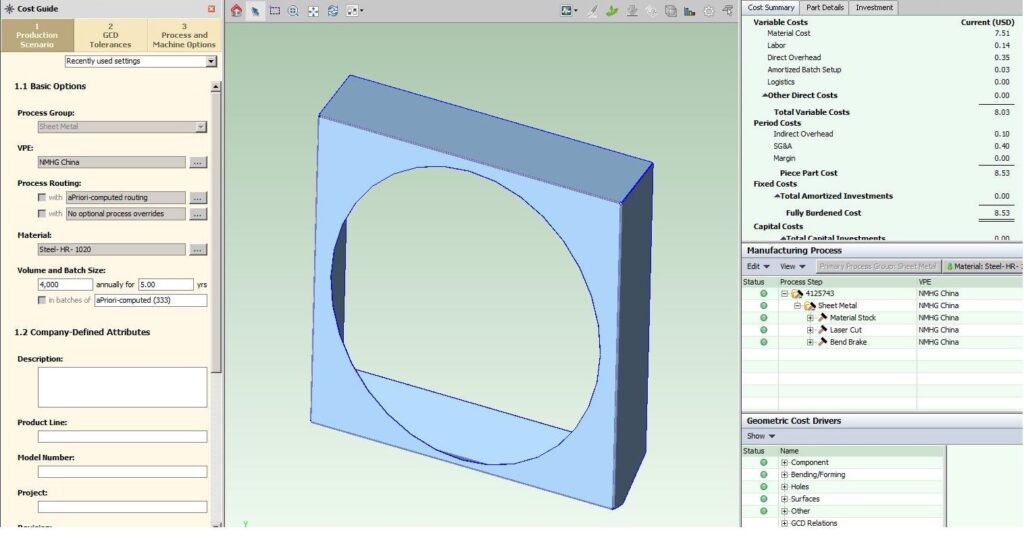
The product developer recognized that the material choice was the primary cost driver and reduced the part size without altering the size of the opening or component mating points. The following screenshot displays his final solution.
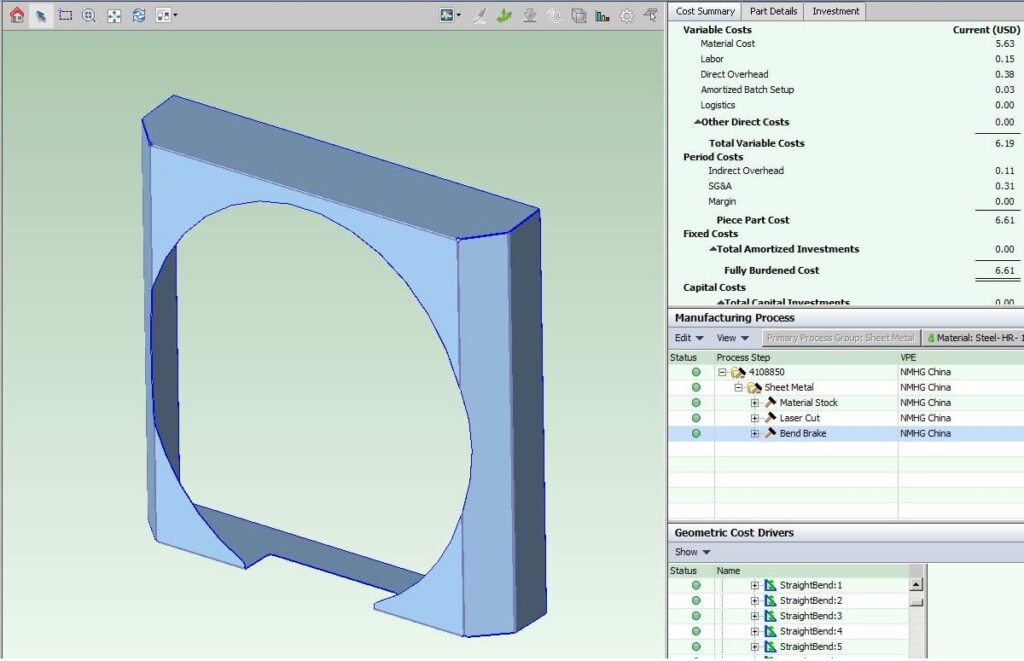
Note that while labor and overhead costs increased from $0.49 to $0.53, the material cost dropped from $7.51 to $5.63, saving $1.88 – which is a 25% savings. This improvement has paid for itself exponentially because the part is still used in tens of thousands of trucks.
This is a great example of how a reliable cost estimate is useful for prioritizing redesign work. A good cost vector (whether the cost is going up or down, by a little or a lot) is sufficient. For example, if the material cost dropped by only $1.50 instead of $1.88, the price reduction would still warrant a redesign.
Material Cost Example Two: Plastic Seat
A manufacturer produces approximately 200,000 seats annually. The digital manufacturing cost model revealed that material is 67% of the total cost.
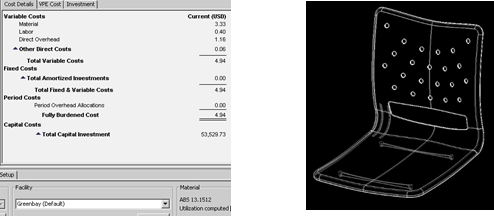
The engineer redesigning the seat has two options:
- Use lower-cost materials. Note: had the conversion cost been the most expensive, you may have wanted a material that cools faster, thereby decreasing the cycle time and production cost.
- Reduce the amount of material without compromising seat integrity.
The engineer tried several alternative designs, including:
She began by reducing the thickness of the plastic from the top edge of the back of the seat down to 2/3 of the way and from the edge of the bottom of the seat to approximately ½ of the way to the middle of the seat. This change decreased the average thickness from 0.18” to 0.15”. It is critical to note that the cost of materials, labor, and overhead was also reduced. That’s because the thinner part cools faster, leading to a double benefit: a reduction in material and manufacturing costs, totaling $0.95 on a $5 component – a nearly 20% reduction.
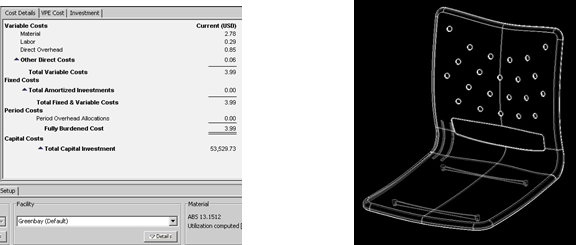
The second design change made the back hole slightly larger from its original 5”–6” in height. However, because this change only shaved a few cents off the original cost, it was not worth the risk of potential quality issues or increased customer discomfort. The value of having real-time cost feedback “at the speed of design” enables you to catch these false starts far earlier in the process and maintain quality control by adhering to the principles of DFM.

4) Hidden Material Cost Drivers
- This approach worked until their factory became overwhelmed and started buying parts or sending them to another internal factory across the country. The parts became much more expensive because they needed to orient the components perpendicular to the bend, which limits the nesting flexibility of the part and requires more material. Simulated production software like aPriori can automatically identify if a bend is too tight and recommend a minimum bend angle.
- The organization suspected an unscrupulous bid from a supplier. Still, upon review, it found that the supplier had to buy a special forging or start with the next size-up standard bar to meet the customer’s requirements. Either way, the cost would be disproportionately impacted. A diameter reduction of just a few millimeters fixed the issue, and the final design still had plenty of inertia margin.
5) DFM Material Conversion Cost Example
Let’s now move into conversion costs. Design engineers make choices that affect a large range of conversion costs, such as:
- Labor cost is proportional to cycle time. And the skill necessary to run the machine affects the wages of the operator. A 5-axis CNC machinist makes more than a 3-axis mill operator, for example.
- Set-up cost includes the number of machines to be set up and the number of times the part needs to be set up. Volume plays a large role in determining the per-product impact of set-up costs.
- Direct overhead cost is proportional to cycle time and the type and size of the machine.
An engineer was assigned to reduce the cost for a part like the one below. A quick design review revealed a 40/60 split between material cost and conversion cost. This implies that there may be opportunities to contain costs on both sides of this split without impacting lead times.
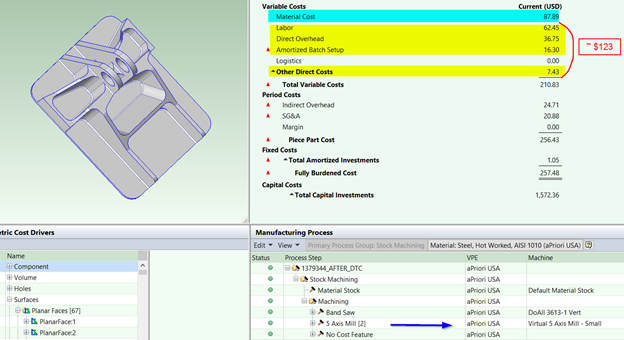
The engineer also noted that because this is a relatively low-volume part (300 units per year), it was being purchased as a machined part. While not very complex, the multiple slants on the surfaces were forcing this part to a 5-axis mill (rather than a comparatively cheap 3-axis mill).
The engineer had three choices to reduce costs:
- Redesign the part to reduce complexity for production on a cheaper machine
- Investigate machining costs further and address those issues in the design
- Identify alternative manufacturing processes for the part if they show promise
Using simulated manufacturing to analyze costs, the engineer discovered that the material utilization was only 11%, meaning that nearly 9 lbs. out of every 10 lbs. of material would be wasted. As expected, most of the cost of making the part was in machining, but from roughing operations, not finishing the part. This demonstrated that getting the part to near net shape was costing a lot in both material and manufacturing costs (see the figures below).
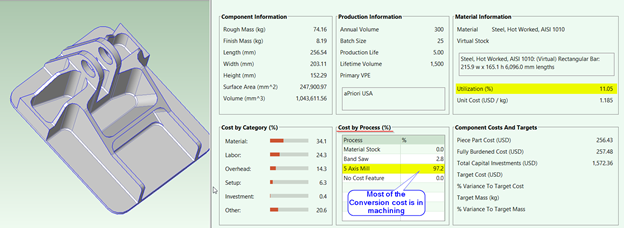
This part had been designated as a machined component because of the relatively low volume production of 300 units per year. However, based on this evidence, the engineer decided to investigate sand casting for the part. To see if it would be worth redoing the design and fatigue analysis to turn this into a casting, he created a cost estimate for sand casting the part.
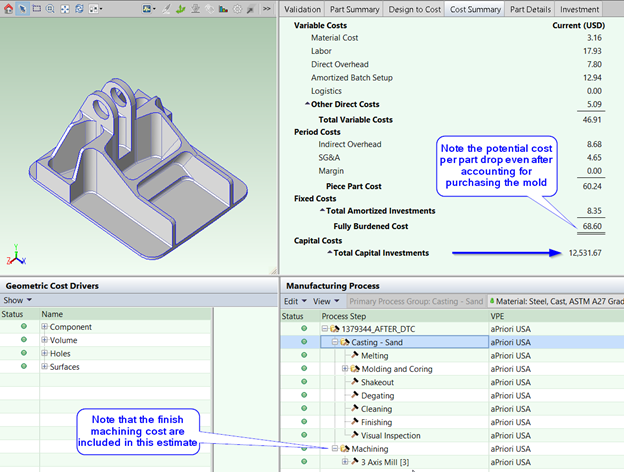
After analyzing the cost difference of approximately $190 per part on 300 parts, which amounted to a potential annual savings of $57,000, the component was redesigned and purchased as a casting, resulting in significant cost savings.
Alternatively, imagine that this part was not a candidate for a casting process due to load and fatigue requirements, as is the part below. The process for reducing costs for the part is similar, except that you need to explore machining costs (some parts may be extruded as well).
Consider how manufacturability issues may be costing you dearly. By evaluating the actual production methods intended for a part, manufacturing insights can identify design features that pose significant challenges. This could involve pinpointing a lack of draft angles, areas with excessive or insufficient thickness, or features that need a side action in plastic injection molding or die casting. For machined parts, issues like sharp corners, obstructed surfaces, or curved surfaces that require ball milling could be highlighted. Addressing these problems early can streamline production and reduce critical costs.
Looking at this part below, we notice a similar ratio of material to conversion cost. And we dig into the features that make it difficult to produce, as casting or extruding it is not an option.
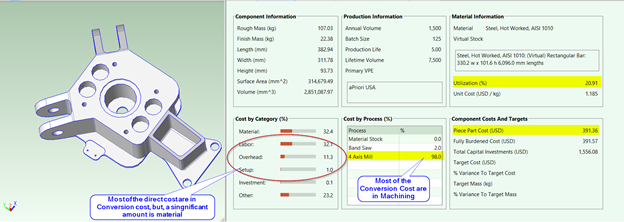
In the interest of time, we will limit ourselves to resolving as many of these L/D ratios as possible. The engineer realizes that the corner radius of those pockets is small, requiring a small tool diameter selection that violates customary L/D ratios and causes slower finishing times. He has the liberty to make those bigger, which won’t change the material consumed. See the figure below for the redesigned part.

Larger corner radii allow for larger diameter selection, which increases the tool’s ability to reach further down without shaking. Cycle time drops, and cost goes down. A 17% cost reduction is certainly worth the effort of the redesign.
6) Other Methods for Cost-Effective Design for Manufacturability
It is possible to affect the size of a machine in manufacturing by considering the design of the part. For example, suppose a part is being produced in China, where labor costs are low, but overhead costs are high due to the use of large, expensive machines. In that case, it may be worth considering features that can influence machine selection.
The die-cast part below has a web in the middle that is not functionally necessary. This web is causing the part to require two-side cores, one on each side. If the web were removed, only one core would be needed, the mold base size would decrease, and the machine size (tonnage) would go down, causing a reduction in tooling and piece part cost with a smaller machine/lower overhead rate. Additionally, you may be able to have more cavities now, which is a big plus if this is a high-volume part.
The number of set-ups can dramatically affect the cost of a low-volume part. A hole that can’t be accessed from an already available set-up direction (aPriori can show you those) can cause an extra set-up.
Too many of these will require a more expensive machine, for example, forcing a move from a 3-axis to a 4-axis or 5-axis. Did you know that if your sheet metal part has an acute angle bend and an obtuse angle bend on the same part, then two bend breaks will have to be set up to make it? This may have minimal cost impact if the part is produced in large volumes, but if this is a low-volume part, it could create serious cost inefficiencies.
7) Learn More About the Power of Digital Manufacturing Insights
DFM is pivotal to identifying cost savings from the initial product design through material selection and manufacturing. By integrating aPriori’s advanced manufacturing insights, product engineers gain a deeper understanding of how seemingly small variables can significantly impact cost and other factors.
This approach provides product design and cost engineers with clear visibility and automated guidance to make informed decisions that enhance both product quality and profitability. The adoption of DFM best practices, supported by aPriori’s insights, can ensure that products are designed for performance, profitability, sustainability, and market success.
This post was originally published on Aug. 12, 2020, and updated on April 18, 2024.
Ready to Unleash the Power of DFM?

More Resources:
- Podcast: How Design for Manufacturing Saves $Millions
- Video: Design Engineering Tips for Reducing Mfg. Costs on New Product Designs
- Webinar: Product Design – Reduce Cost, Carbon, and Risk
Like what you see? Share on:

Subscribe to the aPriori Blog!

What to Look for in Manufacturing Costing Software

Product Engineering: The Road to Achieving a $25K Electric Vehicle

Optimize Your Strategic Sourcing: Our New Report Shows You How

What Is Design to Cost? An Overview With Examples

6 Metrics That Will Help Improve Your Engineering Productivity

How to Conduct a Spend Analysis: Methodology in Detail
- Case Studies
- Customer Reviews
- Manufacturability
- Profitability
- Sustainability
- Design to Cost
- Design for Manufacturability
- Design for Sustainability
- Faster RFQs
- Should Cost Analysis
- Cost and Value Engineering
- Design Engineering
- Manufacturing Engineering
- Quoting Professionals
- Sourcing Professionals
- Sustainable Engineering
- By Industry
- Aerospace & Defense
- Auto & Transportation
- High Tech Electronics
- Industrial Equipment
- Manufacturing Insights
- Digital Factories
- Manufacturing Process Models
- Regional Data Libraries
- Unique Platform Features
- aP Analytics
- aP Generate
- aP Workspace
- News & Events
- Request a Demo
About the Author
- mindful design
- student success
- product design
- ui/ux design
- watch me work
- design careers
- design inspiration
10 Exceptional Product Design Portfolios with Case Study Breakdowns
After working with many designers throughout my own career and helping many more build their job-ready portfolios, there are a few designers that I keep coming back to for inspiration and some that are inspiring a new generation of UI/UX and Product Designers to enter the field.
I've chosen 10 of our favorite UI/UX and Product Designers—a colorful tapestry of digital product architects that have evolved from graphic designers, marketers, architects, engineers and everything in-between. Their unique backgrounds and journey bring something special to our industry and illustrate how we can all do more meaningful, interesting and impactful work:
There are 10 things in particular that make these 10 designers really stand out:
- They have each honed their craft from the bottom up (whether having gone through a traditional academic program, an online course like DesignerUp , or being self-taught).
- They are all at different stages of their careers (some newly minted and others seasoned veterans).
- They continue to learn, grow, push the envelope, document and share their genuine experiences.
- They each hail from a different background (and sometimes non-design industry) that informs who they are and what they work on as a designer.
- Their evolution is apparent in their work.
- They are passionate about the problems they solve and find joy in connecting with the users they serve.
- They are transparent about their processes, thoughtful in their communication about it and not afraid to show what worked and what didn't.
- They have focused portfolios that reveal their unique point of view as a designer.
- They are a diverse group of designers from different cultural, gender and socio-economic backgrounds.
- They have so much to teach us all about design and how to use it to express authenticity and to understand and help others do the same.
Case Study Format: http://simonpan.com/work/uber/
- The Challenge
- Early Insights
- Reframing the Problem
- The Redesign
- Design Strategy
“In a city as busy as San Francisco, over $1 million was wasted per week because of problematic pickups.”
Madeline Wukusick
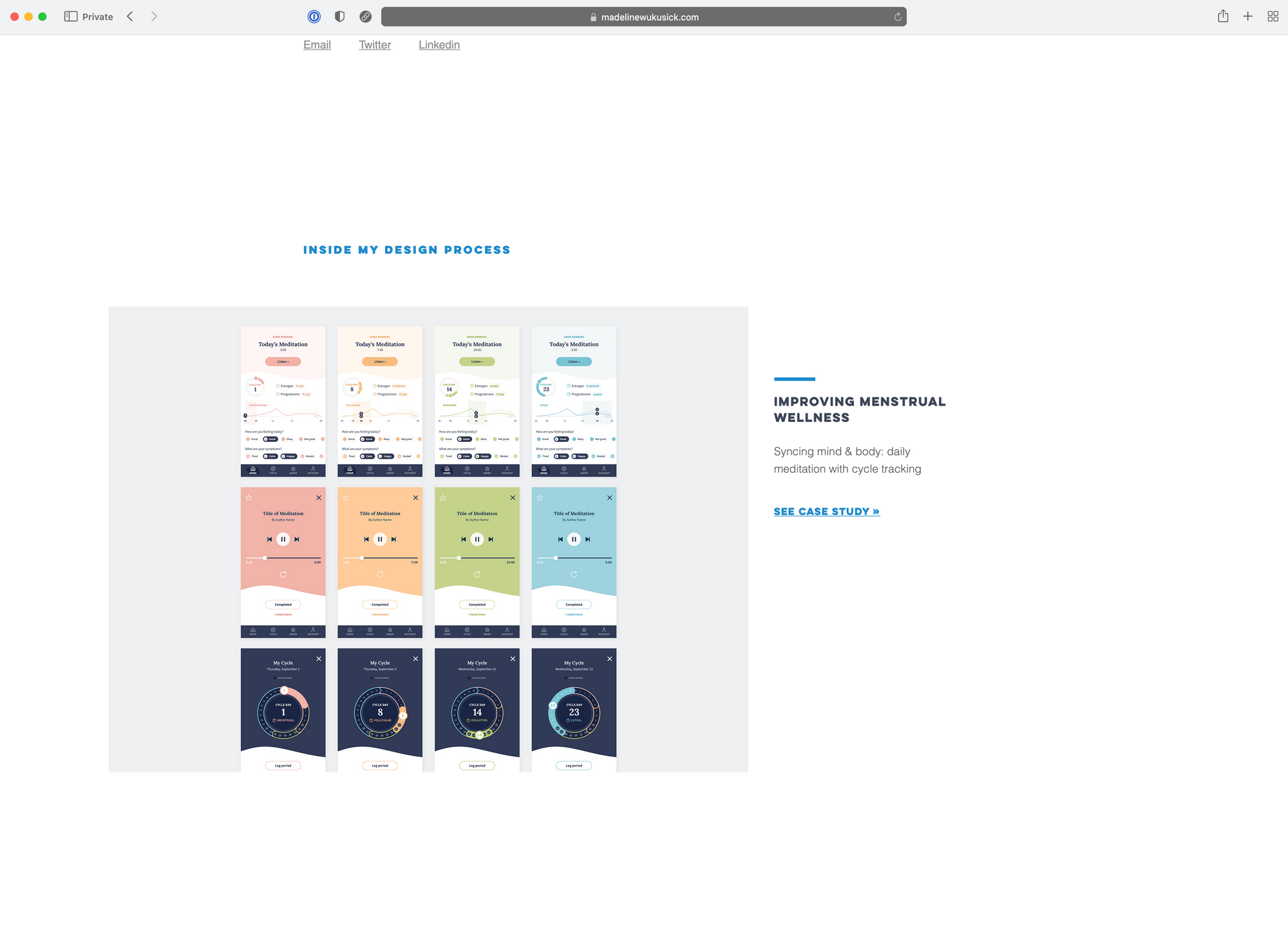
Madeline is a graduate of our DesignerUp Product Design course. She was able to create an incredible portfolio working through our curriculum, blended with her background in graphic and data design that set her up for immediate success landing professional design roles.
Case Study Format:
- The Observed Problem
- The Research
- In the Insights
- The proposed How Might We Statements
- Lean Canvas and Product Strategy
- Business Requirements
- The Solutions and MVP Features
- Things that could be improved
"Thanks for helping me work through these iterations—it's been tremendously helpful! You have such a knack for fine-tuning and teasing out subtle themes that I hadn't noticed before. From these comments, I have a better sense of some of my growth areas to work on and ways in which to push myself. It also helped me realize that I am most interested in hybrid roles, or at least roles with a strong visual component. Really grateful to have discovered this course :-)" - Madeline
Not an Italian mobster; Johny Vino is an engineers' designer. I've been a long time admirer of his work every since his mind-blowing animations and micro-interactions arrested me mid Dribbble scroll many years ago. He is a thoughtful, meticulous designer that understands how to align user and business goals all while transmuting conventional interaction patterns into something that is altogether transcendent yet familiar.
Case Study Format: https://johnyvino.com/
Process, Goal and Task Oriented that varies with each project
- What he worked on
- What he aim to accomplish
- Business Goals
- Representation of complex data
- Integration
Humans are not perfect. I like to apply 3 principles to ever product I design to help me focus on that. Fitt's Law, Mimicry, Aesthetic Usability Effect
Steph Parrott
Steph is a product designer based in Toronto. Currently working on Plantd and most recently at Square in San Francisco.
Case Study Format: https://www.stephparrott.com/plantd/
- Roles and Process
- App Overview
- Feature proposal
- Design to Development
- Looking to the future and what's next
"As someone who hasn’t eaten meat in almost 20 years, I’m highly motivated to put in the work to find plant-based options, but for those starting to dabble, how can we except them to do the same?" - Steph
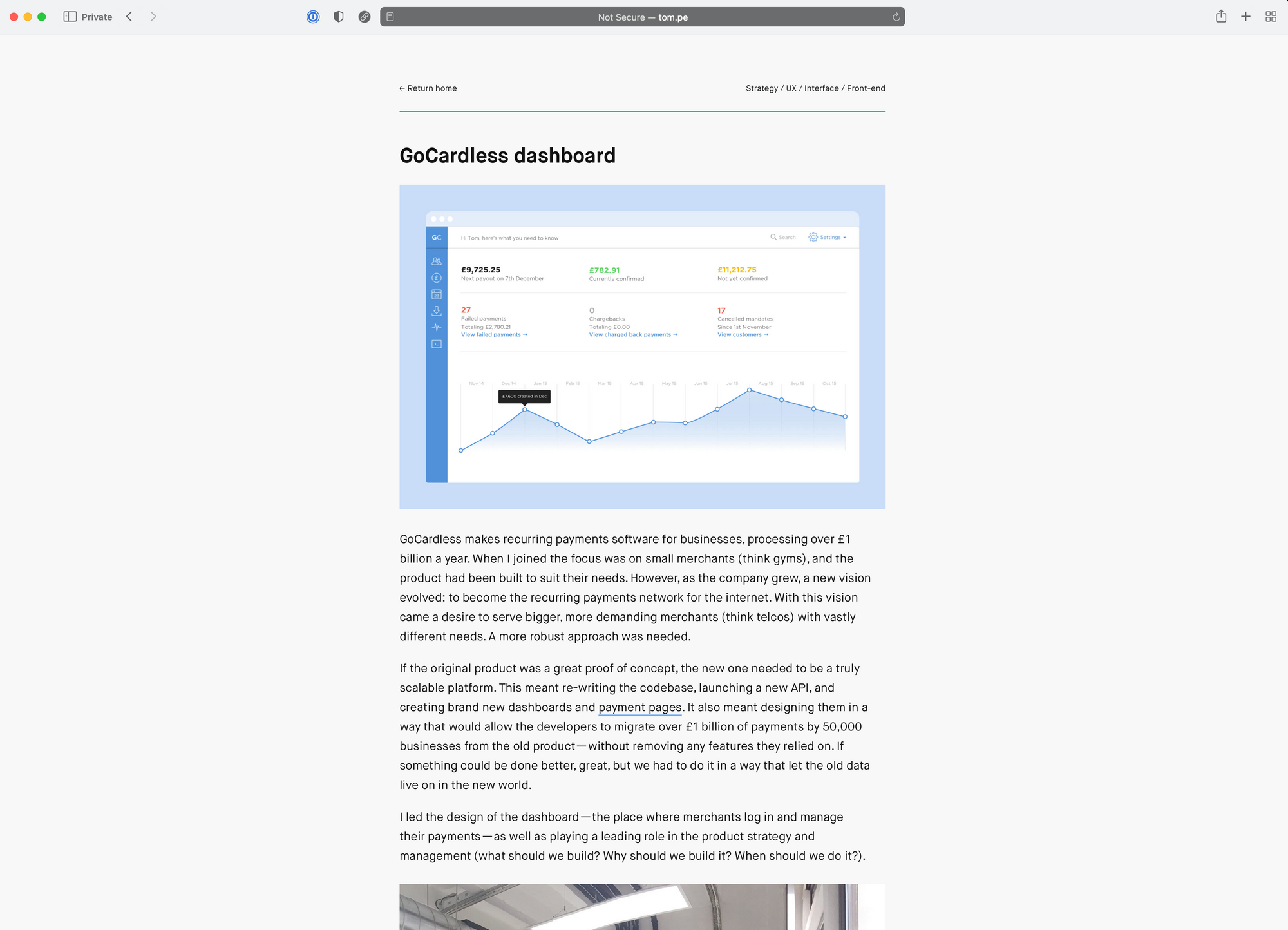
Tom is the co-founder of the community and event series Design Club , and an investor & advisor to Bricklane . He currently works own clients, helping invent, design, and launch new ventures. Before that he held design leadership roles at fashion and fintech startups, and was as a senior designer at a global agency. Case Study Format: http://tom.pe/gc-dashboard.html
- Summary of the problem space, challenges, project and contribution
- Goal and Problem
- Design Principles applied
- Proposed solution and representation of dashboard and data
- Design frameworks used
- Future considerations
"The problem here is that by trying to create something for everyone, we risked helping no one. Avoiding the design equivelant of an identity crisis became a big focus of mine. I'd do this by finding ways to inject a point of view into the product. By knowing what it wasn't, as well as what it was." - Tom Petty
Want to create an incredible portfolio like these full of amazing case studies to get you hired? Enroll in our Product Design Course today!

Garett MacGillivray
Much like myself and other designers of a particular generation, Aussie/Canadian Garett MacGillivray has been around the block and through the evolution of graphic designer, web design, UI/UX Designers and now landed squarely as a full stack Product Designer.
Case Study Format: https://aucadian.com.au/project/goloop
- Exploration and Ideation
- Component Library
- B2B product interface
I've had many labels throughout my career in the digital industry. It's safe to say that I enjoy crafting digital experiences.
Elise is a Bay Area designer that jumped to the bay from NYU. She comes from an advertising and marketing background and has fine tuned her communication skills and processes vast knowledge of the tech and digital product industry has a whole having been on the broadcasting and marketing side of things.
Case Study Format: http://www.elisefu.com/work-komeeda/
- Impact/Metrics
- User testing
- User research
- Information Architecture
- Implementation
- Major Findings
- Formal User Testing
I was driven to design because I felt excited and rewarding to learn about people’s goals and desires, help them solve problems and make their lives easier.
Latiesha Caston
Latisha is a User Experience Designer passionate about holistic, accessible, and inclusive design, based in Seattle.
Case Study Format: https://www.latieshacaston.com/veggie-grill-online-ordering-experience
- The Problem
- The High-level Goal
- The approach
- Pain Points
- Optimizing flow and improving architecture
- Interaction model breakdown
- Looking to the future
"Our high level goal was to design a holistic order-ahead experience that keeps the core of what we've built, while delivering on opportunities, addressing pain points, and setting the stage for the future." - Latisha
Karolis Kosas

Having recently joined the amazing design team at Stripe as a Product designer, Karolis's portfolio is clean and minimal and his case studies really understand the mental model of the user, getting into their heads and revealing the friction points they are feeling and how he can insert a solution that improves on the experience with compromising the soul of the designer.
Case Study Format: http://karoliskosas.com/cinemaclub/
- prototyping
- Built the product from zero
Visual communication is a self-sufficient organism capable of adapting and evolving based on the input of multiple sources.Acting in such context, the designer is an initiator, establishing methods and boundaries for the system to establish itself as an independent entity.
Rohit Singh
One of o ur very own graduates ! Rohit Singh is an up and coming product designer with a focus on helping early-stage startups and new businesses blossom.
Rohit outlines his process for creating an MVP for his digital product from scratch, which serves as a sort of physical and virtual library for the poorest class of India.
Case Study Format: https://work.khadush.in/booksite-an-online-physical-book-sharing-platform/
- Inspiration
- User Interviews
- Lean Canvas
- Visual Design
- Final Words
I specialize in helping early stage startups validate their riskiest assumptions using leading design methods
After analyzing all of these case studies and working with 100s of designers in our product design course to get them ready for the job application process, we've created our own tried-and-true templates to make it easy for designers to replicate the successful format and structure of these top portfolios using Notion .
Each of these amazing portfolios tells the story of the product designer, their evolution, their process and shows what they bring to the world. But it's not easy doing what they do or knowing exactly how to show and tell who you are as designer. Having a solid design education and getting feedback from the design community is the best way to ensure that your work is up to par and being presented in a way the shows your skills and your worth.
Have a look at our partners' advice over at Pathrise on building a strong UX design portfolio .
The best design resources, in your inbox
Tips, tricks, articles and freebies. It's all happening in the DesignerUp Newsletter. View the archives →
We'll only send the occasional email and promise not to spam.
© Copyright 2022 DesignerUp. All Rights Reserved.
🔥 Upgrade to a new style of analyzing VoC with sentiment checks, video clips, custom reports & more!
.png)
6 Product Management Case Studies You Can't Miss

Associate Product Marketer at Zeda.io.
Mahima Arora
Created on:
June 26, 2024
Updated on:
8 mins read
.webp)
Transform Insights into Impact
Build Products That Drive Revenue and Delight Customers!
Product management case studies are detailed analyses of how a product was conceptualized, developed, and marketed. A typical product management case study contains the following:
- The pain points and expectations of the user
- Competing products in the market
- Development , delivery, and iteration methods
- Marketing strategies implemented to relay the product’s value proposition
- How the product was received
- Lessons for the product team
So, why should you learn about the development of a product in so much detail? The answer lies in the sixth bullet.
Let’s look at how reading case studies related to product management can help you.
How product management case studies help you
Here’s why reading product management case studies is a worthwhile investment of your time. A well-written case study:
- Gives you an in-depth understanding of real product problems : Meeting or exceeding the expectations of the customers is always challenging. Whether it is technical complexities, budget limitations, or organizational miscommunication, a case study helps you recognize the source of the problem which led to the development of a less-desirable product.
- Contains practical insights outside of the theory : Even a layman can learn the steps of SaaS product management . However, seasoned product managers know that developing a successful product takes more than learning the development steps. These case studies contain tons of real-life scenarios and the lessons that come with them.
- Educates you and makes you a better product manager: Product management case study examples take you through the journey of developing a product, which helps you improve your existing approach toward product development. You will also learn better ways to manage your team and resources.
In simple terms, a product management case study helps teams learn lessons that they can emulate to develop a more profitable product.
In this article, let’s look at six product management case studies that are a must-read for every product manager.
1. Slack: Initial product launch strategy

Stewart Butterfield started a gaming company called Tiny Speck to change the world of massively multiplayer online role-playing games (MMORPG). Him and his team created Glitch which was quite different from other games in that genre such as World of Warcraft.
Glitch was a 2D game that did not have the violent aspects that typical MMORPG games had at the time. It allowed extensive character personalization and Butterfield described it as “Monty Python crossed with Dr. Seuss on acid”.
While building Glitch, Butterfield and his team used the Internet Relay Chat (IRC), an online chat tool popular in the 80s and 90s. However, it fell short as the team found it difficult to keep track of past conversations, which motivated them to build their own communication tool.
As they developed Glitch, their internal chat tool gained more features based on their needs.
Despite lots of support from investors, Glitch was unable to attract enough players to keep running profitably and Butterfield eventually shut it down in 2012 .
After six months, in early 2013, Butterfield renamed their internal communication tool Slack - acronym for Searchable Log of All Conversation and Knowledge and requested his friends and colleagues to try it out and give feedback — they all loved it.
By May 2013, Slack was ready for the big reveal which posed a new challenge — executing the perfect launch strategy to drive demand.
Slack’s Challenge: Nailing the initial product launch
While launching an app that can have such an impact on how organizations work, it is crucial to get it right. At the time, there weren’t many team messaging apps and most teams had conversations via email.
Slack needed a significant number of early adopters to validate their hypotheses about team collaboration and collect data that will help them improve its services further. Consequently, this increased the stakes for the first launch.
How did Slack do it
CEO Stewart Butterfield revealed that on the first day of the launch, Slack welcomed 8000 new users which rose to 15000 at the end of the second week. The credit for this initial success, he explains, went primarily to social media.
Social media helped Slack deliver its PR pieces through its genuine users. This led to a snowballing effect because people interacted with people.
Slack recorded over 18 million active users in 2020.
Although the impact of social media-based word-of-mouth marketing will have different levels of success as it depends on factors such as the type of product and its use cases, you should have a social media marketing strategy to spread the word.
Suggested Read: Leveraging VoC-driven AI Insights to Build Revenue-generating Products
2. Superhuman: Finding product-market fit

Superhuman is a premium email service for busy teams and professionals who need more of everything; speed, usability, and personalization. Apart from superb design, Superhuman processes and executes any request within 100ms.
Rahul Vohra built Rapportive in 2010 — a plugin that adds social profiles to Gmail which was later acquired by LinkedIn . This gave Vohra an intimate view of email and quickly realized that things will progressively get worse.
In his words, “I could see Gmail getting worse every single year, becoming more cluttered, using more memory, consuming more CPU, slowing down your machine, and still not working properly offline.”
He also brought attention to the number of plugins people used, “And on top of that, people were installing plugins like ours, Rapportive, but also Boomerang, Mixmax, Clearbit, you name it, they had it. And each plugin took those problems of clutter, memory, CPU, performance offline, and made all of them dramatically worse.”
Vohra had one question in his mind — how different would the email experience be if it was designed today instead of 12 years ago?
Superhuman was born to give professionals the email experience that they have been long waiting for. Smooth, easy on the eyes, and most importantly, blazingly fast.
But, there was one elephant in the room.
The idea of building a better email service than the existing players sounded great. However, going against some of the biggest brands of Silicon Valley required more than a bad personal experience with Gmail.
The Superhuman team needed evidence that such a product is actually desirable.
Superhuman’s Challenge: Establishing product-market fit
The team at Superhuman was competing against the email services of Apple, Google, and Microsoft which made the product-market fit quite crucial.
But how do you know whether you have achieved product-market fit?
How did Superhuman do it
Vohra and his team came up with an innovative idea to measure product-market fit by testing crucial hypotheses and focusing on the right target audience.
Superhuman had two hypotheses :
- People are dissatisfied with Gmail and how slow it is.
- People are also dissatisfied with third-party email clients and how buggy they were.
In a product management case study , Vohra explained how to find the right audience — the users who would be ‘very disappointed’ if they could no longer use your product. After identifying them, all you have to do is build the product as they want it.
3. Medium: “Highlights” feature

Evan Williams co-founded Blogger and Twitter which has helped millions of people share their thoughts with the world. Although both platforms became quite popular, they still couldn’t deliver the best reading experience to their users. Blogger allowed readers to browse topics by authors only and Twitter made it difficult for authors to aptly describe themselves.
He quickly recognized the need for a publishing platform that delivers a diverse experience for the readers and allows the authors to speak their hearts.
That’s how Medium was born. It enabled readers to browse articles by topics and authors, helping them to gain different perspectives on any particular subject. It also allowed everyone from professional programmers to amateur chefs to share their insights with the world as they wanted it.
The developers slowly added more features to Medium such as tags, linked images, social cards, and sharing drafts as it evolved through the years.
One of the many notable features of the platform is the “Highlight” feature — where you can select any particular post section and treat it as a mini-post. You can comment on the Highlight or tweet it, which is handy for both personal revision and sharing interesting snippets with others.
Suggested Read: Want to become a Product Coach?
Medium’s Challenge: Determining whether “Highlights” added value
Medium faced a challenge while determining a metric that can give them an accurate assessment of the desirability of this feature. In other words, they needed a metric that would tell them whether the “Highlights” feature made user interactions better and more rewarding.
How did Medium do it
The team at Medium solved the challenge by shifting their focus to one crucial metric rather than multiple vanity metrics such as organic visits and retention time which signifies how much value your users are getting out of your product based on retention rate.
For Medium, it was Total Time Reading (TTR) . It is calculated by estimating the average read time which is the number of words divided by the average reading speed (about 265 WPM) and adding the time spent by the reader lingering over good paragraphs by tracking scrolling speed.
4. Ipsy: Managing distribution

Michelle Phan started her journey as a YouTuber who recognized the importance of makeup in someone’s self-expression. She has been sharing beauty tips and makeup tutorials with her audience since 2007.
While on a trip to Thailand, she observed how little girls scrambled to pay for makeup samples in front of vending machines. Five years later, she launched a subscription-based Glam Bag program — where the customers will receive 4-5 deluxe-sized samples of makeup products.
MyGlam, as it was known back then, quickly gained over half-a-million monthly subscribers which created one of the biggest online beauty communities.
Phan quickly realized what she wanted to do — to build a brand for women who wanted to share their perspectives on beauty and meet like-minded people with similar interests and styles.
Ipsy , which comes from the Latin root “ipse” meaning “self”, was created by Phan, Marcelo Camberos, Jennifer Goldfarb, and Richard Frias to expand the user experience.
Although Phan knew how to convert viewers into paying customers, executing a marketing strategy by scaling it up was challenging.
Ipsy’s Challenge: Managing a content distribution strategy
The first makeup tutorial by Michelle Phan has now over 12 million views. Videos like that helped Phan get her first subscribers on her MyGlam program.
This shows the importance and impact of influencer-led content on revenue for businesses in the beauty industry.
However, running an influencer content distribution strategy involves collaborating with multiple passionate influencers. It was challenging to find like-minded influencers who will promote only one brand. Moreover, when working with influencers, it's important to implement effective content moderation to make sure the posted content aligns with your goals.
Phan and her team had a simple solution for this.
How did Ipsy do it
Phan and Spencer McClung, EVP of Media and Partnerships at Ipsy, partnered with beauty influencers like Bethany Mota, Promise Phan, Jessica Harlow, and Andrea Brooks who were already subscribed to MyGlam to create content exclusively for Ipsy.
In a case study analysis, McClung revealed that it put Ipsy on a content-based growth loop where the content was created by both the influencers and customers for the beauty community.
Sponsored content for products by influencers helped them increase their reach and helped Ipsy get more loyal customers. This growth loop gained Ipsy over 3 million monthly subscribers .
Suggested Read: Pivoting equals failure?🤯
5. Stitch Fix: Mastering personalization

Katrina Lake, the founder of Stitch Fix , realized back in 2011 that apparel shopping needed an upgrade. eCommerce failed to meet the expectations of the shoppers and retail shops were falling short in terms of options.
In an interview with The Cut , she revealed "Searching online for jeans is a ridiculously bad experience. And I realized that if I imagined a different future, I could create it."
After realizing that no one has merged data and fashion shopping, she set out to make a difference. She started a personal styling service out of her apartment in 2011 when she was pursuing her MBA from Harvard.
Lake relied on SurveyMonkey to keep track of her customer’s preferences and charged $20 as a styling fee. In late 2012 Eric Colson, then the VP of data science and engineering at Netflix, joined Lake on her journey of crafting the future of retail.
Lake and Colson wanted to give their customers much more than just personalized recommendations.
Stitch Fix’s Challenge: Building a personalized store
Stitch Fix wanted to give their customers more than just personalized recommendations — they wanted to build a personalized store for them where everything they look at, from clothes to accessories, matches their flavor.
But everyone’s body dimensions, preferences, budgets, and past choices are unique which can make building a personalized store difficult.
The team at Stitch Fix found a simple yet effective solution for this challenge.
How did Stitch Fix do it
Katrina Lake, CEO of Stitch Fix, revealed in a case study that personalization is crucial for the onboarding, retention, and monetization of customers.
When signing up, Stitch Fix asks you a few questions about your fashion choices and picks clothes that look the best on you. Furthermore, the collections in your personal store will keep improving as it continuously learns more about your personal preferences.
Also, there is no subscription fee which makes Stitch Fix a great option for occasional shoppers. Suggested Read: Canva’s Success Tale in the World of Design
6. Pinterest: User retention

Ben Silbermann started his tech career at Google’s customer support department. Although he loved the company and believed in its vision, he quickly became frustrated as he wasn’t allowed to build products.
With support from his girlfriend (now wife) Divya and a college friend Paul Sciarra (co-founder), Ben created an app called “Tote” in 2009 which was described as a “catalog for the phone”. Tote allowed users to catalog their favorite items and will be alerted whenever they were on sale so they can make a purchase.
However, the users used it to share their collections with each other instead. Ben recalled how he collected insects as a kid and loved sharing his collection with others. He recognized how people, in general, love to do that.
And, just like that, Pinterest was born where users can “pin” whatever they are interested in and add it to their personal collections.
Pinterest quickly became a hit and entered the global market.
Despite huge success within the US, Pinterest struggled to retain users globally. The team realized that the primary reason users churned is that something stopped them from getting the product’s core value — building personal collections.
Pinterest’s Challenge: Helping customers quickly realize the core value
There are many things that can prevent a user from accessing a product’s core value and one of them is internal friction within the product.
Pinterest’s product folks zeroed in on the one feature that was the gateway to the product’s core value — the “Pin It” feature.
Users outside the US simply couldn’t relate to the term, even though all it did was save the item they like to their personal collection.
How did Pinterest do it
The “Pin It” feature of Pinterest is linked directly to its brand identity. Casey Winters, former growth product lead at Pinterest, suggested changing it to “Save”, particularly in areas outside of the US.
As of the third quarter of 2022, it has over 445 million monthly users all over the world exploring various “ideas” to build collections for sharing with their friends.
Casey concludes in the product management case study that checking whether the users are getting your product’s core value is pivotal in solving most of your growth challenges.
Key Takeaways
Case studies for product management contain in-depth insights that help product teams improve their approach toward their product’s ideation, analysis , development, and commercialization.
The six product management case study examples we reviewed above give these crucial insights:
- Slack : Don’t forget to use social media for marketing your product before its launch.
- Superhuman : Focus on the users that will be “very disappointed” if they can’t use your product anymore to achieve product-market fit.
- Medium : Track the one metric that tells you whether your users are getting value from your product rather than vanity metrics such as organic traffic.
- Ipsy : Partner with influencers to educate your target audience on how to get the most out of your product.
- Stitch Fix : Learn about what your users want and recommend them just that.
- Pinterest : Continuously experiment by changing multiple variables to uncover new growth opportunities.
To put these lessons into practice, you need to provide your team with the right tools that help them interact with your users, learn about their preferences, monitor their usage data, plan the next steps, and manage product development effectively.
Zeda.io is a product management super-app that allows you to do just that. You can run your entire product management process , from ideation to delivery, in one place. Zeda.io comes with over 5000 integrations with Zapier, enabling you to hit the ground running in no time.
Start your free trial today . Also, looking for the latest trends in AI, UX, product management, and startups? Join our biweekly newsletter now! We distill complex topics into actionable insights just for you. Hit the 'Subscribe' button and never miss out on these valuable updates. Act now – because in the fast-paced world of tech, staying ahead matters! Subscribe here.
- What is a product management case study?
Answer: A product management case study is a detailed analysis of how a product was developed and iterated over time for maximum success. These studies help product managers learn from others and improve their own approach toward product management.
- How do you prepare a product management case?
Answer: You can prepare a product management case study in four steps — understand customer needs, monitor the stages of development, identify the factors that affected the course of product development, and extract takeaways.
- What are the 3 major areas of product management?
Answer: Discovery — recognizing the need for a product, planning — creating a roadmap to plan the product’s development, and development — the various sprints through which a product is developed are three major areas of product management.
- What are the 7 steps of product planning?
Answer: Concept development, competitive analysis, market research, MVP development, introduction, product lifecycle, and sunset are the seven steps of product planning.
- What are the 5 dimensions of product management?
Answer: Reliability, usability, functionality, maintainability, and efficiency are the five dimensions of product management.
- What are the 4 P's of product management?
Answer: Product, price, place, and promotion are the 4Ps of product management which represent four crucial aspects product teams should simultaneously focus on while developing a product.
- What are the 5 phases of the product management process?
Answer: Idea generation, screening, concept development, product development, and commercialization are the five phases of the product management process .

Join Product Café Newsletter!
Sip on the freshest insights in Product Management, UX, and AI — straight to your inbox.
By subscribing, I agree to receive communications by Zeda.
IN THIS ARTICLE:
Latest articles

Product Roadmap Prioritization: How-to Guide (With Frameworks)
Learn the basics of product roadmap prioritization and make informed decisions on what features to focus on next. explore the critical elements of a prioritized product roadmap, equipping you with tools and frameworks to make informed decisions.

5 Best Product Analytics Tools for Data-driven Success
Discover the five best product analytics tools that will help you elevate your business strategies, acquire customers and boost retention.

PM’s Guide to Nail User Experience Research
We’ve created a start-to-finish guide on user experience (UX) research for every user-focused product manager. So, dive in
AI-powered product discovery for customer-focused teams

- Product Engineering

NewVision Team
Previous post cloud infrastructure, next post digital transformation, leave a reply cancel reply.
Save my name, email, and website in this browser for the next time I comment.

Follow us on
Product Engineering Cloud & Infrastructure Data Engineering & Analytics Digital Assurance Intelligent Automation Platform & Technology Competency Oracle Utilities
About NewVision Careers Topics
USA - Atlanta | Palo Alto
India - Pune | Hyderabad | Bhopal
Middle East - Dubai
Phone: +91 - 7030048266
Connect With Us
For Sales/Business Related Inquiries: [email protected]
For Career Related Inquiries: [email protected]
Copyright © 2024 - NewVision Softcom & Consultancy Pvt. Ltd.
Privacy Policy | Terms & Conditions
- Our Approach
- Cloud & Infrastructure
- Data Engineering & Analytics
- Digital Assurance
- Intelligent Automation
- Platform & Technology Competency
- Oracle Utilities
- About NewVision
- Case Studies
.png)
How to solve product management case studies
Preparing for a product manager interview can be a daunting task. With case studies being a critical component of the interview process, it's important to strategize and practice ahead of time to showcase your skills effectively. In this comprehensive guide, we'll cover actionable tips to help you ace product manager case study interviews. From key frameworks to avoid common mistakes, you'll gain the confidence and knowledge needed to land your dream PM role. Whether you're just starting your PM career or a seasoned pro, read on to level up your case study interview skills.
Here at The Product Folks , we're all about empowering product managers to grow in their careers. With resources like case study workshop recordings, mock interviews, and dedicated mentors, we're here to help you put your best foot forward in the interview process. Let's dive in!
Overview of the Product Manager Interview Process
A typical PM interview will include multiple stages designed to thoroughly assess your abilities. Here's what to expect:
- Screening call: Discuss your background and interest in the role. Helps align expectations.
- Case studies: Presented with a hypothetical product challenge to analyze and propose solutions for. Tests core critical thinking and prioritization skills. Varies from estimating market size to designing new features.
- Behavioral questions: Queries about your past experiences working on teams, managing stakeholders, handling conflicts etc. Screens for culture fit.
- Technical questions: Assesses your technical knowledge depending on the product domain. More critical for engineering-heavy roles.
Case studies are particularly crucial, as they demonstrate key skills like structuring complex problems, analyzing tradeoffs, and communicating recommendations. Common mistakes include lack of a methodical approach and failure to articulate underlying thought process. Avoid vagueness and guide interviewers through your thinking.
For training tailored to acing PM case interviews, check out The Product Folks' mentor-led masterclasses. Their experts explain how to navigate various types of case studies and equip you with frameworks to tackle them confidently.
Key Strategies for Acing Case Studies
Here are proven strategies to shine in your PM case study interview:
- Research the company: Review their products, customers, domain etc. Helps tailor your approach to their context. For example, studying an ecommerce company's key metrics will allow you to anchor examples and data points in their specifics.
- Clarify the case parameters: Confirm goals, assumptions and constraints. Prevents misalignment. Ask clarifying questions upfront to avoid veering off track.
- Structure with frameworks: Use templates like PRD format to organize thinking. Keeps it methodical. Outlining stakeholders, requirements and success metrics provides systematic analysis.
- Show your work: Verbalize your analysis to demonstrate thought process. Explains rationale. Walk through each consideration instead of simply stating conclusions.
- Practice aloud: Helps get comfortable articulating complex ideas. Improves narrative flow. The Product Folks mock interviews are great for rehearsing with real-time feedback.
Walking through an example case study is invaluable for internalizing these key strategies in action:
Example Case Study #1 - Food Delivery App
Let's imagine we're the PM for Swiggy, a food delivery app in India. The CEO wants to grow monthly active users (MAU) by 30% this quarter. Here's how I'd approach this case:
First, I'd propose features that provide more value to users:
- Loyalty program : Points for orders that unlock free delivery and discounts. Improves retention.
- Package deals : Meal combos from multiple restaurants at a bundled price. Increases order value.
Next, I'd consider options to improve new user onboarding:
- Free delivery trial : First 3 orders free delivery to hook new users. Lowers barrier to signup.
- Referral bonus : Users earn credits for referrals. Virality leverages network effects.
Comparing the options, I'd recommend prioritizing the loyalty program for the highest ROI. It targets our core goal of increasing MAU by incentivizing repeat orders. According to Swiggy's metrics, existing users drive 80% of orders, so loyalty has the biggest leverage. The referral bonus is more speculative and might require substantial promo budget.
Key risks include existing users thinning order frequency to earn points. We'd need to analyze optimal program tiers and rewards, likely by running A/B tests. Overall, the loyalty program combines high impact on KPIs with ease of implementation. For execution, I'd pilot in Bangalore and Hyderabad first, tracking engagement data to refine the nationwide rollout.
This showcases weighing alternatives against goals, evaluating feasibility and mapping execution steps - all critical PM case study skills. Let's break down another example next.
Example Case Study #2 - Social Media Platform
Imagine we're PMs at Facebook. Engagement from teenagers in the US has dropped 30% this quarter. How can we turn this around?
I'd start by auditing their core needs - sense of identity and community. Some potential solutions:
- Interest-based groups : Connects users with niche interests. Provides targeted sense of belonging.
- Ephemeral content : Stories, polls that disappear after 24 hrs. Creates constant activity.
- Rewards program : Points and badges for engagement milestones. Gamification taps into motivation.
Evaluating the options, interest-based groups seem most promising. While ephemeral content may spike engagement short-term, it likely won't address the root identity needs long-term. Groups are scalable and tap directly into the teenage affinity for communities.
I'd propose a pilot targeting groups around hobbies, causes etc. Success would see group engagement exceed overall platform averages for the teen demographic. Risks include bullying in unmoderated groups. We'd need community guidelines, reporting mechanisms and moderation.
Overall, this matches an audience need with a targeted solution grounded in behavioral data. We walked through ideating options tailored to goals, analyzing feasibility and defining metrics to track outcomes. These frameworks are key for structuring strategic thinking during case interviews.
Key Learnings and Takeaways
Let's recap the core strategies we covered for tackling PM case studies:
- Research the role and company to frame your approach
- Clarify the objectives, constraints early
- Organize analysis using frameworks
- Explain your thinking to demonstrate logic
- Practice case studies regularly to build skills
Avoiding vague responses and clearly articulating your thought process are critical. Use examples and data to back recommendations. Structure your thinking with proven frameworks.
For further practice with feedback from experts, check out The Product Folks' mock case study interviews. Their dedicated mentors can help take your skills to the next level.
Latest Posts
.jpg)
This article will explore how product management side projects can catalyze professional development by allowing you to experiment with new methodologies and enhance your skillset.
.jpg)
This comprehensive guide promises to equip you with a structured approach to tackling product case studies. You'll gain frameworks to methodically analyze prompts and craft insightful solutions.
.jpg)
Through real-world application, valuable feedback, and community engagement with groups like The Product Folks, PMs can significantly accelerate their skill development and expertise in the dynamic field of product management.
Come For the Content Stay For the Community

Product Case Studies: Examples and Best Practices for Success
Discover the power of product case studies with our comprehensive guide.
Posted May 15, 2023

Featuring Karthik R.
Breaking into Product
Starting friday, september 6.
7:00 PM UTC · 45 minutes
Table of Contents
Product case studies are an important tool that businesses use to showcase their products and demonstrate their value. They are especially crucial for companies that have innovative and complex products that require explanation and demonstration to potential customers. A product case study can help potential customers to understand a product's features, benefits, and the results they can expect when using it. In this article, we will explore the importance of product case studies, how to identify the right products for case studies, tips for creating compelling case studies, and best practices for promoting them.
Why Product Case Studies are Important for Businesses
Product case studies provide businesses with a platform to showcase their products in a real-life scenario and demonstrate how they solve customers' problems. By doing so, businesses can communicate the value of their products to potential customers and build trust with them. According to a study by MarketingSherpa, 71% of B2B buyers read case studies during their decision-making process, making them a highly effective marketing tool. Case studies provide social proof and credibility that inspire others to use the product and generate leads. Additionally, product case studies can be repurposed into blog posts, website pages, social media posts, and email marketing campaigns, giving businesses an ongoing source of content to engage their audiences.
How to Identify the Right Products for Case Studies
The first step in creating a successful product case study is identifying the right product to showcase. The ideal product is one that solves a problem that your ideal customer faces, has unique features that set it apart from competitors and generates positive results. It's important to consider the availability of resources, such as time, budget, and personnel. You also need to assess how representative the product is of your business's value proposition and goals. Finally, consider the potential impact of the case study and how well it aligns with the target audience's interests.
Free trial!

From 135 top coaches
Access a library of videos, templates, and examples curated by Leland’s top coaches.
150+ video guides.

Example Resumes

Exercises & Templates
Technical Skill Development

Tips for Choosing the Best Format for Your Product Case Study
The format of your product case study will depend on the product, audience, and objective of your study. Common formats include written case study, video case study, podcast case study, and presentation format. The chosen format should match the objectives of your study, the target audience's preferences, and your available resources. The format should be well-designed, clear, persuasive, and include all relevant information that the reader or viewer needs to know about the product.
Elements of a Compelling Product Case Study
Effective product case studies share certain elements that make them compelling to readers and viewers. The elements include the background of the company and customer, the problem or pain point that the customer faced, the solution offered by the product, the implementation and usage of the product, and the results achieved by the customer. A good product case study should be well-structured, engaging, and informative. It should have a clear and concise message, a call to action, and be supported by data and quotes from the customer or expert.
Steps to Creating a Successful Product Case Study
The process of creating a successful product case study encompasses various steps that businesses should undertake. The first step is to identify the product, identifying the customers who use it and their needs. The second step is to collect data by researching, interviewing customers and experts. The third step is to create a structure or outline that guides the case study, including the key elements mentioned above. The fourth step is to draft the case study, edit it, and get feedback from customers and experts. Finally, businesses should promote the case study to their ideal audience through multiple channels.
Real-life Examples of Successful Product Case Studies
There are numerous examples of successful product case studies that businesses can use to inspire their strategies. One example is the Dropbox case study, a written case study that showcases Dropbox's product's integration with other services, cost savings for businesses, and customer feedback. Another example is the Hubspot case study, a video case study that focuses on the customer's business challenges, the solution, and the results achieved by their partnership with Hubspot. These case studies are well-written, engaging, and informative, providing valuable insights for potential customers.
How to Measure the Success of Your Product Case Study
After creating and promoting a product case study, it's essential to track its success to improve future strategies. Metrics such as the number of views, engagement, clicks, leads generated, sales, and customer retention rate can provide insights into the case study's effectiveness. Additionally, reviewing customer feedback such as testimonials, ratings, and reviews can give businesses valuable insights into the impact their product case study had on customers.
Best Practices for Promoting Your Product Case Studies
After creating a product case study, it's critical to promote it to reach your ideal audience effectively. Best practices for promoting your product case studies include using multiple channels such as social media, email marketing campaigns, press releases, website pages, blog posts, and paid advertising. Additionally, segmenting the audience based on their interests and preferences can increase engagement and lead generation. Finally, businesses should measure and analyze the metrics to adapt their strategies based on the case study's feedback.
Common Mistakes to Avoid in Creating Product Case Studies
Creating compelling and effective product case studies can be challenging, and it's essential to avoid common mistakes that can hinder their impact. Common mistakes include failing to target the right audience, not having a clear message or value proposition, making the case study too sales-oriented, or lacking concrete data and statistics. It's crucial to have a thorough understanding of the product, the customers, and their needs, and providing an objective evaluation of the results to avoid these pitfalls.
How to Use Customer Feedback in Your Product Case Studies
Customer feedback is an essential source of insights for businesses that want to create engaging and effective product case studies. The feedback can be collected through customer satisfaction surveys, interviews, and reviews. By incorporating customer feedback in product case studies, businesses can improve the credibility of the study, provide social proof and build trust with potential customers. Additionally, customer feedback can help businesses to improve their products, services, and marketing strategies based on customer needs and preferences.
The Role of Storytelling in Creating Effective Product Case Studies
Storytelling is a powerful tool in creating compelling and persuasive product case studies. By telling the customer's story, businesses can connect emotionally with potential customers and demonstrate the benefits, value, and relevance of the product. Storytelling can also make the case study more engaging, memorable, and relatable. The story format can help simplify complex concepts and make it easier for customers to understand the product's features and benefits.
Tips for Conducting Interviews with Customers and Experts for Your Product Case Study
Conducting interviews with customers and experts is a crucial step in creating accurate and informative product case studies. Tips for conducting successful interviews include preparing a structured agenda or script, identifying the right experts and customers, asking open-ended questions, listening actively, taking detailed notes, and following up after the interview. By conducting thorough and well-prepared interviews, businesses can gather valuable insights, quotes, and data that can help shape the product case study effectively.
How to Incorporate Data and Statistics in Your Product Case Study
Data and statistics can provide valuable insights that justify the value and impact of the product being showcased in the case study. When incorporating data and statistics in a product case study, it's essential to use credible and reliable sources, present the data in a clear and concise format, and link the data to the customers' needs and challenges. Data and statistics can also help businesses to identify trends and patterns in their customer behavior and preferences, leading to better marketing strategies and product development.
The Benefits of Using Video in Your Product Case Study
Video is a powerful and engaging format that can increase the impact and reach of product case studies. Video case studies can offer a more immersive and engaging experience for potential customers, allowing them to see the product's features, benefits, and value in action. Video case studies can also be easily shared across multiple social media platforms, generating greater brand awareness and recognition. Additionally, video case studies can provide visual data, graphs, and diagrams that can be more impactful than written or spoken testimonies.
How to Leverage Social Media to Amplify your Product Case Study
Social media is a powerful tool that can be used to amplify the reach and engagement of product case studies. Tips for leveraging social media include identifying the right social media platforms, creating shareable content that resonates with the audience, using relevant hashtags, tagging influential people in the industry, and promoting the content to targeted audiences. Social media can also be used to generate feedback, encourage testimonials, and gain insights into customers' views and opinions.
The Importance of A/B Testing in Optimizing your product case study
A/B testing can provide valuable insights into how potential customers interact with product case studies and what elements are most persuasive. A/B testing involves creating two versions of the product case study, each with a slightly different element, such as colors, headlines, or calls to action. By measuring how customers interact with each version, businesses can identify which elements are most effective and optimize the case study accordingly. A/B testing can lead to increased engagement, conversion rates, and customer satisfaction.
Best practices for collecting qualitative data through surveys and interviews
Collecting qualitative data through surveys and interviews is a valuable source of insights for product case studies. Best practices for collecting qualitative data include creating a structured interview process or survey, identifying the right questions, avoiding leading questions, listening actively, encouraging detailed responses, and using open-ended questions. Additionally, businesses should ensure confidentiality and anonymity to encourage honest and objective feedback from customers and experts.
Top mistakes businesses make when creating product case studies
Creating effective and compelling product case studies can be challenging, and businesses can make common mistakes that can hinder their impact. Common mistakes include not targeting the right audience, failing to have a clear message or value proposition, making the case study too sales-oriented, and lacking concrete data and statistics. It's crucial to have a thorough understanding of the product, the customers, and their needs, and providing an objective evaluation of the results to avoid these pitfalls.
The role of branding in creating an effective product case study
Branding plays a crucial role in creating an effective and persuasive product case study. The case study should reflect the brand identity and voice, including logos, fonts, and colors. It should also align with the target audience's preferences and interests and embody the brand's values, mission, and vision. An effective product case study should differentiate the brand from competitors and communicate the unique selling proposition. Lastly, brand consistency should be maintained across all channels and formats used to promote the case study.
Browse hundreds of expert coaches
Leland coaches have helped thousands of people achieve their goals. A dedicated mentor can make all the difference.
Browse Related Articles

May 16, 2023
How to Prepare for Mozilla Product Management Case Interviews
Are you preparing for a Mozilla Product Management case interview? Look no further! Our comprehensive guide provides tips and strategies to help you ace your interview and land your dream job.

How to Prepare for Schmidt Futures Product Management Case Interviews
If you're looking to nail your Schmidt Futures product management case interviews, this article is a must-read.

How to Prepare for Square Product Management Case Interviews
Are you preparing for a Square product management case interview? Look no further! Our comprehensive guide provides tips and strategies to help you ace your interview and land your dream job at Square.

How to Prepare for TikTok Product Management Case Interviews
If you're looking to land a product management role at TikTok, then you need to be prepared for the case interview process.

How to Prepare for Walmart Product Management Case Interviews
If you're looking to land a product management role at Walmart, you'll likely need to ace the case interview.

How to Prepare for Zynga Product Management Case Interviews
If you're preparing for a Zynga product management case interview, this article is a must-read.

May 9, 2024
The Best Newsletters & Podcasts for Product Management
Stay ahead in product management with our expert-curated list of the best newsletters and podcasts. Gain insights, trends, and tips from industry leaders to level up your skills and career.

Best Companies for Product Managers: Top Employers for PMs in 2023
Discover the top companies that offer the best opportunities for product managers in 2023.
How to Prepare for Miro Product Management Case Interviews
If you're preparing for a Miro product management case interview, this article is a must-read.

How to Prepare for Oracle Product Management Case Interviews
Looking to ace your Oracle Product Management case interviews? Our comprehensive guide provides you with expert tips and strategies to help you prepare effectively.

How to Prepare for Shopify Product Management Case Interviews
If you're preparing for a Shopify product management case interview, this article is a must-read.

How to Prepare for Spotify Product Management Case Interviews
Are you looking to land a product management role at Spotify? Our article on how to prepare for Spotify product management case interviews is a must-read.
- Design for Business
- Most Recent
- Presentations
- Infographics
- Data Visualizations
- Forms and Surveys
- Video & Animation
- Case Studies
- Digital Marketing
- Design Inspiration
- Visual Thinking
- Product Updates
- Visme Webinars
- Artificial Intelligence
15 Real-Life Case Study Examples & Best Practices

Written by: Oghale Olori

Case studies are more than just success stories.
They are powerful tools that demonstrate the practical value of your product or service. Case studies help attract attention to your products, build trust with potential customers and ultimately drive sales.
It’s no wonder that 73% of successful content marketers utilize case studies as part of their content strategy. Plus, buyers spend 54% of their time reviewing case studies before they make a buying decision.
To ensure you’re making the most of your case studies, we’ve put together 15 real-life case study examples to inspire you. These examples span a variety of industries and formats. We’ve also included best practices, design tips and templates to inspire you.
Let’s dive in!
Table of Contents
What is a case study, 15 real-life case study examples, sales case study examples, saas case study examples, product case study examples, marketing case study examples, business case study examples, case study faqs.
- A case study is a compelling narrative that showcases how your product or service has positively impacted a real business or individual.
- Case studies delve into your customer's challenges, how your solution addressed them and the quantifiable results they achieved.
- Your case study should have an attention-grabbing headline, great visuals and a relevant call to action. Other key elements include an introduction, problems and result section.
- Visme provides easy-to-use tools, professionally designed templates and features for creating attractive and engaging case studies.
A case study is a real-life scenario where your company helped a person or business solve their unique challenges. It provides a detailed analysis of the positive outcomes achieved as a result of implementing your solution.
Case studies are an effective way to showcase the value of your product or service to potential customers without overt selling. By sharing how your company transformed a business, you can attract customers seeking similar solutions and results.
Case studies are not only about your company's capabilities; they are primarily about the benefits customers and clients have experienced from using your product.
Every great case study is made up of key elements. They are;
- Attention-grabbing headline: Write a compelling headline that grabs attention and tells your reader what the case study is about. For example, "How a CRM System Helped a B2B Company Increase Revenue by 225%.
- Introduction/Executive Summary: Include a brief overview of your case study, including your customer’s problem, the solution they implemented and the results they achieved.
- Problem/Challenge: Case studies with solutions offer a powerful way to connect with potential customers. In this section, explain how your product or service specifically addressed your customer's challenges.
- Solution: Explain how your product or service specifically addressed your customer's challenges.
- Results/Achievements : Give a detailed account of the positive impact of your product. Quantify the benefits achieved using metrics such as increased sales, improved efficiency, reduced costs or enhanced customer satisfaction.
- Graphics/Visuals: Include professional designs, high-quality photos and videos to make your case study more engaging and visually appealing.
- Quotes/Testimonials: Incorporate written or video quotes from your clients to boost your credibility.
- Relevant CTA: Insert a call to action (CTA) that encourages the reader to take action. For example, visiting your website or contacting you for more information. Your CTA can be a link to a landing page, a contact form or your social media handle and should be related to the product or service you highlighted in your case study.

Now that you understand what a case study is, let’s look at real-life case study examples. Among these, you'll find some simple case study examples that break down complex ideas into easily understandable solutions.
In this section, we’ll explore SaaS, marketing, sales, product and business case study examples with solutions. Take note of how these companies structured their case studies and included the key elements.
We’ve also included professionally designed case study templates to inspire you.
1. Georgia Tech Athletics Increase Season Ticket Sales by 80%

Georgia Tech Athletics, with its 8,000 football season ticket holders, sought for a way to increase efficiency and customer engagement.
Their initial sales process involved making multiple outbound phone calls per day with no real targeting or guidelines. Georgia Tech believed that targeting communications will enable them to reach more people in real time.
Salesloft improved Georgia Tech’s sales process with an inbound structure. This enabled sales reps to connect with their customers on a more targeted level. The use of dynamic fields and filters when importing lists ensured prospects received the right information, while communication with existing fans became faster with automation.
As a result, Georgia Tech Athletics recorded an 80% increase in season ticket sales as relationships with season ticket holders significantly improved. Employee engagement increased as employees became more energized to connect and communicate with fans.
Why Does This Case Study Work?
In this case study example , Salesloft utilized the key elements of a good case study. Their introduction gave an overview of their customers' challenges and the results they enjoyed after using them. After which they categorized the case study into three main sections: challenge, solution and result.
Salesloft utilized a case study video to increase engagement and invoke human connection.
Incorporating videos in your case study has a lot of benefits. Wyzol’s 2023 state of video marketing report showed a direct correlation between videos and an 87% increase in sales.
The beautiful thing is that creating videos for your case study doesn’t have to be daunting.
With an easy-to-use platform like Visme, you can create top-notch testimonial videos that will connect with your audience. Within the Visme editor, you can access over 1 million stock photos , video templates, animated graphics and more. These tools and resources will significantly improve the design and engagement of your case study.
Simplify content creation and brand management for your team
- Collaborate on designs , mockups and wireframes with your non-design colleagues
- Lock down your branding to maintain brand consistency throughout your designs
- Why start from scratch? Save time with 1000s of professional branded templates
Sign up. It’s free.

2. WeightWatchers Completely Revamped their Enterprise Sales Process with HubSpot

WeightWatchers, a 60-year-old wellness company, sought a CRM solution that increased the efficiency of their sales process. With their previous system, Weightwatchers had limited automation. They would copy-paste message templates from word documents or recreate one email for a batch of customers.
This required a huge effort from sales reps, account managers and leadership, as they were unable to track leads or pull customized reports for planning and growth.
WeightWatchers transformed their B2B sales strategy by leveraging HubSpot's robust marketing and sales workflows. They utilized HubSpot’s deal pipeline and automation features to streamline lead qualification. And the customized dashboard gave leadership valuable insights.
As a result, WeightWatchers generated seven figures in annual contract value and boosted recurring revenue. Hubspot’s impact resulted in 100% adoption across all sales, marketing, client success and operations teams.
Hubspot structured its case study into separate sections, demonstrating the specific benefits of their products to various aspects of the customer's business. Additionally, they integrated direct customer quotes in each section to boost credibility, resulting in a more compelling case study.
Getting insight from your customer about their challenges is one thing. But writing about their process and achievements in a concise and relatable way is another. If you find yourself constantly experiencing writer’s block, Visme’s AI writer is perfect for you.
Visme created this AI text generator tool to take your ideas and transform them into a great draft. So whether you need help writing your first draft or editing your final case study, Visme is ready for you.
3. Immi’s Ram Fam Helps to Drive Over $200k in Sales

Immi embarked on a mission to recreate healthier ramen recipes that were nutritious and delicious. After 2 years of tireless trials, Immi finally found the perfect ramen recipe. However, they envisioned a community of passionate ramen enthusiasts to fuel their business growth.
This vision propelled them to partner with Shopify Collabs. Shopify Collabs successfully cultivated and managed Immi’s Ramen community of ambassadors and creators.
As a result of their partnership, Immi’s community grew to more than 400 dedicated members, generating over $200,000 in total affiliate sales.
The power of data-driven headlines cannot be overemphasized. Chili Piper strategically incorporates quantifiable results in their headlines. This instantly sparks curiosity and interest in readers.
While not every customer success story may boast headline-grabbing figures, quantifying achievements in percentages is still effective. For example, you can highlight a 50% revenue increase with the implementation of your product.
Take a look at the beautiful case study template below. Just like in the example above, the figures in the headline instantly grab attention and entice your reader to click through.
Having a case study document is a key factor in boosting engagement. This makes it easy to promote your case study in multiple ways. With Visme, you can easily publish, download and share your case study with your customers in a variety of formats, including PDF, PPTX, JPG and more!

4. How WOW! is Saving Nearly 79% in Time and Cost With Visme
This case study discusses how Visme helped WOW! save time and money by providing user-friendly tools to create interactive and quality training materials for their employees. Find out what your team can do with Visme. Request a Demo
WOW!'s learning and development team creates high-quality training materials for new and existing employees. Previous tools and platforms they used had plain templates, little to no interactivity features, and limited flexibility—that is, until they discovered Visme.
Now, the learning and development team at WOW! use Visme to create engaging infographics, training videos, slide decks and other training materials.
This has directly reduced the company's turnover rate, saving them money spent on recruiting and training new employees. It has also saved them a significant amount of time, which they can now allocate to other important tasks.
Visme's customer testimonials spark an emotional connection with the reader, leaving a profound impact. Upon reading this case study, prospective customers will be blown away by the remarkable efficiency achieved by Visme's clients after switching from PowerPoint.
Visme’s interactivity feature was a game changer for WOW! and one of the primary reasons they chose Visme.
“Previously we were using PowerPoint, which is fine, but the interactivity you can get with Visme is so much more robust that we’ve all steered away from PowerPoint.” - Kendra, L&D team, Wow!
Visme’s interactive feature allowed them to animate their infographics, include clickable links on their PowerPoint designs and even embed polls and quizzes their employees could interact with.
By embedding the slide decks, infographics and other training materials WOW! created with Visme, potential customers get a taste of what they can create with the tool. This is much more effective than describing the features of Visme because it allows potential customers to see the tool in action.
To top it all off, this case study utilized relevant data and figures. For example, one part of the case study said, “In Visme, where Kendra’s team has access to hundreds of templates, a brand kit, and millions of design assets at their disposal, their team can create presentations in 80% less time.”
Who wouldn't want that?
Including relevant figures and graphics in your case study is a sure way to convince your potential customers why you’re a great fit for their brand. The case study template below is a great example of integrating relevant figures and data.
This colorful template begins with a captivating headline. But that is not the best part; this template extensively showcases the results their customer had using relevant figures.
The arrangement of the results makes it fun and attractive. Instead of just putting figures in a plain table, you can find interesting shapes in your Visme editor to take your case study to the next level.
5. Lyte Reduces Customer Churn To Just 3% With Hubspot CRM

While Lyte was redefining the ticketing industry, it had no definite CRM system . Lyte utilized 12–15 different SaaS solutions across various departments, which led to a lack of alignment between teams, duplication of work and overlapping tasks.
Customer data was spread across these platforms, making it difficult to effectively track their customer journey. As a result, their churn rate increased along with customer dissatisfaction.
Through Fuelius , Lyte founded and implemented Hubspot CRM. Lyte's productivity skyrocketed after incorporating Hubspot's all-in-one CRM tool. With improved efficiency, better teamwork and stronger client relationships, sales figures soared.
The case study title page and executive summary act as compelling entry points for both existing and potential customers. This overview provides a clear understanding of the case study and also strategically incorporates key details like the client's industry, location and relevant background information.
Having a good summary of your case study can prompt your readers to engage further. You can achieve this with a simple but effective case study one-pager that highlights your customer’s problems, process and achievements, just like this case study did in the beginning.
Moreover, you can easily distribute your case study one-pager and use it as a lead magnet to draw prospective customers to your company.
Take a look at this case study one-pager template below.

This template includes key aspects of your case study, such as the introduction, key findings, conclusion and more, without overcrowding the page. The use of multiple shades of blue gives it a clean and dynamic layout.
Our favorite part of this template is where the age group is visualized.
With Visme’s data visualization tool , you can present your data in tables, graphs, progress bars, maps and so much more. All you need to do is choose your preferred data visualization widget, input or import your data and click enter!
6. How Workato Converts 75% of Their Qualified Leads

Workato wanted to improve their inbound leads and increase their conversion rate, which ranged from 40-55%.
At first, Workato searched for a simple scheduling tool. They soon discovered that they needed a tool that provided advanced routing capabilities based on zip code and other criteria. Luckily, they found and implemented Chili Piper.
As a result of implementing Chili Piper, Workato achieved a remarkable 75–80% conversion rate and improved show rates. This led to a substantial revenue boost, with a 10-15% increase in revenue attributed to Chili Piper's impact on lead conversion.
This case study example utilizes the power of video testimonials to drive the impact of their product.
Chili Piper incorporates screenshots and clips of their tool in use. This is a great strategy because it helps your viewers become familiar with how your product works, making onboarding new customers much easier.
In this case study example, we see the importance of efficient Workflow Management Systems (WMS). Without a WMS, you manually assign tasks to your team members and engage in multiple emails for regular updates on progress.
However, when crafting and designing your case study, you should prioritize having a good WMS.
Visme has an outstanding Workflow Management System feature that keeps you on top of all your projects and designs. This feature makes it much easier to assign roles, ensure accuracy across documents, and track progress and deadlines.
Visme’s WMS feature allows you to limit access to your entire document by assigning specific slides or pages to individual members of your team. At the end of the day, your team members are not overwhelmed or distracted by the whole document but can focus on their tasks.
7. Rush Order Helps Vogmask Scale-Up During a Pandemic

Vomask's reliance on third-party fulfillment companies became a challenge as demand for their masks grew. Seeking a reliable fulfillment partner, they found Rush Order and entrusted them with their entire inventory.
Vomask's partnership with Rush Order proved to be a lifesaver during the COVID-19 pandemic. Rush Order's agility, efficiency and commitment to customer satisfaction helped Vogmask navigate the unprecedented demand and maintain its reputation for quality and service.
Rush Order’s comprehensive support enabled Vogmask to scale up its order processing by a staggering 900% while maintaining a remarkable customer satisfaction rate of 92%.
Rush Order chose one event where their impact mattered the most to their customer and shared that story.
While pandemics don't happen every day, you can look through your customer’s journey and highlight a specific time or scenario where your product or service saved their business.
The story of Vogmask and Rush Order is compelling, but it simply is not enough. The case study format and design attract readers' attention and make them want to know more. Rush Order uses consistent colors throughout the case study, starting with the logo, bold square blocks, pictures, and even headers.
Take a look at this product case study template below.
Just like our example, this case study template utilizes bold colors and large squares to attract and maintain the reader’s attention. It provides enough room for you to write about your customers' backgrounds/introductions, challenges, goals and results.
The right combination of shapes and colors adds a level of professionalism to this case study template.

8. AMR Hair & Beauty leverages B2B functionality to boost sales by 200%

With limits on website customization, slow page loading and multiple website crashes during peak events, it wasn't long before AMR Hair & Beauty began looking for a new e-commerce solution.
Their existing platform lacked effective search and filtering options, a seamless checkout process and the data analytics capabilities needed for informed decision-making. This led to a significant number of abandoned carts.
Upon switching to Shopify Plus, AMR immediately saw improvements in page loading speed and average session duration. They added better search and filtering options for their wholesale customers and customized their checkout process.
Due to this, AMR witnessed a 200% increase in sales and a 77% rise in B2B average order value. AMR Hair & Beauty is now poised for further expansion and growth.
This case study example showcases the power of a concise and impactful narrative.
To make their case analysis more effective, Shopify focused on the most relevant aspects of the customer's journey. While there may have been other challenges the customer faced, they only included those that directly related to their solutions.
Take a look at this case study template below. It is perfect if you want to create a concise but effective case study. Without including unnecessary details, you can outline the challenges, solutions and results your customers experienced from using your product.
Don’t forget to include a strong CTA within your case study. By incorporating a link, sidebar pop-up or an exit pop-up into your case study, you can prompt your readers and prospective clients to connect with you.

9. How a Marketing Agency Uses Visme to Create Engaging Content With Infographics

SmartBox Dental , a marketing agency specializing in dental practices, sought ways to make dental advice more interesting and easier to read. However, they lacked the design skills to do so effectively.
Visme's wide range of templates and features made it easy for the team to create high-quality content quickly and efficiently. SmartBox Dental enjoyed creating infographics in as little as 10-15 minutes, compared to one hour before Visme was implemented.
By leveraging Visme, SmartBox Dental successfully transformed dental content into a more enjoyable and informative experience for their clients' patients. Therefore enhancing its reputation as a marketing partner that goes the extra mile to deliver value to its clients.
Visme creatively incorporates testimonials In this case study example.
By showcasing infographics and designs created by their clients, they leverage the power of social proof in a visually compelling way. This way, potential customers gain immediate insight into the creative possibilities Visme offers as a design tool.
This example effectively showcases a product's versatility and impact, and we can learn a lot about writing a case study from it. Instead of focusing on one tool or feature per customer, Visme took a more comprehensive approach.
Within each section of their case study, Visme explained how a particular tool or feature played a key role in solving the customer's challenges.
For example, this case study highlighted Visme’s collaboration tool . With Visme’s tool, the SmartBox Dental content team fostered teamwork, accountability and effective supervision.
Visme also achieved a versatile case study by including relevant quotes to showcase each tool or feature. Take a look at some examples;
Visme’s collaboration tool: “We really like the collaboration tool. Being able to see what a co-worker is working on and borrow their ideas or collaborate on a project to make sure we get the best end result really helps us out.”
Visme’s library of stock photos and animated characters: “I really love the images and the look those give to an infographic. I also really like the animated little guys and the animated pictures. That’s added a lot of fun to our designs.”
Visme’s interactivity feature: “You can add URLs and phone number links directly into the infographic so they can just click and call or go to another page on the website and I really like adding those hyperlinks in.”
You can ask your customers to talk about the different products or features that helped them achieve their business success and draw quotes from each one.
10. Jasper Grows Blog Organic Sessions 810% and Blog-Attributed User Signups 400X
Jasper, an AI writing tool, lacked a scalable content strategy to drive organic traffic and user growth. They needed help creating content that converted visitors into users. Especially when a looming domain migration threatened organic traffic.
To address these challenges, Jasper partnered with Omniscient Digital. Their goal was to turn their content into a growth channel and drive organic growth. Omniscient Digital developed a full content strategy for Jasper AI, which included a content audit, competitive analysis, and keyword discovery.
Through their collaboration, Jasper’s organic blog sessions increased by 810%, despite the domain migration. They also witnessed a 400X increase in blog-attributed signups. And more importantly, the content program contributed to over $4 million in annual recurring revenue.
The combination of storytelling and video testimonials within the case study example makes this a real winner. But there’s a twist to it. Omniscient segmented the video testimonials and placed them in different sections of the case study.
Video marketing , especially in case studies, works wonders. Research shows us that 42% of people prefer video testimonials because they show real customers with real success stories. So if you haven't thought of it before, incorporate video testimonials into your case study.
Take a look at this stunning video testimonial template. With its simple design, you can input the picture, name and quote of your customer within your case study in a fun and engaging way.
Try it yourself! Customize this template with your customer’s testimonial and add it to your case study!

11. How Meliá Became One of the Most Influential Hotel Chains on Social Media

Meliá Hotels needed help managing their growing social media customer service needs. Despite having over 500 social accounts, they lacked a unified response protocol and detailed reporting. This largely hindered efficiency and brand consistency.
Meliá partnered with Hootsuite to build an in-house social customer care team. Implementing Hootsuite's tools enabled Meliá to decrease response times from 24 hours to 12.4 hours while also leveraging smart automation.
In addition to that, Meliá resolved over 133,000 conversations, booking 330 inquiries per week through Hootsuite Inbox. They significantly improved brand consistency, response time and customer satisfaction.
The need for a good case study design cannot be over-emphasized.
As soon as anyone lands on this case study example, they are mesmerized by a beautiful case study design. This alone raises the interest of readers and keeps them engaged till the end.
If you’re currently saying to yourself, “ I can write great case studies, but I don’t have the time or skill to turn it into a beautiful document.” Say no more.
Visme’s amazing AI document generator can take your text and transform it into a stunning and professional document in minutes! Not only do you save time, but you also get inspired by the design.
With Visme’s document generator, you can create PDFs, case study presentations , infographics and more!
Take a look at this case study template below. Just like our case study example, it captures readers' attention with its beautiful design. Its dynamic blend of colors and fonts helps to segment each element of the case study beautifully.

12. Tea’s Me Cafe: Tamika Catchings is Brewing Glory

Tamika's journey began when she purchased Tea's Me Cafe in 2017, saving it from closure. She recognized the potential of the cafe as a community hub and hosted regular events centered on social issues and youth empowerment.
One of Tamika’s business goals was to automate her business. She sought to streamline business processes across various aspects of her business. One of the ways she achieves this goal is through Constant Contact.
Constant Contact became an integral part of Tamika's marketing strategy. They provided an automated and centralized platform for managing email newsletters, event registrations, social media scheduling and more.
This allowed Tamika and her team to collaborate efficiently and focus on engaging with their audience. They effectively utilized features like WooCommerce integration, text-to-join and the survey builder to grow their email list, segment their audience and gather valuable feedback.
The case study example utilizes the power of storytelling to form a connection with readers. Constant Contact takes a humble approach in this case study. They spotlight their customers' efforts as the reason for their achievements and growth, establishing trust and credibility.
This case study is also visually appealing, filled with high-quality photos of their customer. While this is a great way to foster originality, it can prove challenging if your customer sends you blurry or low-quality photos.
If you find yourself in that dilemma, you can use Visme’s AI image edit tool to touch up your photos. With Visme’s AI tool, you can remove unwanted backgrounds, erase unwanted objects, unblur low-quality pictures and upscale any photo without losing the quality.
Constant Contact offers its readers various formats to engage with their case study. Including an audio podcast and PDF.
In its PDF version, Constant Contact utilized its brand colors to create a stunning case study design. With this, they increase brand awareness and, in turn, brand recognition with anyone who comes across their case study.
With Visme’s brand wizard tool , you can seamlessly incorporate your brand assets into any design or document you create. By inputting your URL, Visme’s AI integration will take note of your brand colors, brand fonts and more and create branded templates for you automatically.
You don't need to worry about spending hours customizing templates to fit your brand anymore. You can focus on writing amazing case studies that promote your company.
13. How Breakwater Kitchens Achieved a 7% Growth in Sales With Thryv

Breakwater Kitchens struggled with managing their business operations efficiently. They spent a lot of time on manual tasks, such as scheduling appointments and managing client communication. This made it difficult for them to grow their business and provide the best possible service to their customers.
David, the owner, discovered Thryv. With Thryv, Breakwater Kitchens was able to automate many of their manual tasks. Additionally, Thryv integrated social media management. This enabled Breakwater Kitchens to deliver a consistent brand message, captivate its audience and foster online growth.
As a result, Breakwater Kitchens achieved increased efficiency, reduced missed appointments and a 7% growth in sales.
This case study example uses a concise format and strong verbs, which make it easy for readers to absorb the information.
At the top of the case study, Thryv immediately builds trust by presenting their customer's complete profile, including their name, company details and website. This allows potential customers to verify the case study's legitimacy, making them more likely to believe in Thryv's services.
However, manually copying and pasting customer information across multiple pages of your case study can be time-consuming.
To save time and effort, you can utilize Visme's dynamic field feature . Dynamic fields automatically insert reusable information into your designs. So you don’t have to type it out multiple times.
14. Zoom’s Creative Team Saves Over 4,000 Hours With Brandfolder

Zoom experienced rapid growth with the advent of remote work and the rise of the COVID-19 pandemic. Such growth called for agility and resilience to scale through.
At the time, Zoom’s assets were disorganized which made retrieving brand information a burden. Zoom’s creative manager spent no less than 10 hours per week finding and retrieving brand assets for internal teams.
Zoom needed a more sustainable approach to organizing and retrieving brand information and came across Brandfolder. Brandfolder simplified and accelerated Zoom’s email localization and webpage development. It also enhanced the creation and storage of Zoom virtual backgrounds.
With Brandfolder, Zoom now saves 4,000+ hours every year. The company also centralized its assets in Brandfolder, which allowed 6,800+ employees and 20-30 vendors to quickly access them.
Brandfolder infused its case study with compelling data and backed it up with verifiable sources. This data-driven approach boosts credibility and increases the impact of their story.
Bradfolder's case study goes the extra mile by providing a downloadable PDF version, making it convenient for readers to access the information on their own time. Their dedication to crafting stunning visuals is evident in every aspect of the project.
From the vibrant colors to the seamless navigation, everything has been meticulously designed to leave a lasting impression on the viewer. And with clickable links that make exploring the content a breeze, the user experience is guaranteed to be nothing short of exceptional.
The thing is, your case study presentation won’t always sit on your website. There are instances where you may need to do a case study presentation for clients, partners or potential investors.
Visme has a rich library of templates you can tap into. But if you’re racing against the clock, Visme’s AI presentation maker is your best ally.

15. How Cents of Style Made $1.7M+ in Affiliate Sales with LeadDyno

Cents of Style had a successful affiliate and influencer marketing strategy. However, their existing affiliate marketing platform was not intuitive, customizable or transparent enough to meet the needs of their influencers.
Cents of Styles needed an easy-to-use affiliate marketing platform that gave them more freedom to customize their program and implement a multi-tier commission program.
After exploring their options, Cents of Style decided on LeadDyno.
LeadDyno provided more flexibility, allowing them to customize commission rates and implement their multi-tier commission structure, switching from monthly to weekly payouts.
Also, integrations with PayPal made payments smoother And features like newsletters and leaderboards added to the platform's success by keeping things transparent and engaging.
As a result, Cents of Style witnessed an impressive $1.7 million in revenue from affiliate sales with a substantial increase in web sales by 80%.
LeadDyno strategically placed a compelling CTA in the middle of their case study layout, maximizing its impact. At this point, readers are already invested in the customer's story and may be considering implementing similar strategies.
A well-placed CTA offers them a direct path to learn more and take action.
LeadDyno also utilized the power of quotes to strengthen their case study. They didn't just embed these quotes seamlessly into the text; instead, they emphasized each one with distinct blocks.
Are you looking for an easier and quicker solution to create a case study and other business documents? Try Visme's AI designer ! This powerful tool allows you to generate complete documents, such as case studies, reports, whitepapers and more, just by providing text prompts. Simply explain your requirements to the tool, and it will produce the document for you, complete with text, images, design assets and more.
Still have more questions about case studies? Let's look at some frequently asked questions.
How to Write a Case Study?
- Choose a compelling story: Not all case studies are created equal. Pick one that is relevant to your target audience and demonstrates the specific benefits of your product or service.
- Outline your case study: Create a case study outline and highlight how you will structure your case study to include the introduction, problem, solution and achievements of your customer.
- Choose a case study template: After you outline your case study, choose a case study template . Visme has stunning templates that can inspire your case study design.
- Craft a compelling headline: Include figures or percentages that draw attention to your case study.
- Work on the first draft: Your case study should be easy to read and understand. Use clear and concise language and avoid jargon.
- Include high-quality visual aids: Visuals can help to make your case study more engaging and easier to read. Consider adding high-quality photos, screenshots or videos.
- Include a relevant CTA: Tell prospective customers how to reach you for questions or sign-ups.
What Are the Stages of a Case Study?
The stages of a case study are;
- Planning & Preparation: Highlight your goals for writing the case study. Plan the case study format, length and audience you wish to target.
- Interview the Client: Reach out to the company you want to showcase and ask relevant questions about their journey and achievements.
- Revision & Editing: Review your case study and ask for feedback. Include relevant quotes and CTAs to your case study.
- Publication & Distribution: Publish and share your case study on your website, social media channels and email list!
- Marketing & Repurposing: Turn your case study into a podcast, PDF, case study presentation and more. Share these materials with your sales and marketing team.
What Are the Advantages and Disadvantages of a Case Study?
Advantages of a case study:
- Case studies showcase a specific solution and outcome for specific customer challenges.
- It attracts potential customers with similar challenges.
- It builds trust and credibility with potential customers.
- It provides an in-depth analysis of your company’s problem-solving process.
Disadvantages of a case study:
- Limited applicability. Case studies are tailored to specific cases and may not apply to other businesses.
- It relies heavily on customer cooperation and willingness to share information.
- It stands a risk of becoming outdated as industries and customer needs evolve.
What Are the Types of Case Studies?
There are 7 main types of case studies. They include;
- Illustrative case study.
- Instrumental case study.
- Intrinsic case study.
- Descriptive case study.
- Explanatory case study.
- Exploratory case study.
- Collective case study.
How Long Should a Case Study Be?
The ideal length of your case study is between 500 - 1500 words or 1-3 pages. Certain factors like your target audience, goal or the amount of detail you want to share may influence the length of your case study. This infographic has powerful tips for designing winning case studies
What Is the Difference Between a Case Study and an Example?
Case studies provide a detailed narrative of how your product or service was used to solve a problem. Examples are general illustrations and are not necessarily real-life scenarios.
Case studies are often used for marketing purposes, attracting potential customers and building trust. Examples, on the other hand, are primarily used to simplify or clarify complex concepts.
Where Can I Find Case Study Examples?
You can easily find many case study examples online and in industry publications. Many companies, including Visme, share case studies on their websites to showcase how their products or services have helped clients achieve success. You can also search online libraries and professional organizations for case studies related to your specific industry or field.
If you need professionally-designed, customizable case study templates to create your own, Visme's template library is one of the best places to look. These templates include all the essential sections of a case study and high-quality content to help you create case studies that position your business as an industry leader.
Get More Out Of Your Case Studies With Visme
Case studies are an essential tool for converting potential customers into paying customers. By following the tips in this article, you can create compelling case studies that will help you build trust, establish credibility and drive sales.
Visme can help you create stunning case studies and other relevant marketing materials. With our easy-to-use platform, interactive features and analytics tools , you can increase your content creation game in no time.
There is no limit to what you can achieve with Visme. Connect with Sales to discover how Visme can boost your business goals.
Easily create beautiful case studies and more with Visme

Trusted by leading brands
Recommended content for you:

Create Stunning Content!
Design visual brand experiences for your business whether you are a seasoned designer or a total novice.
About the Author
Software Engineering Institute
A Case Study in Successful Product Line Development
October 1, 1996 • technical report, by lisa brownsword and paul c. clements, cmu/sei report number, topic or tag.
This report describes the experience of a company that builds large, complex, embedded, real-time shipboard command-and-control systems as a product line.
Part of a Collection
Software Product Lines Collection
Cite This Technical Report
Brownsword, L., & Clements, P. (1996, October 1). A Case Study in Successful Product Line Development. (Technical Report CMU/SEI-96-TR-016). Retrieved September 17, 2024, from https://insights.sei.cmu.edu/library/a-case-study-in-successful-product-line-development/.
@techreport{brownsword_1996, author={Brownsword, Lisa and Clements, Paul}, title={A Case Study in Successful Product Line Development}, month={Oct}, year={1996}, number={CMU/SEI-96-TR-016}, howpublished={Carnegie Mellon University, Software Engineering Institute's Digital Library}, url={https://insights.sei.cmu.edu/library/a-case-study-in-successful-product-line-development/}, note={Accessed: 2024-Sep-17} }
Brownsword, Lisa, and Paul Clements. "A Case Study in Successful Product Line Development." (CMU/SEI-96-TR-016). Carnegie Mellon University, Software Engineering Institute's Digital Library . Software Engineering Institute, October 1, 1996. https://insights.sei.cmu.edu/library/a-case-study-in-successful-product-line-development/.
L. Brownsword, and P. Clements, "A Case Study in Successful Product Line Development," Carnegie Mellon University, Software Engineering Institute's Digital Library . Software Engineering Institute, Technical Report CMU/SEI-96-TR-016, 1-Oct-1996 [Online]. Available: https://insights.sei.cmu.edu/library/a-case-study-in-successful-product-line-development/. [Accessed: 17-Sep-2024].
Brownsword, Lisa, and Paul Clements. "A Case Study in Successful Product Line Development." (Technical Report CMU/SEI-96-TR-016). Carnegie Mellon University, Software Engineering Institute's Digital Library , Software Engineering Institute, 1 Oct. 1996. https://insights.sei.cmu.edu/library/a-case-study-in-successful-product-line-development/. Accessed 17 Sep. 2024.
Brownsword, Lisa; & Clements, Paul. A Case Study in Successful Product Line Development . CMU/SEI-96-TR-016. Software Engineering Institute. 1996. https://insights.sei.cmu.edu/library/a-case-study-in-successful-product-line-development/

Site Search
- How to Search
- Advisory Group
- Editorial Board
- OEC Fellows
- History and Funding
- Using OEC Materials
- Collections
- Research Ethics Resources
- Ethics Projects
- Communities of Practice
- Get Involved
- Submit Content
- Open Access Membership
- Become a Partner
- Advanced Search
- Webinar Series
Case Studies for Engineering Ethics Across the Product Life Cycle
This activity is considered an NAE Exemplar in Engineering Ethics Education and was included in a 2016 report with other exemplary activities. This activity uses reviewed case studies and life cycle assessment tools to help students develop needed ethical decision-making skills.
Exemplary features: Adaptability for use in secondary education; extensive collection of cases on the ethics of lifecycle impacts and sustainability
Why it’s exemplary: Real-world engineering decision making involves multiple actors and, for each, ethical considerations may arise at multiple levels—personal, professional, societal, or global. Our program of case studies and educational materials is exemplary in its interdisciplinary foundation, created collectively by engineers, policy experts, business professionals, and ethicists to provide clear examples for rising engineers to appreciate ethical issues from multiple angles. Accompanying materials are rigorously assessed in the classroom by internal and external evaluators based on national educational goals and guidelines, with versions developed to suit a variety of instructional modes. Full cases are designed for university engineering students, while streamlined versions for secondary schools spread an awareness of lifecycle issues and environmental ethics early in formal education. Widespread dissemination using various media adds to national infrastructure for ethics education in engineering and environmental fields, with the goal of emphasizing societal ethics and indirect effects.
Program description: A central goal of engineering education is to provide students with an understanding of context for their designs and decisions. A common theme currently relates to the environment and public health, specifically what constitutes a fair distribution of emissions or impacts, who or what has value, and what exactly gets counted in an engineering analysis of benefits and costs. These questions can be quite effectively discussed in the context of lifecycle engineering, a design strategy that uses a “cradle-to-grave” approach to evaluate environmental and social impacts, incorporating material, energy, and economic flows as well as social and biological effects at different stages. While the use of lifecycle engineering and lifecycle assessment (LCA) tools is widespread, the modeling structure and interpretation of results involve ethical and value judgments that must be navigated carefully by the analyst and by the receiver of the results.
LCA is increasingly important in corporate and government decision making, yet there is a dearth of materials specifically designed to integrate ethics education into life cycle–oriented coursework. Our ethics education project centers on the integration of life cycle–oriented case studies in design, engineering, management, and public policy fields. Case studies are effective pedagogical tools, and particularly useful in enabling students to develop practical understanding of the ethical challenges they will face as practicing professionals by placing them in mock decision-making roles. We have conducted a thorough review of nearly 1,000 existing case studies from engineering, business, and public policy to determine common topics and themes that relate to product life cycles and environmental and health impacts. Our case studies cover current events and engineering design decisions that involve balancing local or direct effects with larger, indirect effects on society, including (a) mismanagement of industrial waste and ecological impacts from industrial accidents, specifically the inundation of several villages in Hungary from a large-volume spill of red mud, a byproduct of aluminum production (production stage); (b) the upstream implication of material selection for consumer electronics, specifically the tradeoffs between Au-coated antennas and GaIn liquid metal reconfigurable antennas, a new technology being piloted by handset manufacturers (design stage); (c) implementation of state-level policy around compact fluorescent bulbs, balancing state targets for energy efficiency, indirect emissions as a result of reducing electricity demand, and direct potential emissions of Hg during lamp breakage, both accidental and intentional (use and disposal stages); and (d) whether federal/state agencies could and should require labelling of nanomaterials in consumer products, drawing parallels with labelling efforts for pharmaceuticals and food (use and disposal stages).
Following typical case study methods, students are presented with an engineering or design decision that they need to make, accompanied by background material that provides technical, environmental, and policy context. An accompanying teaching note guides instructors with ideas for classroom instruction, emphasizing the ethical concepts that are relevant to the case and written with proper terminology in collaboration with the Ethics Institute at Northeastern and assessed by an external evaluator. Instructional materials and video footage presenting each case, as well as shorter versions for younger audiences, are being created and will be hosted at the Ethics Institute as an additional teaching resource. The creation of the case studies involved a multidisciplinary collaboration among faculty members as well as graduate students. Undergraduate students and high school teachers are assisting in the creation of versions appropriate for secondary schools. These cases have been designed as one-week modules to be incorporated in existing courses and ethics workshops.
The educational goals of this project are to:
(1) Create engaging, practical, and effective case study and workshop materials that examine ethical dimensions of LCA practice and communication, for use in courses in engineering, management, and social science;
(2) Evaluate the effectiveness of these materials through robust educational assessment while improving student learning; and
(3) Engage other secondary school and college/university instructors through demonstration and provision of instructional guides and resources to accompany the case study and workshop materials.
The overall purpose of the project is to enable engineering students and the general public to have an understanding and meaningful discussions of indirect impacts of their activities, and how to balance direct benefits and indirect impacts. Our life cycle–oriented, case-based approach to engineering ethics education will fill gaps in case study resources by addressing fundamental ethical principles and macro-ethical issues on sustainability topics, developing novel, robustly assessed educational materials where few currently exist.
Assessment information: Our case studies and workshops are being piloted in engineering, business, and public policy classrooms. We have also been working with the Center for Advancing Teaching and Learning through Research at Northeastern and our external assessment advisor, Dr. Michael Loui, to develop assessment instruments and evaluation schemes that can be used across all of the cases. We now have a scheme that covers the common ethical concepts introduced in the cases—distributive justice, weighting/balancing risks, moral status, the precautionary principle, responsibility to report, and exploitation. The evaluation scheme is based on the framework presented by the Ethical Reasoning Value rubric published by the Association of American Colleges and Universities and will be applied to five separate classes of students over the coming year in order to test learning outcomes. This project grew out of the team’s experience with trying to fit existing engineering ethics cases into a life cycle–based framework. To provide a baseline for evaluating the new case studies, a review of learning assessments was carried out in spring 2015 for a mechanical/industrial engineering course, which currently uses a case study–based ethics module about the Bhopal chemical disaster, and retrospectively for the 150+ students who have passed through the course over the past several years. Review of assignments and responses informed the creation of case study teaching notes and the draft evaluation scheme. Continuing assessment will allow the project team to adjust the cases and teaching materials as necessary and add further instructional guidance where learning objectives are not being met.
Additional resources:
- Devising State Policy on Compact Fluorescent Lamps: https://us.sagepub.com/sites/default/files/devising-state-policy-on-compact-fluorescent-lamps-case.pdf
Related Resources
Submit Content to the OEC Donate

This material is based upon work supported by the National Science Foundation under Award No. 2055332. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.

- General & Introductory Industrial Engineering
- Industrial Engineering / Project Management

Project Management Case Studies, 6th Edition
ISBN: 978-1-119-82199-1
Digital Evaluation Copy

Harold Kerzner
The latest edition in the gold standard of project management case study collections
As a critical part of any successful, competitive business, project management sits at the intersection of several functional areas. And in the newly revised Sixth Edition of Project Management Case Studies , world-renowned project management professional Dr. Harold Kerzner delivers practical and in-depth coverage of project management in industries as varied as automotive, healthcare, government, manufacturing, communications, construction, chemical, aerospace, and more.
The latest edition of this bestselling book acts as the perfect supplement to any project management textbook or as an aid in the preparation for the PMP certification exam. The author includes new topics, like risk management, information sharing, scope changes, crisis dashboards, and innovation.
The Sixth Edition includes ten new case studies and a wide array of updates to existing cases to meet today’s industry standards and reflect the unique challenges facing modern project management professionals. This new edition:
- Features 10 new case studies from LEGO, NorthStar, Berlin Brandenburg Airport, and more
- Includes over 100 case studies drawn from real companies illustrating successful and poor implementation of project management
- Provides coverage of broad areas of project management as well as focused content on the automotive, healthcare, government, manufacturing, communications, construction, chemical, and aerospace industries
- Offers new topics including risk management, information sharing, scope changes, crisis dashboards, and innovation
Perfect for students taking courses on project management during their undergraduate degrees and at the graduate level as part of an MBA or graduate engineering program, Project Management Case Studies is also an indispensable resource for consulting and training companies who work with other professionals.
Harold Kerzner, PhD, is Senior Executive Director for Project, Program and Portfolio Management at the International Institute of Learning, Inc. (IIL). Dr. Kerzner’s impact on the project management industry inspired IIL to establish the Kerzner International Project Manager of the Year Award. IIL also donated $1M to the Project Management Institute Educational Foundation (PMIEF) to establish the Dr. Harold Kerzner Scholarship Fund.
- Pharmaceutical Engineering Magazine
- Online Exclusives
- Special Reports
- Facilities & Equipment
- Information Systems
- Product Development
- Supply Chain
- White Papers
- iSpeak Blog
- Editorial Calendar
- Article of the Year
- Submit an Article
- Editorial Team
Industry Perspectives on Practical Application of Platform Analytical Procedures

Pharmaceutical and biotechnology companies employ platform analytical procedures in the development stages of their synthetic and biological drug products and are beginning to leverage them for commercial products. This shift is supported by the acceptance of platform procedures in the recently adopted ICH Q2(R2) and ICH Q14. Six case studies are shared in this article to highlight how platform procedures are developed, applied to products in development, and assessed for extent of validation needed to determine if appropriate for use.
The recent revision to ICH Q2(R2) Validation of Analytical Procedures 1 and new guideline ICH Q14 Analytical Procedure Development 2 have provided a pathway for pharmaceutical and biotechnology companies to develop and use platform analytical procedures for commercial products.
ICH Q2(R2) defines a platform analytical procedure as “an analytical procedure that is suitable to test quality attributes of different products without significant change to its operational conditions, system suitability, and reporting structure. This type of analytical procedure can be used to analyze molecules that are sufficiently alike with respect to the attributes that the platform analytical procedure is intended to measure.” 1
Platform analytical procedures have been commonly used across industry to streamline clinical development by making analytical development and validation activities more efficient. The inclusion of the platform analytical procedure concept in ICH Q2(R2) formalizes the idea in guidance for the first time. ICH Q2(R2) states, “when an established platform analytical procedure is used for a new purpose, validation testing can be abbreviated, if scientifically justified.” 1
ICH Q14 further describes the rationale for application of platform analytical procedures and reduction of subsequent development activities: “In certain cases, an analytical procedure can be applied to multiple products with little or no modification of measurement conditions. For a new application of such platform analytical procedures, the subsequent development can be abbreviated, and certain validation tests can be omitted based on a science- and risk-based justification.” 2
The Food and Drug Omnibus Reform Act (FDORA)—a US Food and Drug administration (FDA) “rider” that is part of the Consolidated Appropriations Act, 2023—required the US FDA to establish a program for the designation of platform technologies. 3 It states that the sponsor of a drug application should demonstrate that the platform technology has the potential to be incorporated in, or utilized by, more than one drug without an adverse effect on quality, manufacturing, or safety. Although FDORA applies to manufacturing technologies, some concepts may also relate to platform analytical procedures, which this article aims to explore.
- 1 a b c International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. “ICH Harmonised Tripartite Guideline Q2(R2): Validation of Analytical Procedures.” Published November 2023. https://database.ich.org/sites/default/files/ICH_Q2%28R2%29_Guideline_2023_1130.pdf
- 2 a b International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. “ICH Harmonised Tripartite Guideline Q14: Analytical Procedure Development.” Published November 2023. https://database.ich.org/sites/default/files/ICH_Q14_Guideline_2023_1116.pdf
- 3 Consolidated Appropriations Act of 2023, H.R. 2617, 117th Cong. (2023).
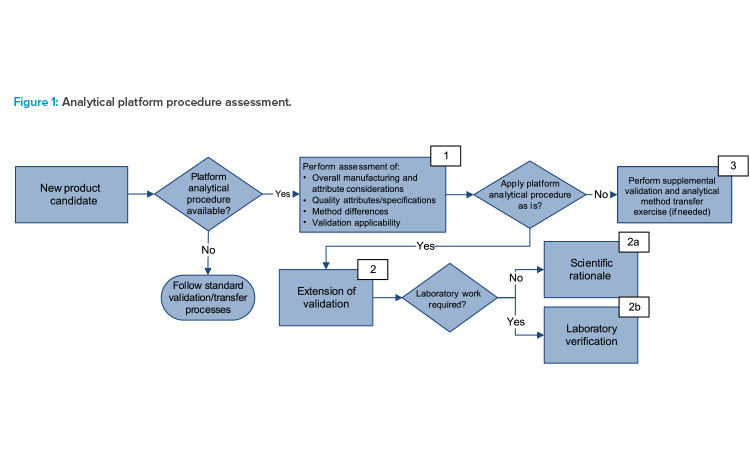
To date, industry has used the platform analytical procedure approach primarily for biological products and nearly exclusively in clinical development. 4 , 5 , 6 However, recent research applied a platform approach to the analysis of residual solvents in small molecules 7 and has offered suggestions for submitting information on analytical platform technologies in regulatory filings. 5 Monoclonal antibodies (mAbs) are good candidates for platform analytical procedures. This is because most attributes can be analyzed by an established set of conditions where the mAbs are of similar size and structure.
In a recent guideline from the World Health Organization (WHO), platform technologies were defined for methods and processes as they apply to mAbs. 8 Similarly, messenger RNA (mRNA) vaccines are good candidates to adopt platform analytical procedure strategies, as they may have common structural elements with only the codon-optimized sequence encoding the target antigen being unique to each new mRNA construct/variant. Moreover, many platform analytical procedures with established conditions, system suitability, and reporting structure have been used to analyze mRNA attributes for a commercial product. 9
The article showcases examples from six different companies to demonstrate the efficiencies and opportunities afforded by platform analytical procedures across techniques and modalities, leveraging prior knowledge and historical data. Although a pathway has been recognized through the adoption of ICH Q2(R2) and Q14, challenges lie ahead in preparing and gaining approval for global submissions.
By sharing these examples, the aim is to provide awareness of and create dialogue around global acceptance of platform analytical procedures. The objective of this article is to describe how the implementation of platform analytical procedures benefits industry and regulators alike.
Pfizer: Platform Analytical Procedures for mRNA Vaccines
The following case study shows the use of platform analytical procedures to support mRNA vaccine technology. All analytical procedures supporting re-lease and stability testing for an mRNA COVID-19 vaccine were fully validated in conformance with existing ICH guidelines. Platforms have been established through extensive method development, validation, transfer, and testing of mRNA vaccine products—and their effectiveness was demonstrated through an application to change an mRNA strain for the already approved product.
The approach outlined in this example can be used for any mRNA vaccine product or product change by following the appropriate assessment, described next.
The critical elements of a procedure and the rationale to support any reduced validation requirements are determined using a science- and risk-based approach. Figure 1 shows a decision tree used to apply the strategy with consistency and considers the following elements in box 1:
- Overall considerations of the manufacturing processes, matrices, and attributes
- Quality attributes and alignment of acceptance criteria (specification) to the analytical procedure’s intended use
- Changes to analytical methodology, reference materials, and critical reagents
- Applicability and coverage of the validation, e.g., validated range and transfer activities
The assessment described in box 1 of Figure 1 concluded that there is no change for the changed mRNA product other than the sequence in the platform manufacturing process, the target concentration, and the specification. The analytical procedures have already been established at the commercial testing sites. Next, one example of each outcome—scientific rationale (Figure 1, box 2a), laboratory verification (Figure 1, box 2b), and supplemental validation (Figure 1, box 3) and the rationale are given.
By sharing these examples, the aim is to provide awareness of and create dialogue around global acceptance of platform analytical procedures.
Extension of Validation: Scientific Rationale
For box 2a in Figure 1, RNA concentration by ultraviolet (UV) spectroscopy was selected as the example. The sequence change from mRNA-to-mRNA products does not alter the basic structural components—the purine and pyrimidine bases that absorb UV—of DNA or RNA. This allows for determination of RNA concentration by the Beer–Lambert law when measuring absorbance at 260 nanometers (nm).
The UV spectroscopy platform procedure was previously validated per ICH Q2 and transferred to the testing site. No changes to the platform analytical procedure are required to support testing of the new product. The procedure could be run without impacting system suitability criteria, operating conditions, and parameters.
Extension of Validation: Laboratory Verification
For box 2b in Figure 1, mRNA integrity (purity) using capillary gel electrophoresis (CGE) was selected as the example. Considering no significant change in size/valency, the sequence length of the new mRNA product/strain may affect migration time and the electrophoretic profile of fragments and intact species without impacting the ability of the method to accurately measure RNA integrity.
Verification was performed to confirm that the potentially impacted characteristics of the platform analytical procedure remain in a validated state. For this example, precision and specificity were challenged under a protocol with the same acceptance criteria. The CGE platform procedure was previously validated per ICH Q2 and transferred to the testing site.
No changes to the platform analytical procedures are required to support testing of the new product. The same assay control, sample preparations, and instrument conditions are used. The analytical procedure is applied without significant change to its operational conditions, system suitability, and reporting structure.
Supplemental Validation
For box 3 in Figure 1, identification of mRNA using digital droplet polymerase chain reaction (ddPCR) was selected as the example. The procedure requires mRNA product/strain-specific primers and probes (for specificity). Challenge validation characteristics (e.g., specificity) were considered under a protocol with acceptance criteria against both the formulation buffer and other mRNA constructs. The ddPCR platform procedure was previously validated per ICH Q2 and transferred to the intended testing site.
Platform Documentation for Regulatory Submissions
Although there are multiple ways to document this work within a company, the documentation within a regulatory submission should be considered early in the process. The full validation for each analytical procedure is a combination of the platform procedure validation, scientific rationale, and product-specific data.
For any extension of validation or supplemental validation leveraging historical knowledge and/or previous validations, a validation report or summary in the appropriate common technical document sections should be provided. This includes the scientific rationale justifying the strategy for each analytical procedure. In addition, product-specific results from laboratory verifications and supplemental validations should be shared within the submission.
The mRNA technology presents an application of the platform analytical procedure approach for an already approved product upon a variant change. The same principles can be applied to other products generated with a similar manufacturing process (for example, a new mRNA vaccine candidate). The subsequent case studies aim to expand the approach to other modalities.
Genentech: Capillary Electrophoresis Sodium Dodecyl Sulfate (CE-SDS) Methods
The platform analytical procedure workflow consists of three phases: development, validation, and utilization (see Figure 2). Measurement requirements for a given quality attribute or a set of quality attributes can be defined in an analytical target profile (ATP). This predefined approach to analytical development can ensure that the analytical procedure is robust across its life cycle.
To achieve the optimal procedural conditions, a design of experiments (DOE) for screening of critical method parameters is executed. During the second phase, a validation strategy is devised per ICH Q2. To ensure suitability of the platform analytical procedure for new molecules, an assessment along with a generic control is completed.
The platform analytical procedure control is used for system suitability testing, which ensures the proper performance of the analytical procedure during assessment. The platform procedure is monitored to maintain performance and prevent potential drifting through the system suitability criterion. Using a common control for system suitability across multiple labs, instruments, and analysts enables collecting more relevant data and improves the precision assessment of the platform analytical procedure. Furthermore, the control enables evaluation of any changes introduced by suppliers, such as column resins or equipment configurations.
- 4 Carnes, K. A., L. D. Oliver, Jr., T. A. Brown, R. F. Delgadillo, M. S. Ward, S. Mortazavi, et al. “A Platform Analytical Method for Intact Polysorbates in Protein-Containing Biopharmaceutical Products via HPLC-CAD.” Journal of Liquid Chromatography and Related Technologies 45, no. 17–20 (2023):259–70. doi:10.1080/10826076.2023.2207024
- 5 a b Krause, S. O. “Using Analytical Platform Technologies to Support Accelerated Product Development–Concept Review and Case Study.” PDA Journal of Pharmaceutical Science and Technology 76, no. 3 (2022):248–62. doi:10.5731/pdajpst.2021.012647
- 6 Ritter, N., S. J. Advant, J. Hennessey, H. Simmerman, J. McEntire, A. Mire-Sluis, et al. “What is Test Method Qualification? Proceedings of the WCBP CMC Strategy Forum, 24 July 2003.” BioProcess International. September 2004. www.casss.org/docs/default-source/cmc-strategy-forum-north-america/cmc-north-america-summary-papers/cmc-summary-paper-cmc-strategy-forum-north-america-summer-2003.pdf?sfvrsn=3b313f96_3
- 7 Bernardoni, F, H. H. Halsey, R. Hartman, T. Nowak, and E.K. Regalado. “Generic Gas Chromatography Flame Ionization Detection Method Using Hydrogen as the Carrier Gas for the Analysis of Solvents in Pharmaceuticals.” Journal of Pharmaceutical and Biomedical Analysis 165 (2019):366–73.
- 8 World Health Organization. “Guidelines on the Nonclinical and Clinical Evaluation of Monoclonal Antibodies and Related Products Intended for the Prevention or Treatment of Infectious Diseases.” Published March 2023. https://cdn.who.int/media/docs/default-source/biologicals/who-ecbs-mar-2023---annex-2---mab-evaluation-guidelines---clean-for-posting---1-may-2023.pdf
- 9 Ripley, D. “Platform Analytical Method Lifecycle: Trends, Strategies, and Case Study Through the Lens of mRNA Vaccines.” Presented at the CASSS WCPB Meeting, Washington, D.C., January 2023.
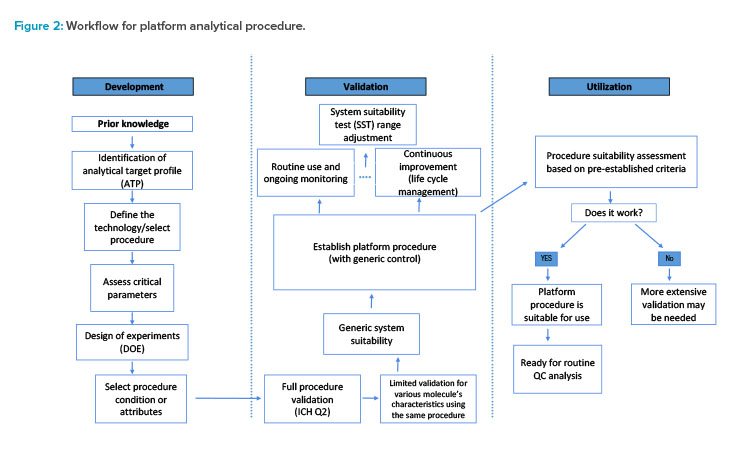
Application
CE-SDS analytical procedures are commonly used to evaluate the size heterogeneity, purity, and manufacturing consistency of biologics, under non-reduced and/or reduced conditions. The non-reduced CE-SDS method separates, detects, and quantitates distribution of product-related size variants (e.g., hinge fragments, main peak, or partially reduced cysteine-linked subunits). Reduced CE-SDS separates and quantitates light chain (LC), non-glycosylated heavy chain (HC), HC components, and other size variants such as non-specific polypeptide cleavages (e.g., LC and HC clips).
CE-SDS is a good candidate for a platform analytical procedure due to wide applicability and well-established robustness for usage in testing of mAbs. During the developmental phase, a combination of single- and multi-factorial studies are completed to identify critical method parameters, minimize the method variation, and increase applicability of usage as a platform across multiple mAb formats.
This approach was applied to the determination of critical dye-labeling conditions for a fluorescent-based platform CE-SDS method. 10 Critical parameters such as SDS-protein complexation, dye labeling, gel-lot variation, and auto-sampler stability are confirmed during validation.
The conditions for platform analytical procedures are established by leveraging prior learnings and knowledge from previous product-specific and platform-validation studies. During the utilization phase, the feasibility of the platform analytical procedure for new mAbs is evaluated through limited assessment studies, such as precision, specificity, stability indication, and comparison of UV and fluorescence electropherograms to ensure proper dye labeling.
Inclusion of a preestablished platform assay control during the assessment studies ensures proper performance of the analytical procedure. This approach creates a rigorous process to ensure that new molecules that meet the predefined criterion during the utilization phase can be tested by the platform CE-SDS method.
Eli Lilly and Company: Capillary Electrophoresis to Investigate Fragmentation of MABS
As previously mentioned, reduced and non-reduced CE-SDS are standard techniques employed in the purity assessment for large molecules. The following example offers another approach to establish a platform analytical procedure for CE-SDS analysis of size variants. The principle of the CE-SDS separation for both conditions—reducing (the addition of 2-mercaptoethanol [BME] to break disulfide linkages) and non-reducing—is based on the use of an electric field to separate proteins through a polymeric gel matrix based on their hydrodynamic radius.
The objective of this article is to describe how the implementation of platform analytical procedures benefits industry and regulators alike.
SDS is a negatively charged ionic detergent used in sample preparation to promote denaturation and achieve uniform protein coating. This is to obtain a uniform mass-to-charge ratio. Under such conditions, molecular hydrodynamic volume is the determining factor, resulting in a size-based separation. “Ignoring” the diversity in the complementarity-determining regions that make mAbs unique allows for common method conditions to be applied to mAbs of similar size and structure.
US Pharmacopoeia (USP) <1053> provides guidance and procedures used for characterization of biotechnology-derived articles by capillary electrophoresis. 11 That chapter is harmonized with the corresponding chapters in Japanese Pharmacopoeia (General Information 4) and European Pharmacopeia <2.2.47>. 12 , 13
Four platform analytical procedures are established (reducing and non-reducing conditions with pressure or electrokinetic injection) based on prior knowledge and experience with allowable ranges around the main parameters. When a new molecule enters the portfolio, experiments (e.g., exploratory DOEs) are conducted. This is to confirm that the two main factors found to be molecule-
dependent—heating time and temperature—fall within the established allowable ranges for the platform analytical procedures. The same is true for slight alterations of other method conditions that are covered by the underlying dataset used, e.g., minor adjustments in BME concentration.
Molecule-specific instructions for sample preparation that differ from the platform analytical procedures text are documented. The analysts are pointed to this documentation when executing the platform analytical procedure. If more significant modifications were needed to achieve the intended separation (e.g., changes to gel buffer detergent, reductant, wavelength, polarity, separation voltage, or instrument model), non-platform method conditions would have to be developed. These changes would result in a molecule-specific procedure.
Based on scientific evaluation (e.g., matrix components, analyte concentration, and selectivity need), a decision is made to use the pressure or electrokinetic injection platform analytical procedures. They are used throughout development for final drug substance/drug product, as well as in-process and characterization testing. The procedures are stability indicating, consistent with global regulatory guidance and industry expectations for mAbs.
During the clinical stages of development, method qualification for the platform analytical procedure is abbreviated. Linearity across the purity range, extrapolated linearity, accuracy, and matrix specificity are not executed. The only experiments performed are those to determine precision/intermediate precision, prepared sample stability, and stability indication using a stressed sample.
Molecule-specific procedure codes are then introduced at the time of marketing authorization. The validation exercise employs a mixed model of using historical data, generated across the platform, in addition to molecule-specific data. Robustness parameters are determined based on platform knowledge: the experiments conducted for each molecule.
Amgen: Leveraging a Platform-Size Exclusion Chromatography (SEC) Analytical Procedure for MAB Analysis
Purity methods, such as SEC, are ideal candidates for platform execution. SEC can be consistently applied across different mAbs due to its versatility and ability to handle diverse protein samples. SEC methods developed for one mAb can often be transferred and applied to others without any changes to the method.
This makes SEC a convenient and time-saving approach when analyzing different mAbs within a project or across multiple projects. SEC is compatible with a wide range of sample matrices, including different formulations, buffers, and excipients. It can handle mAbs derived from various sources (e.g., mammalian and microbial) or subclasses (immunoglobulin G (IgG)1 and IgG2). This versatility allows for the use of SEC as a consistent platform method across a mAb portfolio.
By selecting an appropriate column, laboratories can achieve optimal resolution and separation for different mAbs to generate a platform procedure, ensuring accurate and reliable analysis. SEC is extensively used in quality control laboratories for mAb analysis. Its ability to provide quantitative information about mAb size distribution, aggregate content, and purity makes it a reliable method for routine analysis of different mAb samples for both re-lease and stability purposes. By using SEC as a platform method, laboratories can streamline their analytical processes and maintain consistent performance.
Method Development
Typically, the development of analytical procedure commences after preclinical development for new active substances. Method development should align with the expectations of an analytical platform, which encompasses the ability to monitor relevant quality attributes, attribute sensitivity, and reporting intervals. The initiation of qualification and robustness experiments should occur after finalized method conditions and initial system suitability criteria are established.
The completion of initial robustness is a prerequisite before the method can be used for release and stability testing of clinical materials. As illustrated in Figure 3, if the method is applicable for platform use, the initial qualification and robustness lay the foundation for implementation. After the platform method is adopted in the laboratory, the method can be applied to a new molecule with minimal method qualification and robustness to demonstrate that the method is fit for purpose.
- 10 Michels, D. A., M. Parker, and O. Salas-Solano. “Quantitative Impurity Analysis of Monoclonal Antibody Size Heterogeneity By CE-LIF: Example of Development and Validation Through a Quality-By-Design Framework.” Electrophoresis 33, no 5. (2012):815–826. doi:10.1002/elps.201100528
- 11 US Pharmacopeial Convention. USP <1053>: Capillary Electrophoresis. Rockville, MD: US Pharmacopeial Convention, 2020.
- 12 The Japanese Pharmacopoeia. 17th Edition, April 2016. www.pmda.go.jp/files/000217650.pdf
- 13 European Pharmacopoeia. 11th Edition, July 2022. pheur.edqm.eu
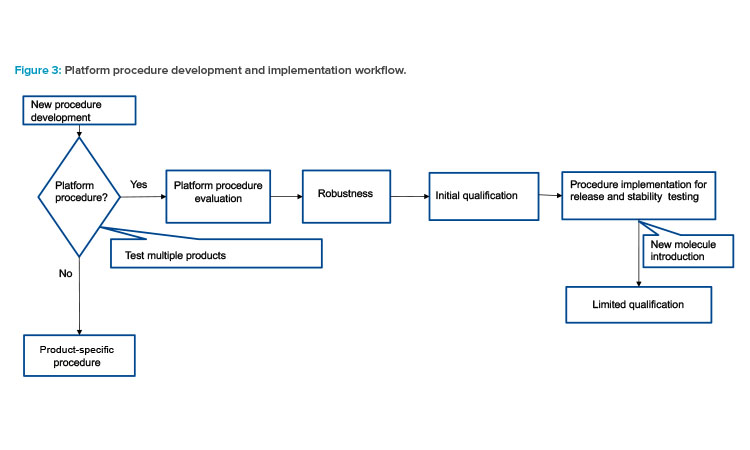
Having a subset of method qualification parameters tested prior to new product introduction is generally adequate in development. If prod-uct-specific information, such as example chromatograms and integration events, is needed to facilitate attribute testing of the new molecule, it can be achieved by maintaining specific supplemental documents. For regulatory submissions, platform and any product-specific qualification data can be provided to justify use of the platform procedure.
In summary, SEC’s compatibility, transferability, and suitability for diverse mAb samples make it an excellent candidate as a platform methodology. Its consistency and reliability contribute to efficient and robust analysis across different projects, as shown in Figure 4. It facilitates comparability studies and quality control and ensures the product quality of mAb-based therapies.
Sanofi: Peptide Mapping as a Global Platform Analytical Procedure
Peptide mapping has been developed and implemented as a platform procedure for identity testing of mAb and mAb-like molecules to support lot release at Sanofi. Despite being a comparative and platform procedure, peptide mapping is not a “generic” method. A specific map, or a fingerprint, needs to be developed for each protein under the same/similar operational conditions, system suitability, and reporting requirements. General guidance and procedures for this method are provided in USP <1055>: Peptide Mapping 14 and harmonized with corresponding chapters in the Japanese Pharmacopeia <G3-3-14> and European Pharmacopoeia <2.2.55>. 12 , 13
Platform Procedure
The platform procedure consists of disulfide reduction under denaturing conditions, cysteine alkylation, buffer exchange, enzymatic digestion, chromatographic separation of peptide fragments with UV detection, and data analysis. Identity is confirmed based on a comparison of the chromatographic profile and the relative peak area ratios of selected marker peptides of a test sample against a reference standard.
To ensure the platform procedure is robust and fit for its intended purpose, an enhanced approach—as delineated in ICH Q14—was used during development. Sample preparation (from reduction to digestion), separation and detection of peptide fragments, and data analysis are three major parts of the procedure. The potential variations that may occur in the first two parts are known to introduce artifacts or impact the method performance (e.g., chemical modifications, miscleavage, and poor resolution of peptides), and thus may lead to invalid results.
The risks associated with performance were assessed based on prior knowledge using a systematic approach (e.g., failure mode and effects analysis [FMEA] or Ishikawa diagram), and risk probability numbers were generated for the various method parameters. The method operable design region (MODR) was then defined based on the results from three representative mAbs (two IgG subtypes) using DOE and one-factor-at-a-time experiments. These MODRs are expected to be applicable to many other mAbs. Thus, the platform approach affords reduced method development for new mAbs. For molecules that behave differently and do not fit the platform, a product-specific method will be developed.
Specificity, as a key requirement for identity, was assessed based on the overall chromatographic profile and through a set of purposely selected marker peptides. The finalization of marker peptides was based on ultra performance liquid chromatography with ultraviolet detection and mass spectrometry (UPLC-UV-MS), e.g., quadrupole time-of-flight MS detector, data from the target molecule, and their chromatographic behaviors. The development of the peptide map generally requires the identification of most, if not all, peptide fragments by MS.
To prepare the platform procedure for release testing of new mAbs, an abbreviated qualification (e.g., only the specificity performance characteristic with a limited number of samples) is performed for early-stage programs. A validation is completed with additional parameters considered (e.g., inter-mediate precision, column lot-to-lot variations) to support late-stage programs. The robustness data from development may be included into the validation report as ICH Q14 states. The platform procedure has been implemented for multiple mAbs and mAb-like molecules across different UPLC instrument platforms and data analyses applications.
Merck & Co: Platform Gas Chromatography Procedure for Residual Solvent Quantification in Small-Molecule Pharmaceuticals
Organic solvents are commonly used in the production of small-molecule pharmaceuticals, primarily for drug substance and occasionally for formulated drug product. Ensuring that the levels of residual solvents are within acceptable limits, as defined by ICH Q3C (R8), 15 is crucial for both clinical and commercial release. Therefore, it is essential to analyze and control the organic solvents used in the manufacturing process.
Gas chromatography with flame ionization detection (GC-FID) is the preferred technique for quantifying residual solvent levels. This is due to its compatibility with solvents of different boiling points, availability of various stationary phases for different selectivity, separation capability of capillary columns, and the universality and sensitivity of FID.
Analyzing Residual Solvents
When analyzing residual solvents with GC-FID, two main factors to consider are sample preparation/introduction (direct injection or static headspace sampling) and chromatographic procedure. The chromatographic procedure typically employs single capillary columns and an oven temperature program for baseline resolution of the analytes.
USP <467>: Residual Solvents, 16 which is harmonized with European Pharmacopoeia Chapter 2.4.24, Identification and Control of Residual Solvents, 13 describes three headspace GC-FID procedures that employ capillary columns and static headspace injection. These procedures use hydrogen, nitrogen, or helium as the carrier gas for the determination of solvents. They have a chromatographic runtime of approximately 60 minutes and use capillary columns with different phases and coating thickness as well as temperature programs.
During development, the drug substance process undergoes frequent changes and optimization to enhance cost, yield, and process robustness. This necessitates the rapid analysis of samples produced under diverse conditions. It involves conducting solvent tests for isolated intermediates at different stages of synthesis and for the final drug substance. Having a platform GC-FID procedure simplifies analysis for residual solvents under these varying conditions, minimizing method development, accelerating key results, and reducing validation activities.
Developing a Platform GC-FID Procedure
A platform GC-FID procedure based on direct injection has been developed and validated using five different diluents to achieve this goal. 7 The procedure uses hydrogen as the carrier gas and can resolve over 30 commonly used solvents within a short runtime (eight minutes), which also makes it amenable to high-throughput analysis.
- 14 US Pharmacopeial Convention. USP <1055>: Peptide Mapping. Rockville, MD: US Pharmacopeial Convention, 2024.
- 12
- 15 International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. “ICH Harmonised Tripartite Guideline Q3C(R8): Impurities: Guideline for Residual Solvents.” Published April 2021. https://database.ich.org/sites/default/files/ICH_Q3C-R8_Guideline_Step4_2021_0422_1.pdf
- 16 US Pharmacopeial Convention. USP <467>: Residual Solvents. Rockville, MD: US Pharmacopeial Convention, 2022. European Pharmacopoeia. 11th Edition, July 2022.
- 7
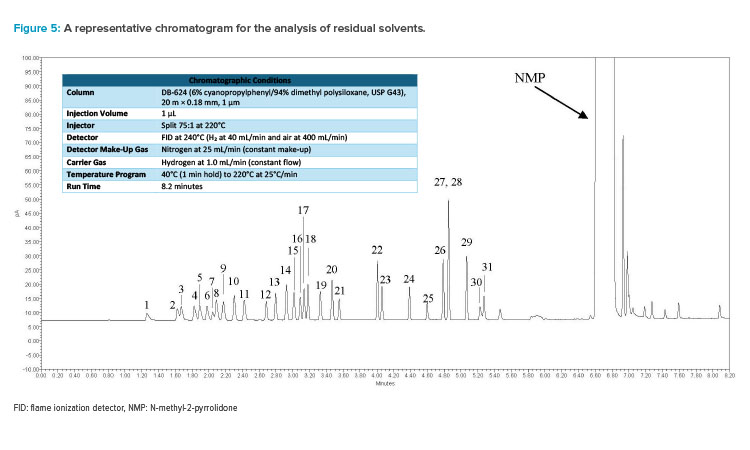
The procedure was established by conducting extensive development and validated using standards for sample independent attributes, such as line-arity, sensitivity (limit of detection/limit of quantitation [LOD/LOQ]), and precision. Various clinical-stage compounds, approved drug substances, and commercially available materials were used as model compounds to assess matrix effects, and the method’s accuracy was demonstrated through spike and recovery experiments for solvents likely to be present in the respective compound (see Figure 5 for a representative chromatogram and Table 1 for procedure details).
The platform GC-FID procedure is commonly used during development of small-molecule programs at Merck & Co. When considering a new program in early clinical development, the suitability of the platform procedure is evaluated based on its ability to separate the desired solvent(s) from process impurities and sample matrix interferences (specificity). Additionally, the stability of the sample solution is assessed to ensure an appropriate analysis window after preparation. If the peak resolution and solution stability results are acceptable, the method can be implemented without the need for further validation.
For commercial programs, supplemental validation of the platform procedure has been performed for application to drug substance. This includes accuracy assessment through spike/recovery, intermediate precision, and robustness evaluation using specific solvents of interest. Additional pro-gram-specific requirements are determined through a case-by-case assessment, and prior knowledge of the platform procedure is leveraged to support validation studies as needed.
Analyte Number | Analyte | Analyte Number | Analyte | Analyte Number | Analyte |
|---|---|---|---|---|---|
1 | Methanol | 11 | n-Propanol | 21 | 1,4-Dioxane |
2 | Ethanol | 12 | Ethyl Acetate | 22 | Toluene |
3 | Diethyl Ether | 13 | Tetrahydrofuran (THF) | 23 | i-Butyl Acetate |
4 | Acetone | 14 | Cyclohexane | 24 | n-Butyl Acetate |
5 | i-Propanol | 15 | i-Butanol | 25 | Dimethylformamide |
6 | Acetonitrile | 16 | Isopropyl acetate (IPAc) | 26 | Ethyl Benzene |
7 | Dichloromethane | 17 | 2-Methyltetraydrofuran | 27,28 | p-Xylene/m-Xylene |
8 | t-Butanol | 18 | n-Heptane | 29 | o-Xylene |
9 | Methyl tertiary-butyl ether | 19 | n-Butanol | 30 | Dimethyl sulfoxide |
10 | n-Hexane | 20 | Methylcyclohexane | 31 | Dimethylacetamide |
With accelerating clinical development, streamlining analytical activities without negatively impacting the assessment of product quality becomes essential.
In many cases, no further validation may be needed to apply this method to regulatory starting materials, intermediates, and in-process testing. Using a platform procedure for residual solvents has significantly accelerated process development and reduced validation requirements for introducing new small-molecule compounds to clinic as well as for commercialization.
Discussion and Example Comparison
Six companies shared examples that highlight how platform procedures are developed, applied to products across modalities in development, and assessed for extent of validation needed to demonstrate fitness. The two CE-SDS examples illustrate that multiple approaches are suitable to establish platform analytical procedures for the assessment of the same quality attributes.
Although one example was applied to commercial registration of mRNA analytical procedures, the remaining examples describe use of platform procedures for the clinical development of mAbs and small molecules. However, consistent with ICH Q2(R2), the principles described in these examples can be applied to commercial products beyond the modalities discussed previously.
Each example leveraged a science- and risk-based approach and followed similar approaches to those outlined in the presented flow charts. Extensive prior knowledge is employed in the development of the platform analytical procedure. This consists of knowledge of the modality as well as requirements of the measurement, analytical technique, and procedure parameters. Measurement requirements for the quality attribute(s) can be described in an ATP, which guides technology selection. An evaluation can be performed if the platform analytical procedure meets the performance requirements of the ATP.
ICH Q14 states, “prior product knowledge plays an important role in identifying the appropriate analytical technique. Knowledge of best practices and current state-of-the-art technologies as well as current regulatory expectations contributes to the selection of the most suitable technology for a given purpose. Existing platform analytical procedures (e.g., protein content determination by UV spectroscopy for a protein drug) can be leveraged to evaluate the attributes of a specific product without conducting additional procedure development.” 2 A risk analysis is completed to evaluate if the platform can be applied to a product with or without any modifications, then the extent of validation experiments required can be determined.
Benefits of Platform Analytical Procedures
There are many benefits to the application of platform analytical procedures, particularly in the commercial environment. With accelerating clinical development, streamlining analytical activities without negatively impacting the assessment of product quality becomes essential. Platform analytical procedures can enable rapid support of new products during development and subsequent commercialization. This also leads to an increased probability for enhanced robustness. They offer operational, compliance, and training advantages for analysts that do not need to learn unique procedure parameters for every test of every product.
Beyond a reduction in validation activities, transfer activities may be streamlined where testing is consolidated in the same commercial testing laboratory. Additionally, platform analytical procedures provide opportunities for automation that can increase throughput. These advantages may lead to greater reliability of the supply chain, providing value to companies, regulators, and patients. Using the same procedure parameters and validation results additionally leads to more rapid completion of registration documents and inspection readiness. This increases efficiency for health authorities during review and inspection where the procedure has previously been registered and implemented.
Once the product and procedure are registered, there are additional benefits throughout the product life cycle. Continuous monitoring of analytical procedures in the commercial environment is a resource-intensive, yet necessary, activity. Continually monitoring a platform across multiple products is more efficient and increases detectability of performance issues requiring mitigation. Management of this knowledge is critical due to the larger amount of data for one procedure and should lead to rapid and effective troubleshooting with any issues that may arise during routine use.
Although there are many benefits to the use of platform analytical procedures, there are challenges moving forward with this approach in commercial registrations. ICH Q2(R2) allows for validation testing to be abbreviated if scientifically justified. However, the extent of studies required to satisfy health authorities from non-ICH members may delay global acceptance of platform procedures and realization of their benefits. Applicants would also need to address documentation concerns, as the validation results would be provided in the dossier of another product. How that information is linked or copied for a new product would be of concern to the applicant.
The analytical procedure control strategy for the platform, including system suitability, and specific control and/or reference materials used to ensure acceptable performance should be considered. Also, the risk analysis for why a platform analytical procedure was applied to the new product and the extent of validation required would need to be communicated. Perhaps the most challenging aspect would be addressing the change management strategy.
The need to change parameters in a platform procedure for one product would require assessment for all other products that use the platform. These concerns may be reason for some applicants to reconsider or delay registering platform procedures. However, the six examples provide an excellent overview of the science- and risk-based approach that the authors believe are representative of the principles described in ICH Q2(R2) and Q14.
The recent adoption of ICH Q2(R2) and ICH Q14 supports the potential for registering platform analytical procedures. This publication describes six examples that outline varying yet scientifically sound approaches for developing, applying, and implementing platform procedures. Primarily, these examples have been successfully used in clinical development, but the principles can be applied to commercial registration.
Applying platform analytical procedures from one product to another has many benefits that facilitate efficient pathways to registration. Beyond registration, a more streamlined path for postapproval changes is important for the life cycle management of the product. With increasing pressures of cost, speed to market, and product quality, the use of analytical procedure platforms is one aspect of a science- and risk-based strategy for product commercialization that will ultimately benefit patients today.
Acknowledgments
The authors would like to acknowledge the ISPE Product Quality Lifecycle Implementation (PQLI)® Analytical Methods technical team for its contributions to the subject matter discussed in this paper, and David Michels, Anjana Patel, and Matt Sampson for their support with manuscript preparation and review of the publication.
Learn more about PQLI ®
Conflicts of Interest
All authors are employees of their respective companies. All authors contributed to the writing of the report, approved the final version of the manuscript, and agreed to submit the manuscript for publication.
- 2
Not a Member Yet?
To continue reading this article and to take advantage of full access to Pharmaceutical Engineering magazine articles, technical reports, white papers and exclusive content on the latest pharmaceutical engineering news, join ISPE today. In addition to exclusive access to all of the content in Pharmaceutical Engineering magazine, you will get online access to 24 ISPE Good Practice Guides, exclusive networking events, regulatory resources, Communities of Practice, and more.
Learn more about the valuable benefits you'll receive with an ISPE membership.
Join Today Member Login
Related Articles

In 2011, the US Food and Drug Administration (FDA) introduced the revised “Guidance for Industry: Process Validation: General Principles and Practices.” 1 The document incorporated principles from...
- 1 US Food and Drug Administration. “Guidance for Industry: Process Validation: General Principles and Practices.” January 2011. www.fda.gov/media/71021/download

The pharmaceutical industry faces considerable challenges throughout the development, manufacturing, and supply of medicines, largely due to the intricate and divergent global regulatory landscape. The adoption of structured data standards and utilization of cloud-based platforms offer immense potential to overcome these challenges by facilitating faster and more efficient global...

This article offers an overview of the benefits of Pharmaceutical Inspection Co-operation Scheme (PIC/S) membership for regulatory authorities and industry. It also highlights Latin American regulators’ current perspectives on PIC/S membership to increase awareness and encourage open dialogue about harmonization, recognition agreements, and potential increase for export facilitation, all which...
Information
- Author Services
Initiatives
You are accessing a machine-readable page. In order to be human-readable, please install an RSS reader.
All articles published by MDPI are made immediately available worldwide under an open access license. No special permission is required to reuse all or part of the article published by MDPI, including figures and tables. For articles published under an open access Creative Common CC BY license, any part of the article may be reused without permission provided that the original article is clearly cited. For more information, please refer to https://www.mdpi.com/openaccess .
Feature papers represent the most advanced research with significant potential for high impact in the field. A Feature Paper should be a substantial original Article that involves several techniques or approaches, provides an outlook for future research directions and describes possible research applications.
Feature papers are submitted upon individual invitation or recommendation by the scientific editors and must receive positive feedback from the reviewers.
Editor’s Choice articles are based on recommendations by the scientific editors of MDPI journals from around the world. Editors select a small number of articles recently published in the journal that they believe will be particularly interesting to readers, or important in the respective research area. The aim is to provide a snapshot of some of the most exciting work published in the various research areas of the journal.
Original Submission Date Received: .
- Active Journals
- Find a Journal
- Proceedings Series
- For Authors
- For Reviewers
- For Editors
- For Librarians
- For Publishers
- For Societies
- For Conference Organizers
- Open Access Policy
- Institutional Open Access Program
- Special Issues Guidelines
- Editorial Process
- Research and Publication Ethics
- Article Processing Charges
- Testimonials
- Preprints.org
- SciProfiles
- Encyclopedia

Article Menu

- Subscribe SciFeed
- Recommended Articles
- Google Scholar
- on Google Scholar
- Table of Contents
Find support for a specific problem in the support section of our website.
Please let us know what you think of our products and services.
Visit our dedicated information section to learn more about MDPI.
JSmol Viewer
Inertial motion capture-driven digital human for ergonomic validation: a case study of core drilling.

1. Introduction
2. digital human model generation, 2.1. establishment of three-dimensional virtual human model, 2.2. motion capture device-driven generation of a digital human, 3. evaluation of human comfortability, 3.1. calculation of joint angle in the human upper limb, 3.2. calculation of upper extremity joint moments, 4. experiment, 4.1. data acquisition, 4.2. calculation of upper extremity joint angles and torques, 4.3. assessment criteria, 4.4. analysis of visible and reachable domains, 5. discussion, 6. conclusions, author contributions, institutional review board statement, informed consent statement, data availability statement, conflicts of interest.
- Jennifer, L.; Hans, R. Visual ergonomics on-the-go. Work 2019 , 63 , 321–324. [ Google Scholar ] [ CrossRef ]
- Qiu, B.B.; Zhang, Y.F.; Shen, H.; Zhou, J.; Chu, L. Ergonomic researches in agricultural machinery—A systematic review using the PRISMA method. Int. J. Ind. Ergon. 2023 , 95 , 103446. [ Google Scholar ] [ CrossRef ]
- Fabio, G.; Margherita, P.; Luca, Z.; Marcello, P. An automatic procedure based on virtual ergonomic analysis to promote human-centric manufacturing. Procedia Manuf. 2019 , 38 , 488–496. [ Google Scholar ] [ CrossRef ]
- Manocha, M.; Soni, S.; Beck, M.; Ramirez, X. Scenario Based Training in Drilling Rig Simulator Improves Drillers Core Competency. In Proceedings of the SPE/IADC International Drilling Conference and Exhibition, Stavanger, Norway, 7–9 March 2023. [ Google Scholar ] [ CrossRef ]
- Bazazan, A.; Dianat, I.; Feizollahi, N.; Mombeini, Z.; Shirazi, A.M.; Castellucci, H.I. Effect of a posture correction-based intervention on musculoskeletal symptoms and fatigue among control room operators. Appl. Ergon. 2019 , 76 , 12–19. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Dai, M.Y.; Lu, Y.L.; Wang, S.; Zhang, H. Research progress on engineering mechanical ergonomic design based on virtual reality technology. J. Mach. Des. 2023 , 37 , 116–123. [ Google Scholar ] [ CrossRef ]
- Li, F.Y.; Li, Y.F. A Computer Aided Conceptual Evaluation of Excavator Cab Based on Ergonomics. Appl. Mech. Mater. 2012 , 121–126 , 1844–1848. [ Google Scholar ] [ CrossRef ]
- Chatterjee, T.; Bhattacharyya, D.; Majumdar, D.; Pal, M. Ergonomic Assessment of Multi Calibre Individual Weapon System in Virtual Reality Platform. Def. Ence J. 2019 , 69 , 240–248. [ Google Scholar ] [ CrossRef ]
- Rahal, R.; Matarese, G.; Gabiccini, M.; Artoni, A.; Prattichizzo, D.; Giordano, P.R.; Pacchierotti, C. Caring about the human operator: Haptic shared control for enhanced user comfort in robotic telemanipulation. IEEE Trans. Haptics 2020 , 13 , 197–203. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Nadeau, S.; Salmanzadeh, H.; Ahmadi, M.; Landau, K. Aviation deicing workers, global risk assessment of musculoskeletal injuries. Int. J. Ind. Ergon. 2019 , 71 , 8–13. [ Google Scholar ] [ CrossRef ]
- Ma, Q.G.; Sun, X.L.; Fu, H.J.; Zhao, D.C.; Guo, J.F. Manufacturing process design based on mental and physical workload analysis. Appl. Mech. Mater. 2013 , 345 , 482–485. [ Google Scholar ] [ CrossRef ]
- Ellegast, R.P.; Kraft, K.; Groenesteijn, L.; Krause, F.; Berger, H.; Vink, P. Comparison of four specific dynamic office chairs with a conventional office chair: Impact upon muscle activation, physical activity and posture. Appl. Ergon. 2012 , 43 , 296–307. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Caputo, F.; D’Amato, E.; Spada, S.; Sessa, F. Upper Body Motion Tracking System with Inertial Sensors for Ergonomic Issues in Industrial Environments. In Advances in Intelligent Systems and Computing ; Springer: Berlin/Heidelberg, Germany, 2016; Volume 489. [ Google Scholar ] [ CrossRef ]
- Gong, L.; Chen, B.; Xu, W.; Liu, C.; Li, X.; Zhao, Z.; Zhao, L. Motion Similarity Evaluation between Human and a Tri-Co Robot during Real-Time Imitation with a Trajectory Dynamic Time Warping Model. Sensors 2022 , 22 , 1968. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Dahibhate, G.; Shinde, R.; Sakle, N. The Use of Digital Human Modeling in Ergonomic Design and Product Development. J. Inst. Eng. India Ser. C 2023 , 104 , 1133–1138. [ Google Scholar ] [ CrossRef ]
- ISO 13485:2016 ; Medical Devices—Quality Management Systems—Requirements for Regulatory Purposes. International Organization for Standardization: Geneva, Switzerland, 2016.
- Sers, R.; Forrester, S.; Moss, E.; Ward, S.; Ma, J.; Zecca, M. Validity of the Perception Neuron inertial motion capture system for upper body motion analysis. Measurement 2020 , 149 , 107024. [ Google Scholar ] [ CrossRef ]
- ISO 11228-1:2003 ; Ergonomics—Manual Handling—Part 1: Lifting and Carrying. International Organization for Standardization: Geneva, Switzerland, 2003.
- ISO 9241-210:2019 ; Ergonomics of Human-System Interaction—Part 210: Human-Centered Design for Interactive Systems. International Organization for Standardization: Geneva, Switzerland, 2019.
- Li, S.; Hao, Y.; Mo, H.; Li, Q.; Lyu, Y.; Wang, X.; Li, H. Fast non-rigid human motion 3D reconstruction. J. Comput.-Aided Des. Comput. Graph. 2018 , 30 , 1505–1513. [ Google Scholar ] [ CrossRef ]
- Kavan, L.; Collins, S.; Zara, J.; O’Sullivan, C. Geometric Skinning with Approximate Dual Quaternion Blending. ACM Trans. Graph. 2008 , 27 , 105-1–105-23. [ Google Scholar ] [ CrossRef ]
- Tanaka, Y.; Nishikawa, K.; Yamada, N.; Tsuji, T. Analysis of Operational Comfort in Manual Tasks Using Human Force Manipulability Measure. IEEE Trans. Haptics 2015 , 8 , 8–19. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Gates, D.H.; Walters, L.S.; Cowley, J.; Wilken, J.M.; Resnik, L. Range of Motion Requirements for Upper-Limb Activities of Daily Living. Am. J. Occup. Ther. 2016 , 70 , 1–10. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- GB/T14779-1993 ; Sitting Posture and Human Body Template Design Requirements. China Standards Press: Beijing, China, 1993.
- Zhang, R.M.; Liu, Z.Q.; Zhou, Q.X.; Shen, Y.H.; Li, C.M. Comfortableness evaluation of range of motion of human upper limb joint. China Saf. Sci. J. 2018 , 28 , 75–80. [ Google Scholar ] [ CrossRef ]
- Qing, T.; Li, Z.; Kang, J. Evaluation of human upper limb motion comfort based on motion capture system. In Proceedings of the IEEE International Conference on Automation and Computing, Newcastle upon Tyne, UK, 6–7 September 2018. [ Google Scholar ] [ CrossRef ]
Click here to enlarge figure
| Anthropometry Parameters/Human of the 95th Percentile (cm) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Stature | Weight | Head Length | Acromion Height | Biacromial Breadth | Arm Length | Elbow Span | Buttock-Popliteal Length | Thigh Clearance |
| 177.5 | 75.0 | 19.8 | 147.1 | 39.7 | 79.9 | 135.0 | 49.0 | 16.2 |
| Position Data of Each Node at the Same Time | Random Sampling Node Number | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |||
| Operating rocker | Left Hand | x | −0.124 | −0.122 | −0.118 | −0.112 | −0.108 | −0.103 | −0.098 | −0.093 | −0.090 | −0.089 |
| y | 0.843 | 0.847 | 0.854 | 0.864 | 0.873 | 0.884 | 0.896 | 0.910 | 0.925 | 0.938 | ||
| z | 0.194 | 0.192 | 0.191 | 0.189 | 0.188 | 0.186 | 0.187 | 0.189 | 0.193 | 0.199 | ||
| Left Lower Arm | x | −0.126 | −0.123 | −0.119 | −0.114 | −0.110 | −0.105 | −0.100 | −0.095 | −0.092 | −0.091 | |
| y | 0.839 | 0.843 | 0.849 | 0.859 | 0.867 | 0.878 | 0.890 | 0.904 | 0.919 | 0.931 | ||
| z | 0.188 | 0.186 | 0.184 | 0.183 | 0.182 | 0.181 | 0.181 | 0.184 | 0.189 | 0.194 | ||
| Left Upper Arm | x | −0.292 | −0.292 | −0.292 | −0.293 | −0.294 | −0.295 | −0.297 | −0.299 | −0.301 | −0.303 | |
| y | 0.931 | 0.930 | 0.930 | 0.929 | 0.930 | 0.930 | 0.931 | 0.933 | 0.935 | 0.938 | ||
| z | 0.011 | 0.010 | 0.009 | 0.009 | 0.010 | 0.013 | 0.018 | 0.026 | 0.035 | 0.044 | ||
| Left Shoulder | x | −0.183 | −0.184 | −0.184 | −0.184 | −0.184 | −0.184 | −0.184 | −0.184 | −0.184 | −0.184 | |
| y | 1.175 | 1.174 | 1.173 | 1.173 | 1.173 | 1.172 | 1.172 | 1.172 | 1.172 | 1.172 | ||
| z | −0.001 | −0.002 | −0.002 | −0.003 | −0.004 | −0.004 | −0.005 | −0.005 | −0.005 | −0.005 | ||
| Twist knob | Left Hand | x | −0.092 | −0.092 | −0.091 | −0.091 | −0.091 | −0.092 | −0.092 | −0.093 | −0.093 | −0.093 |
| y | 0.836 | 0.836 | 0.837 | 0.837 | 0.838 | 0.836 | 0.839 | 0.839 | 0.839 | 0.840 | ||
| z | 0.034 | 0.034 | 0.038 | 0.039 | 0.041 | 0.042 | 0.043 | 0.044 | 0.045 | 0.045 | ||
| Left Lower Arm | x | −0.092 | −0.092 | −0.092 | −0.092 | −0.092 | −0.092 | −0.092 | −0.092 | −0.092 | −0.093 | |
| y | 0.831 | 0.831 | 0.831 | 0.832 | 0.832 | 0.833 | 0.833 | 0.833 | 0.834 | 0.834 | ||
| z | 0.028 | 0.028 | 0.032 | 0.033 | 0.035 | 0.036 | 0.037 | 0.038 | 0.039 | 0.040 | ||
| Left Upper Arm | x | −0.231 | −0.231 | −0.230 | −0.230 | −0.229 | −0.229 | −0.228 | −0.228 | −0.228 | −0.229 | |
| y | 0.929 | 0.929 | 0.928 | 0.927 | 0.927 | 0.927 | 0.927 | 0.927 | 0.926 | 0.926 | ||
| z | −0.166 | −0.166 | −0.164 | −0.163 | −0.163 | −0.162 | −0.162 | −0.161 | −0.161 | −0.160 | ||
| Left Shoulder | x | −0.136 | −0.136 | −0.136 | −0.135 | −0.135 | −0.135 | −0.135 | −0.134 | −0.134 | −0.134 | |
| y | 1.166 | 1.166 | 1.166 | 1.166 | 1.166 | 1.165 | 1.165 | 1.165 | 1.165 | 1.165 | ||
| z | −0.090 | −0.090 | −0.091 | −0.091 | −0.090 | −0.090 | −0.090 | −0.090 | −0.090 | −0.090 | ||
| Push button | Left Hand | x | −0.359 | −0.360 | −0.360 | −0.359 | −0.359 | −0.358 | −0.356 | −0.354 | −0.352 | −0.349 |
| y | 0.868 | 0.868 | 0.869 | 0.869 | 0.869 | 0.869 | 0.869 | 0.870 | 0.870 | 0.871 | ||
| z | 0.198 | 0.201 | 0.202 | 0.204 | 0.204 | 0.204 | 0.205 | 0.205 | 0.205 | 0.206 | ||
| Left Lower Arm | x | −0.357 | −0.357 | −0.358 | −0.357 | −0.356 | −0.355 | −0.353 | −0.351 | −0.349 | −0.347 | |
| y | 0.863 | 0.863 | 0.864 | 0.864 | 0.864 | 0.865 | 0.865 | 0.866 | 0.866 | 0.866 | ||
| z | 0.192 | 0.194 | 0.196 | 0.197 | 0.198 | 0.198 | 0.198 | 0.198 | 0.198 | 0.199 | ||
| Left Upper Arm | x | −0.296 | −0.297 | −0.297 | −0.298 | −0.298 | −0.298 | −0.299 | −0.299 | −0.299 | −0.301 | |
| y | 0.974 | 0.975 | 0.976 | 0.976 | 0.977 | 0.977 | 0.976 | 0.975 | 0.975 | 0.975 | ||
| z | 0.011 | 0.010 | 0.009 | 0.009 | 0.010 | 0.013 | 0.018 | 0.026 | 0.035 | 0.044 | ||
| Left Shoulder | x | −0.128 | −0.128 | −0.128 | −0.129 | −0.129 | −0.129 | −0.130 | −0.130 | −0.130 | −0.131 | |
| y | 1.171 | 1.171 | 1.172 | 1.173 | 1.173 | 1.172 | 1.172 | 1.172 | 1.172 | 1.172 | ||
| z | −0.097 | −0.097 | −0.097 | −0.096 | −0.096 | −0.095 | −0.095 | −0.096 | −0.094 | −0.094 | ||
| Upper Limb Movement | Joint Parameter | Experimental Data Statistics | ||
|---|---|---|---|---|
| Average Value | Minimum Value | Maximum Value | ||
| Operating rocker | Shoulder angle (°) | 66.04 | 48.01 | 98.91 |
| Elbow angle (°) | 86.32 | 67.98 | 113.62 | |
| Wrist angle (°) | 31.02 | 22.99 | 43.47 | |
| Shoulder torques (N·cm) | 253.39 | 0 | 857.79 | |
| Elbow torques (N·cm) | 119.66 | 0 | 483.62 | |
| Twist knob | Shoulder angle (°) | 95.46 | 65.79 | 130.06 |
| Elbow angle (°) | 108.65 | 82.81 | 133.29 | |
| Wrist angle (°) | 78.82 | 58.12 | 104.85 | |
| Shoulder torques (N·cm) | 296.78 | 0 | 1108.45 | |
| Elbow torques (N·cm) | 189.35 | 0 | 746.83 | |
| Push button | Shoulder angle (°) | 66.04 | 48.01 | 98.91 |
| Elbow angle (°) | 82.61 | 50.83 | 117.43 | |
| Wrist angle (°) | 36.30 | 14.32 | 63.18 | |
| Shoulder torques (N·cm) | 213.80 | 0 | 738.64 | |
| Elbow torques (N·cm) | 101.33 | 0 | 250.21 | |
| Joint | Mode of Motion | Limiting Angle | Comfort Zone |
|---|---|---|---|
| Shoulder joint | Front and rear pendulum | 140°~40° | 40°~90° |
| Elbow joint | Bend and stretch | 140°~40° | 80°~110° |
| Wrist joint | Wrist flexion and extension | 80°~70° | 10°~30° |
| Joint | Upper Limb Movement | ||
|---|---|---|---|
| Operating Rocker | Twist Knob | Push Button | |
| Shoulder joint | 0.782 | 0.758 | 0.800 |
| Elbow joint | 0.833 | 0.797 | 0.815 |
| Comfort Level | I | II | III | IV | V |
|---|---|---|---|---|---|
| Comfort index | |||||
| Comfort description | Very uncomfortable | Not comfortable | Generally comfortable | More comfortable | Very comfortable |
| The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
Share and Cite
Zhao, Q.; Lu, T.; Tao, M.; Cheng, S.; Wen, G. Inertial Motion Capture-Driven Digital Human for Ergonomic Validation: A Case Study of Core Drilling. Sensors 2024 , 24 , 5962. https://doi.org/10.3390/s24185962
Zhao Q, Lu T, Tao M, Cheng S, Wen G. Inertial Motion Capture-Driven Digital Human for Ergonomic Validation: A Case Study of Core Drilling. Sensors . 2024; 24(18):5962. https://doi.org/10.3390/s24185962
Zhao, Quan, Tao Lu, Menglun Tao, Siyi Cheng, and Guojun Wen. 2024. "Inertial Motion Capture-Driven Digital Human for Ergonomic Validation: A Case Study of Core Drilling" Sensors 24, no. 18: 5962. https://doi.org/10.3390/s24185962
Article Metrics
Article access statistics, further information, mdpi initiatives, follow mdpi.

Subscribe to receive issue release notifications and newsletters from MDPI journals
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Down to Business
- Published: 16 September 2024
Treating stress urinary incontinence by tissue engineering
- Deana Mohr-Haralampieva ORCID: orcid.org/0009-0006-0856-8137 1 , 2 ,
- Steve Kappenthuler 1 &
- Marcus Droege 1
Nature Reviews Bioengineering ( 2024 ) Cite this article
Metrics details
- Business and management
- Muscle stem cells
- Regeneration
Stress urinary incontinence (SUI), a frequently underdiagnosed condition that mainly affects women, lacks effective and long-term treatment options. MUVON Therapeutics has developed a tissue-engineered advanced therapy medicinal product for the treatment of SUI, based on autologous cells, which is being tested in a phase II clinical study — a challenging development effort.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
111,21 € per year
only 9,27 € per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Danforth, K. N. et al. Risk factors for urinary incontinence among middle-aged women. Am. J. Obstet. Gynecol. 194 , 339–345 (2006).
Article Google Scholar
Norton, P. & Brubaker, L. Urinary incontinence in women. Lancet 367 , 57–67 (2006).
Patel, U. J., Godecker, A. L., Giles, D. L. & Brown, H. W. Updated prevalence of urinary incontinence in women: 2015–2018 national population-based survey data. Female Pelvic Med. Reconstr. Surg. 28 , 181–187 (2022).
Schmid, F. A. et al. Transurethral injection of autologous muscle precursor cells for treatment of female stress urinary incontinence: a prospective phase I clinical trial. Int. Urogynecology J. 34 , 2197–2206 (2023).
US National Library of Medicine. ClinicalTrials.gov , https://clinicaltrials.gov/study/NCT03439527 (2021).
Iglesias-Lopez, C., Agustí, A., Vallano, A. & Obach, M. Current landscape of clinical development and approval of advanced therapies. Mol. Ther. Methods Clin. Dev. 23 , 606–618 (2021).
Download references
Acknowledgements
We thank all team members of MUVON Therapeutics for their expertise and personal achievements in each of the topics mentioned, while translating an academic process towards a commercial one. We also thank M. Graf for his input on the literature search and T. Haralampieva, N. Schwarz and J. Hegetschweiler for graphic design. We acknowledge funding and support from Horizon2020 for the phase 1 trial, and Wyss Zurich Translational Center and University of Zurich for the phase 2 study.
Author information
Authors and affiliations.
MUVON Therapeutics AG, Zürich, Switzerland
Deana Mohr-Haralampieva, Steve Kappenthuler & Marcus Droege
University of Zurich, Wyss Zurich Translational Center, Zurich, Switzerland
Deana Mohr-Haralampieva
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Deana Mohr-Haralampieva .
Ethics declarations
Competing interests.
D.M.-H., S.K. and M.D. are involved with the company MUVON Therapeutics AG. This relationship could potentially influence the interpretation and presentation of the research findings discussed in this manuscript.
Additional information
Related links.
Women are affected twice as often as men, with an estimated 40% of women above the age of 40 experiencing SUI: https://www.swanstudy.org/urinary-incontinence-problematic-for-many-women-over-40-study-finds/
World Federation of Incontinence and Pelvic Problems (WFIPP): https://wfipp.org/
Rights and permissions
Reprints and permissions
About this article
Cite this article.
Mohr-Haralampieva, D., Kappenthuler, S. & Droege, M. Treating stress urinary incontinence by tissue engineering. Nat Rev Bioeng (2024). https://doi.org/10.1038/s44222-024-00246-6
Download citation
Published : 16 September 2024
DOI : https://doi.org/10.1038/s44222-024-00246-6
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
Learning and Teaching
- Curriculum Design
- Embedding sustainability
Case study: Student focus groups in Chemical Engineering map path to enhanced climate and sustainability teaching in undergraduate curriculum.
The Department of Chemical Engineering at University of Bath has run focus groups with students to understand how staff can enhance climate and sustainability teaching provision in their courses. In May 2024, the Equality, Diversity, and Inclusion (ED&I) Committee Chair Dr Hannah Leese and departmental Climate Advocate Dr Sandhya Moise co-organised a consultation session with final-year Chemical Engineering students, one of whom also sits on the ED&I Committee. Participants were drawn from the MEng Chemical Engineering and MEng Environmental Engineering courses. The focus groups prioritised discussion of the links between sustainability and social justice in these disciplinary contexts.
Published on: 16/09/2024 · Last updated on: 16/09/2024
Questions of social justice are central to the relationship between engineering, climate, and sustainability.
The impetus for the consultation, which aligned climate change, sustainability, and ED&I efforts across the department, stemmed from the recognition that climate change is intricately linked with questions of social justice: climate change disproportionately affects vulnerable and marginalised communities, limiting their access to resources such as clean water, food, energy, and healthcare. As such, engineering solutions must be inclusive and avoid exacerbating inequalities. Future engineers must be equipped with the skills and tools to evaluate climate change solutions through a lens of equality, diversity, and inclusivity. How, then, can engineering curricula respond to this challenge?
Insights on curriculum design and delivery
The student focus groups identified key areas that will feed into future curriculum design in the department:
- Whilst Chemical Engineering curricula have successfully embedded knowledge and tools to assess various aspects of environmental sustainability, the discipline and its curricula would benefit from increased focus on people and social justice.
- Adopting principles of course-wide design, the links between environmental sustainability and social justice should be signposted at all levels of the curriculum.
- Students are especially excited by novel and innovative areas of study (renewable energies, hydrogen), and less motivated by incremental and well-established fields (e.g. water treatment).
- Students value explicit marking criteria around sustainability and inclusive design in engineering.
Prioritising student voice and co-creation in climate and sustainability education initiatives
Student feedback on the session was very positive: “I found the EDI-Sustainability focus group to be valuable, as it provided a safe space to voice our views and experiences, and to reassure us that diverse student perceptions and viewpoints are heard and valued. My main takeaways were a deeper understanding of existing EDI and sustainability initiatives in the department, and how these aspects are currently recognised and embedded into the Chemical Engineering curriculum. I appreciated the opportunity to give constructive feedback on how this can be built upon, with the understanding this can be used for continuous improvement for future cohorts.”
Next steps: consolidating climate and sustainability provision and ED&I perspectives across Chemical Engineering courses
Following the focus groups, the Department is introducing further mechanisms to ensure that students are at the heart of its initiative to align climate and sustainability education with ED&I and social justice perspectives. The Department of Chemical Engineering will continue working alongside the Chemical Engineering Student Group (CESG) membership to enable students to drive the sustainability agenda forward within the department. Dr Moise runs a ‘Climate change and Chemical Engineering’ talk in Freshers Week to foreground this aspect of the curriculum and to signpost the CESG. The CESG President is also regularly invited to appropriate lectures across year groups to remind students of opportunities for engagement. In parallel, the ED&I Committee Chair attends all Head of Department Welcome Talks for Freshers and returning students and is working closely with teaching staff across the Department to support students to reflect on inclusive design, and ED&I experiences. Provision in this area is driven by a broad belief that both ED&I perspectives and climate and sustainability education necessitate an engagement with students’ broader lived experience, an insight borne out by the focus groups.
Find out more
Do you want to know more about embedding climate and sustainability skills in your teaching? Find guidance and resources for embedding sustainability in the curriculum on the Teaching Hub, or learn more about Climate Action Education at Bath.

Related Articles
- Case Study: A course-wide approach to embedding climate and sustainability skills in Electronic and Electrical Engineering degrees
- Embedding sustainability in the curriculum
- Case study: Empowering students with design thinking
- Identifying sustainability in the curriculum
Article Contents

IMAGES
VIDEO
COMMENTS
View Case Studies. "One of the great things about Simplexity is that their expertise is fairly wide ranging throughout the product development process. It's great to have a partner like Simplexity who has so much expertise in electro-mechanical systems as well as embedded software because it brings so much capability to my engineering team in ...
Here's a brief overview of three companies that excelled in product engineering, along with the strategies they employed and the lessons learned: 1. Apple Inc. Case Study: Apple's Product Engineering Excellence. Strategies Employed: Integrated Ecosystem: Apple designs its products, software, and services to work seamlessly together. This ...
20 Product Management Case Studies [Detailed Analysis] [2024] 1. Apple Inc. - Reinventing the Smartphone. Task/Conflict: Apple's entry into the already crowded mobile phone market was a bold move, particularly with the objective of introducing a product that wasn't just another addition but a complete redefinition of what a mobile phone ...
That's where Deloitte's Product Engineering and Development practice comes in. ... and business case Using this process, we are able to leverage our unique, pre-configured and industry-tested platforms—DPLM and DMES—, processes, ... Case studies Global automotive OEM Large life sciences company Large telecom services provider Our client ...
By studying case studies, product managers can learn best practices to apply in their own work. ... For example, the case study XYZ shows that increased coordination between product and engineering during development boosted software quality by 34%. Similarly, early designer inclusion at ACME refined the user interface and improved conversion ...
Journey Mapping: A Product Development Process Case Study. Product teams should regularly assess the product development process itself. Sebastian Gherman, a Toptal senior product manager, outlines his approach. Sebastian Gherman is one of the longest-tenured product managers on Toptal's core team. A former software engineer, he focuses ...
Product engineering is a process of applying engineering principles, design thinking, and project management to improve user experience, software quality and enable businesses to deploy high-performance systems. Product engineering provides key benefits of cost-effectiveness, quality improvement, and enhanced security.
Agile Case Study Examples. 1. Moving towards Agile: Managing Loxon Solutions. Following is an Agile case study in banking: Problem: Loxon Solutions, a Hungarian technology startup in the banking software industry, faced several challenges in its journey towards becoming an agile organization.
We curated 50 product management case studies that will help you improve as a product manager in different stages of your career. airbnb. 50 Product Management Case Studies. Producter is a product management tool designed to become customer-driven. It helps you collect feedback, manage tasks, sharing product updates, creating product docs, and ...
Product engineering is a phase of product development in which engineering principles and practices are applied to a product lifecycle. The process includes design, development, testing, and optimization. Product engineering can refer to software engineering or the design of new products like electronics, industrial equipment, or automobiles.
Design for Manufacturing Examples: Real-Life Engineering Case Studies. Abe Chaves. | April 18, 2024. Learn more about aPriori's Manufacturability Insights capabilities in this brochure. Key Takeaways: More than 70% of a part's cost can be locked in during the early design phase. Adopt a robust DFM process using digital manufacturing ...
After analyzing all of these case studies and working with 100s of designers in our product design course to get them ready for the job application process, we've created our own tried-and-true templates to make it easy for designers to replicate the successful format and structure of these top portfolios using Notion.
A product management case study shares how a product was planned, built, and promoted, with key lessons for product managers. Here are six unmissable case studies. ... In late 2012 Eric Colson, then the VP of data science and engineering at Netflix, joined Lake on her journey of crafting the future of retail.
This study sheds light on how product outcomes shape the direction of innovation and markets for technology. In the drug development industry in particular, negative product shocks appear to spur investment changes both within the directly affected firm and in competing firms in the same R&D markets.
Product engineering is the end-to-end process of creating and maintaining software products. It encompasses everything from conceptualization to ongoing support, ensuring that the software meets user needs and aligns with market demands effectively. ... Case Studies; Contact Us Topics Product Engineering. By NewVision Team September 5, 2023 ...
Key Strategies for Acing Case Studies. Here are proven strategies to shine in your PM case study interview: Research the company: Review their products, customers, domain etc. Helps tailor your approach to their context. For example, studying an ecommerce company's key metrics will allow you to anchor examples and data points in their specifics.
Keywords: product generation engineering, case study, dual mass flywheel, contact and channel approach. 1. Introduction and motivation. Innovations are the result of transferring recognized customer value (product profile) into a new product (invention) and distributing and establishing that product in the market (diffusion) [Schumpeter 1934].
There are numerous examples of successful product case studies that businesses can use to inspire their strategies. One example is the Dropbox case study, a written case study that showcases Dropbox's product's integration with other services, cost savings for businesses, and customer feedback. Another example is the Hubspot case study, a video ...
Case studies are more than just success stories.They are powerful tools that demonstrate the practical value of your product or service. Case studies help attract attention to your products, b. We've put together 15 real-life case study examples to inspire you. These examples cover a variety of industries and formats, plus templates to ...
In fact, studies have shown that product dissection can help students relate classroom material to real-life engineering problems [21], help engage first year engineering students in learning [22 ...
L. Brownsword, and P. Clements, "A Case Study in Successful Product Line Development," Carnegie Mellon University, Software Engineering Institute's Digital Library.Software Engineering Institute, Technical Report CMU/SEI-96-TR-016, 1-Oct-1996 [Online].
Why it's exemplary: Real-world engineering decision making involves multiple actors and, for each, ethical considerations may arise at multiple levels—personal, professional, societal, or global.Our program of case studies and educational materials is exemplary in its interdisciplinary foundation, created collectively by engineers, policy experts, business professionals, and ethicists to ...
The latest edition in the gold standard of project management case study collections As a critical part of any successful, competitive business, project management sits at the intersection of several functional areas. And in the newly revised Sixth Edition of Project Management Case Studies , world-renowned project management professional Dr. Harold Kerzner delivers practical and in-depth ...
This case study outlines how the Department of Electronic and Electrical Engineering at University of Bath redesigned their core undergraduate curriculum to offer students practical skills related to climate and sustainability. It offers practical insights into how these skills can be embedded in engineering degrees.
The primary objective of this case is to introduce the notion of why and how businesses are transforming by leveraging digital information technologies. Additionally, the case reinforces concepts typically covered in today's introductory information technology and systems (ITS) courses. The secondary objective of the case is to illustrate the importance of integrating information technology ...
Background. The following case study illustrates how the Predicted Crash Frequency with CMF Adjustment method has been used to explicitly consider the safety impacts of opportunities during the Value Engineering (VE) process. Specifically, it focuses on the quantification of safety in the evaluation phase when safety is a project factor and crash frequency is the related performance measure.
Pharmaceutical and biotechnology companies employ platform analytical procedures in the development stages of their synthetic and biological drug products and are beginning to leverage them for commercial products. This shift is supported by the acceptance of platform procedures in the recently adopted ICH Q2(R2) and ICH Q14. Six case studies are shared in this article to highlight how ...
In the evolving realm of ergonomics, there is a growing demand for enhanced comfortability, visibility, and accessibility in the operation of engineering machinery. This study introduces an innovative approach to assess the ergonomics of a driller's cabin by utilizing a digital human. Through the utilization of inertial motion capture sensors, the method enables the operation of a virtual ...
At the University of Zurich, over a decade of pre-clinical and proof-of-concept studies led to Swiss authorities approving a phase I clinical trial for this tissue-engineering therapy for female ...
Case study: Student focus groups in Chemical Engineering map path to enhanced climate and sustainability teaching in undergraduate curriculum. The Department of Chemical Engineering at University of Bath has run focus groups with students to understand how staff can enhance climate and sustainability teaching provision in their courses.