Suggestions or feedback?

MIT News | Massachusetts Institute of Technology
- Machine learning
- Sustainability
- Black holes
- Classes and programs
Departments
- Aeronautics and Astronautics
- Brain and Cognitive Sciences
- Architecture
- Political Science
- Mechanical Engineering
Centers, Labs, & Programs
- Abdul Latif Jameel Poverty Action Lab (J-PAL)
- Picower Institute for Learning and Memory
- Lincoln Laboratory
- School of Architecture + Planning
- School of Engineering
- School of Humanities, Arts, and Social Sciences
- Sloan School of Management
- School of Science
- MIT Schwarzman College of Computing
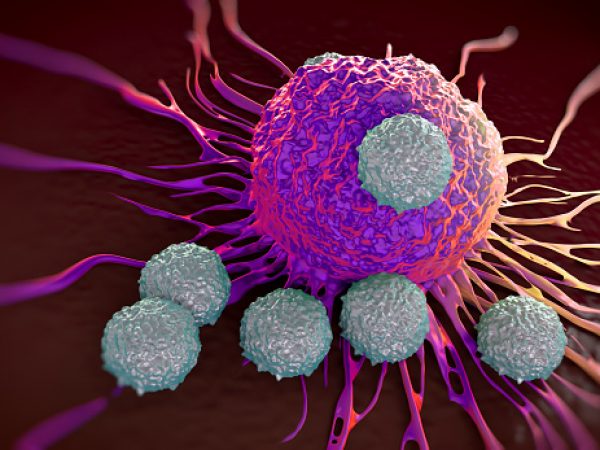
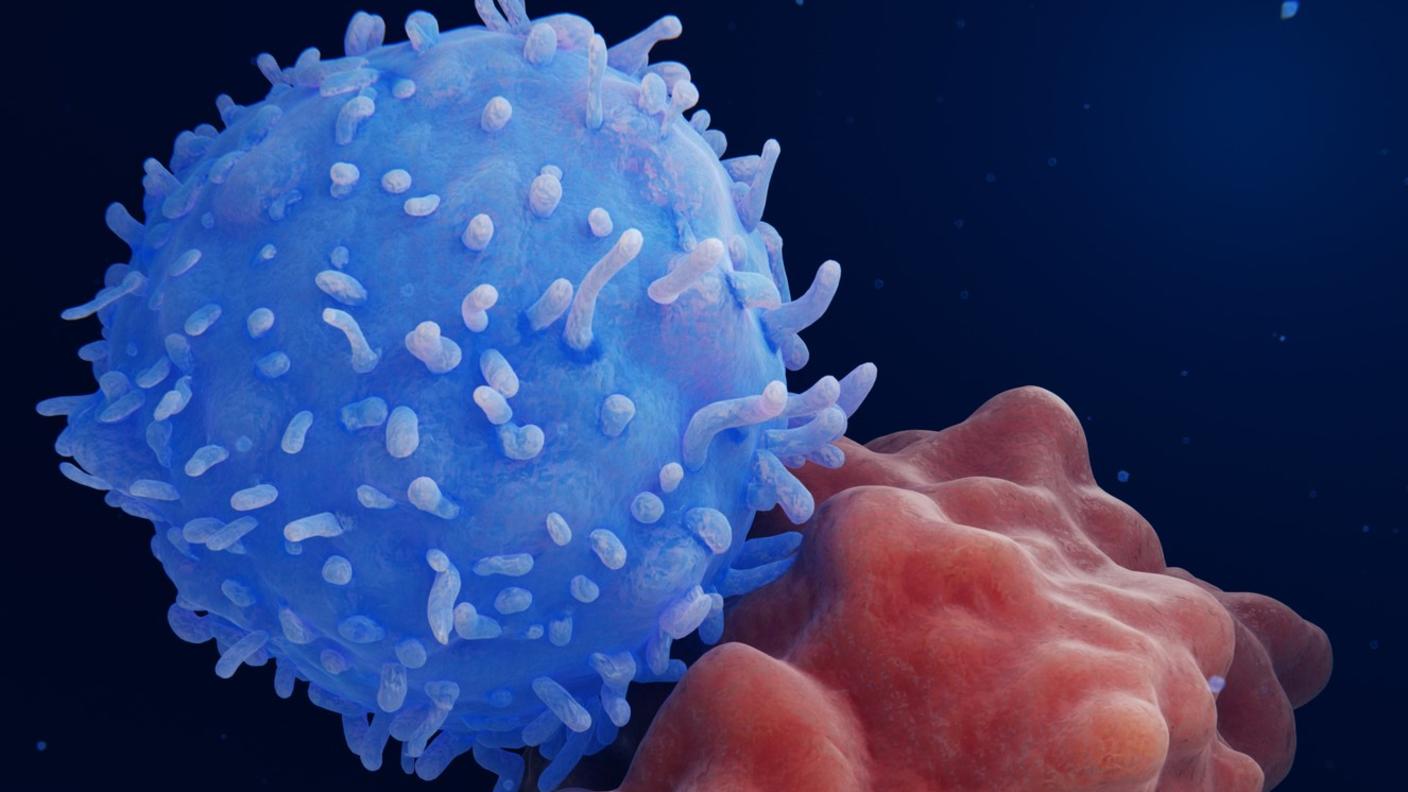
New cancer treatment may reawaken the immune system
Press contact :, media download.
*Terms of Use:
Images for download on the MIT News office website are made available to non-commercial entities, press and the general public under a Creative Commons Attribution Non-Commercial No Derivatives license . You may not alter the images provided, other than to crop them to size. A credit line must be used when reproducing images; if one is not provided below, credit the images to "MIT."
Previous image Next image
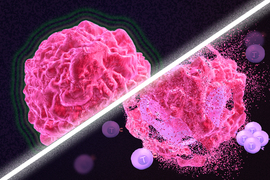
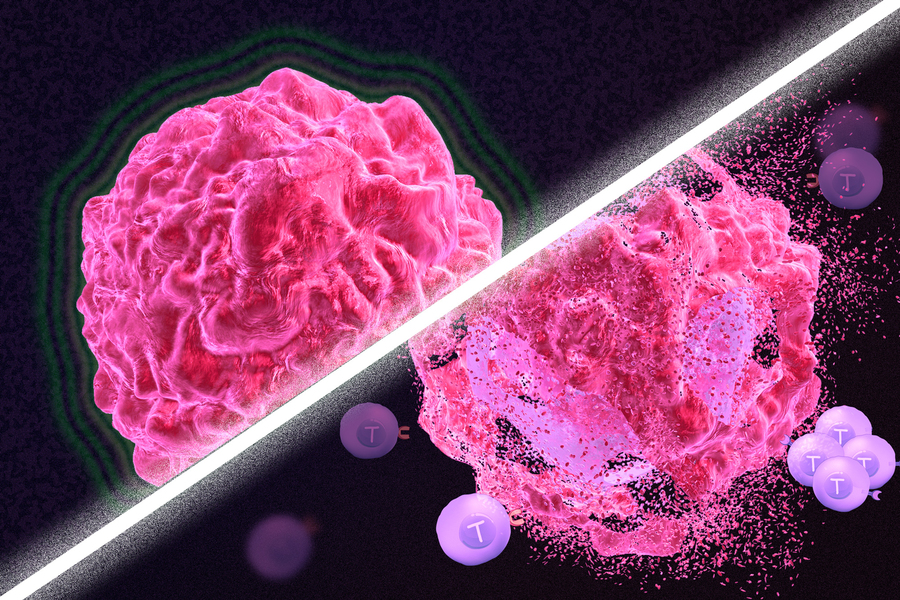
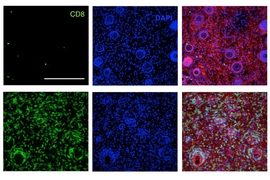
Immunotherapy is a promising strategy to treat cancer by stimulating the body’s own immune system to destroy tumor cells, but it only works for a handful of cancers. MIT researchers have now discovered a new way to jump-start the immune system to attack tumors, which they hope could allow immunotherapy to be used against more types of cancer.
Their novel approach involves removing tumor cells from the body, treating them with chemotherapy drugs, and then placing them back in the tumor. When delivered along with drugs that activate T cells, these injured cancer cells appear to act as a distress signal that spurs the T cells into action.
“When you create cells that have DNA damage but are not killed, under certain conditions those live, injured cells can send a signal that awakens the immune system,” says Michael Yaffe, who is a David H. Koch Professor of Science, the director of the MIT Center for Precision Cancer Medicine, and a member of MIT’s Koch Institute for Integrative Cancer Research.
In mouse studies, the researchers found that this treatment could completely eliminate tumors in nearly half of the mice.
Yaffe and Darrell Irvine, who is the Underwood-Prescott Professor with appointments in MIT’s departments of Biological Engineering and Materials Science and Engineering, and an associate director of the Koch Institute, are the senior authors of the study, which appears today in Science Signaling . MIT postdoc Ganapathy Sriram and Lauren Milling PhD ’21 are the lead authors of the paper.
T cell activation
One class of drugs currently used for cancer immunotherapy is checkpoint blockade inhibitors, which take the brakes off of T cells that have become “exhausted” and unable to attack tumors. These drugs have shown success in treating a few types of cancer but do not work against many others.
Yaffe and his colleagues set out to try to improve the performance of these drugs by combining them with cytotoxic chemotherapy drugs, in hopes that the chemotherapy could help stimulate the immune system to kill tumor cells. This approach is based on a phenomenon known as immunogenic cell death, in which dead or dying tumor cells send signals that attract the immune system’s attention.
Several clinical trials combining chemotherapy and immunotherapy drugs are underway, but little is known so far about the best way to combine these two types of treatment.
The MIT team began by treating cancer cells with several different chemotherapy drugs, at different doses. Twenty-four hours after the treatment, the researchers added dendritic cells to each dish, followed 24 hours later by T cells. Then, they measured how well the T cells were able to kill the cancer cells. To their surprise, they found that most of the chemotherapy drugs didn’t help very much. And those that did help appeared to work best at low doses that didn’t kill many cells.
The researchers later realized why this was so: It wasn’t dead tumor cells that were stimulating the immune system; instead, the critical factor was cells that were injured by chemotherapy but still alive.
“This describes a new concept of immunogenic cell injury rather than immunogenic cell death for cancer treatment,” Yaffe says. “We showed that if you treated tumor cells in a dish, when you injected them back directly into the tumor and gave checkpoint blockade inhibitors, the live, injured cells were the ones that reawaken the immune system.”
The drugs that appear to work best with this approach are drugs that cause DNA damage. The researchers found that when DNA damage occurs in tumor cells, it activates cellular pathways that respond to stress. These pathways send out distress signals that provoke T cells to leap into action and destroy not only those injured cells but any tumor cells nearby.
“Our findings fit perfectly with the concept that ‘danger signals’ within cells can talk to the immune system, a theory pioneered by Polly Matzinger at NIH in the 1990s, though still not universally accepted,” Yaffe says.
Tumor elimination
In studies of mice with melanoma and breast tumors, the researchers showed that this treatment eliminated tumors completely in 40 percent of the mice. Furthermore, when the researchers injected cancer cells into these same mice several months later, their T cells recognized them and destroyed them before they could form new tumors.
The researchers also tried injecting DNA-damaging drugs directly into the tumors, instead of treating cells outside the body, but they found this was not effective because the chemotherapy drugs also harmed T cells and other immune cells near the tumor. Also, injecting the injured cells without checkpoint blockade inhibitors had little effect.
“You have to present something that can act as an immunostimulant, but then you also have to release the preexisting block on the immune cells,” Yaffe says.
Yaffe hopes to test this approach in patients whose tumors have not responded to immunotherapy, but more study is needed first to determine which drugs, and at which doses, would be most beneficial for different types of tumors. The researchers are also further investigating the details of exactly how the injured tumor cells stimulate such a strong T cell response.
The research was funded, in part, by the National Institutes of Health, the Mazumdar-Shaw International Oncology Fellowship, the MIT Center for Precision Cancer Medicine, and the Charles and Marjorie Holloway Foundation.
Share this news article on:
Related links.
- Department of Biology
- Department of Biological Engineering
- Department of Materials Science and Engineering
- Koch Institute
- Ragon Institute
Related Topics
- Biological engineering
Related Articles
Cancer biologists identify new drug combo
A boost for cancer immunotherapy
Fighting cancer with the power of immunity
Previous item Next item
More MIT News

Duane Boning named vice provost for international activities
Read full story →

Q&A: Undergraduate admissions in the wake of the 2023 Supreme Court ruling
Study reveals the benefits and downside of fasting

MIT engineers’ new theory could improve the design and operation of wind farms

Engineering and matters of the heart

Creating connection with science communication
- More news on MIT News homepage →
Massachusetts Institute of Technology 77 Massachusetts Avenue, Cambridge, MA, USA
- Map (opens in new window)
- Events (opens in new window)
- People (opens in new window)
- Careers (opens in new window)
- Accessibility
- Social Media Hub
- MIT on Facebook
- MIT on YouTube
- MIT on Instagram

- Cancer Biology
Latest articles
Identification of novel myelodysplastic syndromes prognostic subgroups by integration of inflammation, cell-type composition, and immune signatures in the bone marrow, deuterium metabolic imaging phenotypes mouse glioblastoma heterogeneity through glucose turnover kinetics.
- Cell Biology
Topological stress triggers persistent DNA lesions in ribosomal DNA with ensuing formation of PML-nucleolar compartment
Inhibition of ulk1/2 and kras g12c controls tumor growth in preclinical models of lung cancer, exploring the role of the immune microenvironment in hepatocellular carcinoma: implications for immunotherapy and drug resistance, rgs10 deficiency facilitates distant metastasis by inducing epithelial–mesenchymal transition in breast cancer.
- Neuroscience
Tumor-infiltrating nerves functionally alter brain circuits and modulate behavior in a mouse model of head-and-neck cancer
- Genetics and Genomics
Circulating small extracellular vesicle RNA profiling for the detection of T1a stage colorectal cancer and precancerous advanced adenoma
Super-enhancer-driven zfp36l1 promotes pd-l1 expression in infiltrative gastric cancer.
- Computational and Systems Biology
Robust estimation of cancer and immune cell-type proportions from bulk tumor ATAC-Seq data
Reproducibility in cancer biology.

eLife publishes final papers from the Reproducibility Project: Cancer Biology
Toxicity: fishing for protective compounds, senior editors.
- See more editors
Be the first to read new articles from eLife

Select "Patients / Caregivers / Public" or "Researchers / Professionals" to filter your results. To further refine your search, toggle appropriate sections on or off.
Cancer Research Catalyst The Official Blog of the American Association for Cancer Research

Home > Cancer Research Catalyst > New Dimensions in Cancer Biology: Updated Hallmarks of Cancer Published
New Dimensions in Cancer Biology: Updated Hallmarks of Cancer Published
The new year brings a new chapter in the holy book of cancer biology with the publication in the AACR journal Cancer Discovery of Hallmarks of Cancer: New Dimensions , an update to the “Hallmarks of Cancer” series. The latest edition was saluted with great enthusiasm by the scientific community as the new piece of an iconic saga, with many scientists taking to Twitter to share their excitement about seeing their field of study acknowledged among the fundamentals of cancer biology.
The original article of the seminal series was published in 2000 by Robert Weinberg, PhD, FAACR , from the Whitehead Institute for Biomedical Research and the Massachusetts Institute of Technology, and Douglas Hanahan, PhD, FAACR , from EPFL, the Swiss Federal Institute of Technology in Lausanne. The authors, both cancer research pioneers, organized state-of-the-art knowledge on cancer into a logical framework that recapitulated the extraordinary complexity of the disease in a small set of fundamental traits shared by most, if not all, human cancers. In addition, they introduced the concept of “enabling characteristics,” or means that enable premalignant cells to acquire the six hallmarks of cancer.
This landmark review soon became an essential resource for cancer researchers, with tens of thousands of citations, providing a comprehensive foundation for understanding and studying cancer biology. To account for new discoveries and progress in the field, the authors provided a first update in 2011, adding two emerging hallmarks and a new enabling characteristic.
In “Hallmarks of Cancer: New Dimensions,” Hanahan further revisited the list, proposing one new emerging hallmark and two additional enabling characteristics.
Read on to learn more about the hallmarks of cancer, how they were expanded over time, and the latest additions.
The original hallmarks (2000)
Writing about the overwhelming complexity of the scientific literature on cancer in 2000, Weinberg and Hanahan forecasted that, rather than adding more information in a chaotic fashion, research in the next quarter century would bring a conceptual shift towards a more logical approach to decipher such complexity “in terms of a small number of underlying principles.” The original “ Hallmarks of Cancer ” review was the authors’ effort and contribution to this shift, leading to the enumeration of six core “rules” that orchestrate the multistep process of the transformation of normal cells into malignant cells:
- Self-sufficiency in growth signals . While normal cells depend on external growth signals for proliferation, cancer cells can generate most of the growth signals by themselves, greatly reducing, or eliminating, their dependence on external stimuli. A corollary to this observation was a new view of cancer as a complex tissue in which malignant cells co-opt the surrounding normal cells to provide the necessary growth signals, serving as active collaborators, rather than passive bystanders.
- Insensitivity to growth suppressive signals . Multiple antiproliferative signals maintain the homeostasis in normal tissues, pushing cells out of the cell cycle and into a temporary quiescent state, or sending them into their terminal, post-mitotic differentiation state. Transformed cells evade these antiproliferative signals by subverting the mechanisms that control cell cycle progression—for example, by disrupting the pRb pathway, and overexpressing growth-stimulating factors such as c-myc.
- Ability to evade programmed cell death . Apoptosis is a major anticancer barrier, as becoming immortal is another way through which cancer cells continue to expand in number. Further, the authors proposed that the redundancy in cell death mechanisms could be exploited for therapeutic purposes.
- Enabling replicative immortality. In order for cancer to grow, malignant cells have to proliferate indefinitely. While normal cells possess a limited proliferative potential and will stop dividing at some point if cultured in vitro, cancer cells have lost that restrain mechanism, governed by telomere shortening. To become immortal, malignant cells rely on the telomerase enzyme to maintain the length of their telomeres above a critical threshold that allows them to go on dividing.
- Sustained angiogenesis. The growing tumor tissue has increased oxygen and nutrient needs and, to keep expanding, it needs to trigger the formation of new vasculature by releasing pro-angiogenic signals. At the time the authors codified this feature, it had been established that tumors go through an “angiogenic switch” that allows them to grow from microscopic to macroscopic lesions.
- Tissue invasion and metastasis . Metastasis is the cause of the vast majority of cancer deaths. The ability to invade, settle in, and grow in distant tissues is therefore one of the main features of cancer and relies on modifications in the cancer cell interactions with their surrounding environment through e-cadherin, integrins, and other adhesion molecules, and the production of matrix-degrading proteases.
The acquisition of multiple mutations through the loss of one or more mechanisms designated to protecting genome integrity was presented by the authors as an enabling characteristic that allows cancer cells to reach the six “biological endpoints” illustrated above.
Weinberg and Hanahan described the six capabilities acquired by cancer cells as the successful breaching of just as many anticancer defense mechanisms wired into our cells, and suggested these characteristics were shared by the more than 100 distinct types of cancer known at the time. Thus, the hallmarks of cancer provided a few unifying concepts toward which future cancer research could gravitate.
“The next generation” (2011)
In 2011, Weinberg and Hanahan published an update discussing the progress made over the preceding decade in the knowledge about the six original hallmarks. They also incorporated two emerging hallmarks:
- Reprogramming energy metabolism . While normal cells use oxygen to process glucose and produce energy, malignant cells can switch to aerobic glycolysis even in the presence of oxygen (what is known as the Warburg effect). Though this mechanism is less efficient, it is faster and originates several intermediate precursors used by cancer cells as building blocks to make proteins, DNA, and lipids to support their fast proliferation. Other cancer cells can use lactate as their main energy source.
- Evading immune destruction . Hanahan and Weinberg discussed evidence supporting the central role played by the immune system as a barrier to tumorigenesis, including studies in mouse models demonstrating that carcinogen-induced tumors developed and grew more rapidly in immunodeficient mice, especially if they lacked cytotoxic and helper T cells or natural killer cells, and observations that human tumors with high immune infiltration had better prognosis.
The 2011 edition also identified tumor-promoting inflammation as a new enabling characteristic. While immune infiltrates were historically considered a sign of the immune system reacting against the tumor, at the time the second review was published, the tumor-promoting effect of certain inflammatory cells had become clear. The authors discussed how inflammation favors multiple hallmark capabilities by providing growth, survival, and proangiogenic factors, and releasing chemicals, such as reactive oxygen species, that can cause additional mutations in the nearby cancer cells.
The review also contains a paragraph on the tumor microenvironment, which in the previous decade had become the subject of extensive research showing that, when studying the biology of a tumor, one needs to consider both the cancer cells and the microenvironment they construct around them.
“New Dimensions:” Expanding the Frontiers of Cancer Biology (2022)
Ten years later, Hanahan goes back to the hallmarks once more, recognizing the great progress made in the study of cancer through big data, and reaffirming the impact of the hallmarks of cancer in conceptualizing the new discoveries and “helping to distill this complexity into an increasingly logical science.” In the latest article, the two hallmarks added as emerging in 2011 were definitively incorporated as core hallmarks, as research in the past 10 years has largely confirmed the importance of metabolic reprogramming and avoiding immune destruction in cancer. In addition, Hanahan proposed an additional emerging hallmark:
- Phenotypic plasticity and disrupted differentiation . Terminal differentiation in normal cells is associated with a permanent proliferation arrest, and increasing evidence indicates that malignant cells evade differentiation and unlock what is known as phenotypic plasticity to continue to grow. In other words, they can change their identity into something that is more inclined to proliferate. This can happen in different ways: Cells that are approaching full differentiation can de-differentiate back to a progenitor-like state; neoplastic cells originating from an undifferentiated progenitor cell can halt the differentiation process and remain in that partially differentiated, progenitor-like state; and cells that were committed to a certain differentiation phenotype can switch developmental programs, or transdifferentiate, acquiring traits that are not associated with their cell of origin. Hanahan notes that, as it is true for other hallmark capabilities, cellular plasticity is not a “novel invention” of cancer cells, but rather a malignant twist on existing mechanisms that some normal cells can activate to repair and regenerate normal tissues.
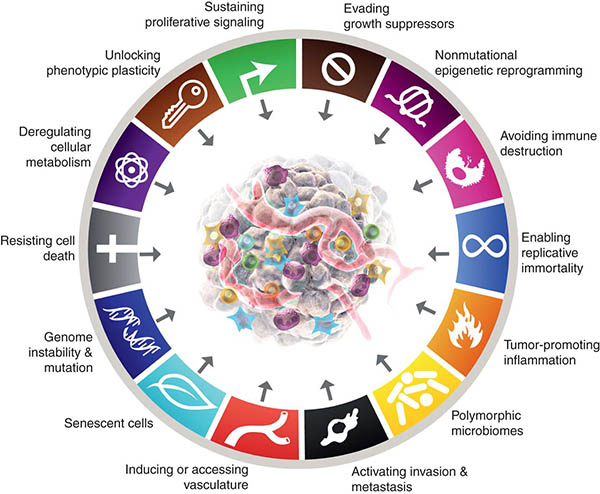
The new article also highlights two new enabling characteristics:
- Non-mutational epigenetic reprogramming. Global changes in the epigenetic landscape are indeed recognized as a common feature of many cancers. Reproducing what happens during normal embryogenesis and development, cancer cells can reprogram a large number or gene-regulation networks to alter gene expression and favor the acquisition of hallmark capabilities.
- The microbiome. Our body is colonized by a vast array of microorganisms—nearly 40 trillion cells—that live in and on us. Their profound contribution to human health and disease is now appreciated. For example, researchers have found that some of these microorganisms can exert protective or deleterious effects on cancer development, progression, and response to therapy.
The new edition acknowledged the importance of senescent cells as instrumental components of the tumor microenvironment. While in the 2000 edition the authors discussed senescence as a possible anticancer barrier, they did not rule out the possibility of it being an artifact of cell culture that did not represent a real cell phenotype in vivo. More than two decades later, the role of cellular senescence in tissue homeostasis and cancer is well recognized, and significant morphological and metabolic features associated with it have been uncovered. Research has also shown how, in certain contexts, senescent cells can stimulate tumor development and malignant progression. Therefore, Hanahan proposed that senescent cells should be included as significant components of the tumor microenvironment.
In the concluding remarks, Hanahan explains how, while some of the hallmarks are now well validated, the newest features added as emerging hallmarks are meant to serve as “trial balloons” to stimulate debate within the cancer research community and inspire new investigations that will keep refining our understanding of cancer biology.
On to the next decade of discoveries.
- About This Blog
- Blog Policies
- Tips for Contributors
AACR Annual Meeting 2017: Immunotherapy Provides Long-lasting Responses to Certain Cancer Types

Cancer Research: Back to Basics

Experts Forecast Cancer Research and Treatment Advances in 2018
Cancel reply
Your email address will not be published. Required fields are marked *
Join the Discussion (max: 750 characters)...
This site uses Akismet to reduce spam. Learn how your comment data is processed .
Cancers are a complicated disease that causes rapid multiplication and growth of specific cells of the body. It can lead to cancer or neoplasm, an unusual mass of tissues. Oncology is a medical discipline dealing with the diagnosis and management of cancer. If left untreated, it can progress into serious conditions endangering the life of the patients. Understanding disease at the cellular level can upgrade. The accuracy of disease diagnosis, management, and prevention of cancers. Additionally, a solid understanding of cancer with the assistance of disease tests from patients encourages clinical interpretations to forestall, treat and alleviate disease and related intricacies.
https://centralbiohub.de/biospecimens/cancer-samples
- Understanding How Cancer Develops
- Scientific Publishing
- Understanding the Path to Cancer Development
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List

Recent Developments in Cancer Systems Biology: Lessons Learned and Future Directions
Kazim y. arga.
1 Department of Bioengineering, Marmara University, Istanbul 34722, Turkey
Raghu Sinha
2 Department of Biochemistry and Molecular Biology, Penn State College of Medicine, Hershey, PA 17033, USA
Cancer is a complex disease involving multiple mechanisms and critical players, at broad genomic, transcriptional, translational and/or biochemical levels. One could envision discovering new biomarkers for early detection by understanding the behavior of cancer development and progression, but to date, there are few biomarkers approved for use in the clinical setting. Therefore, there is a critical need to improve strategies and methods by using novel state-of-the-art tools and strategies to identify and validate newer biomarkers. In addition to biomarkers, there is also a demand for effective methods to identify new targets to inhibit tumor growth. Technically, there is a growing requirement to find new targets using personalized approaches in a targeted and much more effective manner, as existing drugs often become resistant over time in cancer patients. Opportunities to improve this strategy would, therefore, be to find better druggable targets and provide options for drug combinations and/or drug repurposing. More importantly, the ultimate goal of an oncologist and the desire of the cancer patient is to improve overall survival and this could be achieved in part through better prognostic models. Cancer systems biology has undoubtedly emerged as an integrative tool to achieve such advances.
This Special Issue on “recent developments in cancer systems biology” has compiled several novel approaches that use cutting-edge technologies to build a strong foundation of systems biology in cancer research. The issue includes eight original research articles and four literature reviews on recent efforts that use a variety of in silico tools along with experimental approaches to discover novel biomarker candidates for diagnosis and prognosis and to identify drugs and their targets for treatments that could be used in thirteen cancers and their subtypes.
Several “omics” investigations, including genomics, proteomics, metabolomics, glycomics and metagenomics, provide potential candidate biomarkers that can be measured in plasma, tissue and saliva in several lethal cancer types including Pancreatic Cancer [ 1 ]. Integrative analysis of these “omics” data would likely discover novel biomarkers for diagnosis and prognosis as well as targets for effective therapy. Moreover, distinguishing clinically similar cancers can be challenging and focusing on genomic and transcriptomic variations may prove beneficial, this issue describes details on various methods available for ovarian and breast cancers [ 2 ] and types of lung cancer [ 3 , 4 ] and renal cell carcinoma [ 5 ] for identifying key genes and pathways that might assist in proposing diagnostic and prognostic predictions. In addition, integrating multi-omics is important particularly in the use of patient-derived experimental models [ 6 ] that can be used in the clinical setting to provide personalized treatment options. Another genome-level advancement that surpasses next-generation sequencing is the identification of somatic structural variants (SVs) that influence functional and cancer-related genes [ 7 ]. This optical genome mapping and SVs analysis can be applied to a variety of solid tumors for better cancer prognosis and treatment.
Discovering new targets in cancers provide opportunities especially for recurrences since the drug resistance is proving to be challenging to treat. Several drug targets have been identified using transcriptomics and biological networks in different cancer types including miR-1246 targeting several genes [ 4 ] and Sestrin-2 [ 8 ] in lung adenocarcinomas, ELK1 [ 9 ] and ETS [ 10 ] genes in glioma. Additionally, drug repurposing strategies are not only extensively used to discover new uses for already approved drugs, but also provide opportunities for potentially treatment of drug resistance in various cancers. In another article [ 11 ], drug repurposing efforts were reviewed in triple-negative breast cancer, an aggressive breast cancer subtype that has a high rate of recurrence and metastasis. These authors compared different repurposing strategies, including structure-based, transcription signature-based, biological network-based and data mining-based drug repositioning. In another study, seven distinct gene programs representing different biological processes involved in drug-induced changes in AML were identified [ 12 ]. Furthermore, a data-driven dynamic model of acquired resistance to combined drugs was constructed by these authors and revealed several interventions that can specifically disrupt portions of the system-wide drug response, which could allow co-targeting and lead to synergistic treatments that can overcome resistance and prevent potential recurrence.
In conclusion, all of the articles published in this Special Issue cover recent developments with attractive approaches to a wide range of topics encompassing the Cancer Systems Biology. These articles and reviews propose a variety of biomarkers for clinical diagnosis, prognosis and therapeutic strategies including “drug repurposing“ for various cancers that pose a major health challenge with significant socioeconomic consequences. We would like to make an appeal to researchers around the world to join forces and contribute to the development of a common platform for personalized medicine using a combination of the different biomarkers proposed in this Special Issue in a diagnostic and/or prognostic setting, allowing the identification of patients at risk, which would facilitate the early initiation of personalized treatments. This Special Issue also highlights the various predictive models and the use of integrated biological network analysis to identify target genes and correlate them with prognosis. It is of utmost importance that all predictive models must undergo extensive validation.
Acknowledgments
We are very grateful to all the authors who have provided excellent contributions to this Special Issue. Moreover, we would like to thank Journal of Personalized Medicine for offering us the opportunity to make this Special Issue a reality and in particular, Esther Cao for her availability, professionalism, help and constant presence, support and kindness. Finally, we would like to acknowledge the excellent and efficient work of the expert reviewers who reviewed submissions in a timely, fair and constructive manner.
The research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Support Biology
Dei council and dei faculty committee, biology diversity community, mit biology catalyst symposium, honors and awards, employment opportunities, faculty and research, current faculty, in memoriam, areas of research, biochemistry, biophysics, and structural biology, cancer biology, cell biology, computational biology, human disease, microbiology, neurobiology, stem cell and developmental biology, core facilities, video gallery, faculty resources, undergraduate, why biology, undergraduate testimonials, major/minor requirements, general institute requirement, advanced standing exam, transfer credit, current students, subject offerings, research opportunities, biology undergraduate student association, career development, why mit biology, diversity in the graduate program, nih training grant, career outcomes, graduate testimonials, prospective students, application process, interdisciplinary and joint degree programs, living in cambridge, graduate manual: key program info, graduate teaching, career development resources, biology graduate student council, biopals program, postdoctoral, life as a postdoc, postdoc associations, postdoc testimonials, workshops for mit biology postdocs entering the academic job market, responsible conduct of research, postdoc resources, non-mit undergraduates, bernard s. and sophie g. gould mit summer research program in biology (bsg-msrp-bio), bsg-msrp-bio gould fellows, quantitative methods workshop, high school students and teachers, summer workshop for teachers, mit field trips, leah knox scholars program, additional resources, mitx biology, department calendar, ehs and facilities, graduate manual, resources for md/phd students, preliminary exam guidelines, thesis committee meetings, guidelines for graduating, mentoring students and early-career scientists, remembering stephen goldman (1962 – 2022).
cancer gene discovery • tumorigenesis • cancer therapy and resistance • oncogenes • tumor suppressor genes • cancer models • growth control and cell proliferation • metastasis • cell proliferation • cell death • cell-cell and cell-matrix interactions • microenvironment •DNA repair and replication • transcription • chromosome stability • metabolism • immunology and cancer • immunotherapy • cancer stem cells
Eliezer Calo
Lindsay case, jianzhu chen, michael t. hemann, whitney henry, david housman, richard o. hynes, tyler jacks, sally kornbluth, douglas lauffenburger, jacqueline lees, alison e. ringel, francisco j. sánchez-rivera, phillip a. sharp, yadira soto-feliciano, stefani spranger, matthew vander heiden, robert a. weinberg, michael b. yaffe, omer h. yilmaz, richard a. young.

Douglas Lauffenburger fosters the interface of bioengineering, quantitative cell biology, and systems biology to determine fundamental aspects of cell dysregulation — identifying and testing new therapeutic ideas.
New technique reveals how gene transcription is coordinated in cells

“Rosetta Stone” of cell signaling could expedite precision cancer medicine
Taking RNAi from interesting science to impactful new treatments

Scientists develop a rapid gene-editing screen to find effects of cancer mutations
News brief: Calo Lab
How early-stage cancer cells hide from the immune system

How phase separation is revolutionizing biology
Study explains why certain immunotherapies don’t always work as predicted

Share Options
- Share to Facebook
- Share to Linkedin
UVA and NCI-Funded SASCO Center Marks Milestones in Cancer Cell Organelles Research
UVA’s Center for Systems Analysis of Stress-adapted Cancer Organelles (SASCO) recently hosted its first annual symposium, sharing updates on major projects and work to improve research inclusivity as the program enters its third year.
Founded and co-led by Kevin Janes , Ph.D., the John Marshall Money Professor of Biomedical Engineering, and Matthew J. Lazzara , Ph.D., professor of chemical and biomedical engineering, SASCO is funded through a $12 million National Cancer Institute (NCI) U54 grant to research functional sub-cellular compartments in cancer. Its ultimate goal: to improve fundamental understanding of the inner workings of cancer cells to pave the way for more effective therapies.
The July 23 symposium drew about 100 live and virtual participants, including UVA faculty, students and staff, as well as guests from the NCI, various U.S. universities and the National University of Singapore.
SASCO’s Latest Discoveries in Tumor Biology and Therapy Development
Keynote speaker Donita Brady, Ph.D., associate professor of cancer biology and assistant dean for inclusion, diversity and equity in research at the University of Pennsylvania’s Perelman School of Medicine, presented on “Harnessing Chemical Interactions to Explore Tumor Cell Biological Responses to Fluctuations in Metal and Micronutrient Availability.” Brady discovered that kinase enzymes require copper to function, raising the possibility of targeting tumor growth by altering copper import.
Janes shared an update on SASCO’s first two years, which saw three publications and five preprints, with program participation by 20 faculty, seven staff and 28 trainees. Faculty and students presented updates on SASCO’s three primary research projects, each focused on the role of different organelles in a specific type of cancer: the spindle assembly checkpoint in breast cancer; mitochondrial fragmentation colorectal cancer; and membrane stress in glioblastoma. Presentations were introduced by project co-leads P. Todd Stukenberg, Ph.D., professor of biochemistry and molecular genetics, David Kashatus, Ph.D., associate professor of microbiology, immunology, and cancer Biology, and B.J. Purow, M.D., professor of neurology, respectively.
Key SASCO research findings to date include hidden genetic and pharmacologic susceptibilities in chromosome segregation, new models of cancer cell metabolism, and a form of “cell death by drinking” tied to a brain cancer gene.
SASCO’s Commitment to Inclusive Cancer Studies
Associate professor of biomedical engineering and genome sciences Kristen Naegle , Ph.D., and SASCO center co-lead, presented on SASCO’s prioritization of inclusiveness in cell lines, tissues and sample data in computational research models, noting dramatic overrepresentation of white patients in available tissue samples, cell models and clinical data. To improve research outcomes for impacting all patients, SASCO is building awareness within the program, explicitly requesting that project aims include the populations burdened by the cancers they are studying and align their models to capture the diversity of that population.
SASCO's ‘Hot Ones’ Panel Heats Up the Symposium
The day continued with lighting talks and a poster session, followed by a career panel inspired by the celebrity talk show “Hot Ones.” Brave panelists, who included Brady and Lecia Robinson, Ph.D., assistant professor of Biology at Tuskegee University and a visiting faculty member with the Cancer Center’s Short-Term Research Initiative for Visiting Educators (STRIVE) program, took ten-minute turns fielding questions, each accompanied by a taste of progressively hotter salsa. Four interviews took place, and the questions were designed and asked by members of SASCO from broadly different career stages.
Janes said SASCO considers the inaugural symposium a success and hopes to convene an even larger group of researchers in 2025. In the meantime, the team looks forward to advancing their computer models and cell-culture experiments into real tumor settings and patient-derived material.
Related: NIH Invests $12M in UVA’s Cancer Research
UVA researchers are studying the tiniest parts inside cancer cells, or the cells’ “organelles,” to see if there is a way to stop tumors from forming.
UQ research could lead to a more effective treatment for bladder cancer
- Download PDF Copy
University of Queensland research could lead to a more effective immunotherapy treatment against bladder cancer.
A team led by Associate Professor Fernando Guimaraes from UQ's Frazer Institute has found the mechanism used by bladder cancer to suppress the immune system's natural killer (NK) cells.
We found that cancer suppresses immune cells by releasing a protein called TGF-β – and blocking that protein may offer a new therapy for some of the worst types of the cancer. NK cells are components of the immune system that naturally attack cancer cells and are a focus of emerging cancer treatments, including antibody-based immunotherapies. Our research is also a step towards clinical trials to explore the effectiveness of TGF-β inhibitors combined with NK cell-based therapies. Our work is all about unlocking the power of immunotherapy and our hope is that this leads to new bladder cancer therapies within 3 to 5 years." Dr. Fernando Guimaraes, Associate Professor, UQ's Frazer Institute
Bladder cancer is the ninth most common malignancy in the world – in 2023 more than 3000 people were diagnosed in Australia, with more than 1000 deaths.
The bladder cancer immune suppression mechanism was discovered by Dr Guimaraes' PhD candidate Joshua Wong while working at Queensland's Translational Research Institute (TRI).
Related Stories
- New trial explores BAFF CAR T-cell therapy for relapsed myeloma
- Advancements in neoantigen vaccines offer hope for personalized cancer therapy
- Ketogenic diet can boost efficacy of pancreatic cancer therapy
"It really was a eureka moment and it's extremely motivating to come in here every day and do your best work, because ultimately we are trying to find a cure for cancer," Mr Wong said.
The standard treatment for early-stage bladder cancer remains endoscopic surgery, occasionally combined with chemotherapy or immunotherapy.
For advanced forms of the disease, more invasive surgery or radiotherapy is combined with chemotherapy or immunotherapy, which is associated with significant ill health and life-long impacts on quality of life.
The UQ team collaborated with Princess Alexandra Hospital urologists including Dr Handoo Rhee, Professor Eric Chung and Dr Alice Nicol.
The research was published in The Lancet's eBioMedicine .
The University of Queensland
Joshua K.M. et al. (2024). TGF-β signalling limits effector function capacity of NK cell anti-tumour immunity in human bladder cancer. eBioMedicine . doi.org/10.1016/j.ebiom.2024.105176 .
Posted in: Medical Research News | Medical Condition News
Tags: Antibody , Bladder , Bladder Cancer , Cancer , Cell , Chemotherapy , Hospital , Immune System , Immunotherapy , Protein , Radiotherapy , Research , Surgery
Suggested Reading

Cancel reply to comment
- Trending Stories
- Latest Interviews
- Top Health Articles

Global and Local Efforts to Take Action Against Hepatitis
Lindsey Hiebert and James Amugsi
In this interview, we explore global and local efforts to combat viral hepatitis with Lindsey Hiebert, Deputy Director of the Coalition for Global Hepatitis Elimination (CGHE), and James Amugsi, a Mandela Washington Fellow and Physician Assistant at Sandema Hospital in Ghana. Together, they provide valuable insights into the challenges, successes, and the importance of partnerships in the fight against hepatitis.

Addressing Important Cardiac Biology Questions with Shotgun Top-Down Proteomics
In this interview conducted at Pittcon 2024, we spoke to Professor John Yates about capturing cardiomyocyte cell-to-cell heterogeneity via shotgun top-down proteomics.

A Discussion with Hologic’s Tim Simpson on the Future of Cervical Cancer Screening
Tim Simpson
Hologic’s Tim Simpson Discusses the Future of Cervical Cancer Screening.

Latest News

Newsletters you may be interested in
Your AI Powered Scientific Assistant
Hi, I'm Azthena, you can trust me to find commercial scientific answers from News-Medical.net.
A few things you need to know before we start. Please read and accept to continue.
- Use of “Azthena” is subject to the terms and conditions of use as set out by OpenAI .
- Content provided on any AZoNetwork sites are subject to the site Terms & Conditions and Privacy Policy .
- Large Language Models can make mistakes. Consider checking important information.
Great. Ask your question.
Azthena may occasionally provide inaccurate responses. Read the full terms .
While we only use edited and approved content for Azthena answers, it may on occasions provide incorrect responses. Please confirm any data provided with the related suppliers or authors. We do not provide medical advice, if you search for medical information you must always consult a medical professional before acting on any information provided.
Your questions, but not your email details will be shared with OpenAI and retained for 30 days in accordance with their privacy principles.
Please do not ask questions that use sensitive or confidential information.
Read the full Terms & Conditions .
Provide Feedback

News & Events
Latest blog posts & events.

In mice, chemotherapy made a KRAS inhibitor much more effective at shrinking pancreatic cancer.
NCI’s Dr. Stephanie Goff explains the science of, and data behind, CAR T cells and second cancers.

The combination shrank lymphoma and lung tumors in people and in mice.
Latest Press Releases

Immunotherapy approach shows potential in some people with metastatic solid tumors
- July 11, 2024
A new cellular immunotherapy approach shrank tumors in 3 of 7 patients with metastatic colon cancer, in a small NCI clinical trial. Normal white blood cells from each patient were genetically engineered to produce receptors that recognize and attack their specific cancer cells.

For healthy adults, taking multivitamins daily is not associated with a lower risk of death
- June 26, 2024
A large analysis of data from nearly 400,000 healthy U.S. adults followed for more than 20 years has found no association between regular multivitamin use and lower risk of death.

Combination targeted treatment produces lasting remissions in people with resistant aggressive B-cell lymphoma
- June 19, 2024
More than half of clinical trial participants treated with venetoclax, ibrutinib, prednisone, obinutuzumab, and lenalidomide (ViPOR) had substantial tumor shrinkage. Of those, 38% had tumors that disappeared completely.

Inherited genetic factors may predict the pattern of X chromosome loss in older women
- June 12, 2024
An NCI study explores the causes and effects of a phenomenon known as mosaic loss of chromosome X, or mLOX, which may lead to several health conditions, including cancer.

NIH scientists develop AI tool to predict how cancer patients will respond to immunotherapy
- June 3, 2024
AI tool developed by NCI researchers uses routine clinical data, such as that from a simple blood test, to predict whether someone’s cancer will respond to immune checkpoint inhibitors, a type of immunotherapy.
Media Resources
- Resources & Contacts
- Images and B-roll
- NCI Budget Data
- News Releases
Subscribe to NCI News Releases
NCI YouTube
Cancer Currents Blog
Find news and updates on the latest cancer research.
- View all posts
Subscribe to Cancer Currents
- Scientific Meetings and Lectures
- Conferences
- Advisory Board Meetings
- Register for an NCI Event
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
Research articles
Integrative single-cell analysis of human colorectal cancer reveals patient stratification with distinct immune evasion mechanisms
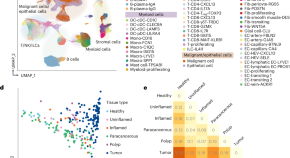
Cheng and colleagues performed an integrative analysis of human colorectal cancer samples to characterize the tumor microenvironment (TME) and stratify patients according to their heterogeneous TMEs, which exploit different immune evasion mechanisms.
- Xiaojing Chu
- Xiangjie Li
- Sijin Cheng

Co-targeting SOS1 enhances the antitumor effects of KRAS G12C inhibitors by addressing intrinsic and acquired resistance
Hofmann and colleagues describe the mechanism underlying the therapeutic benefit of combinatorial use of SOS1 and KRAS-G12C inhibitors in the context of KRAS-G12C mutant-driven lung and colorectal cancer.
- Venu Thatikonda
- Marco H. Hofmann
Building a translational cancer dependency map for The Cancer Genome Atlas
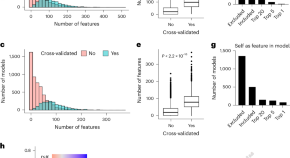
Shi et al. present a hybrid dependency map based on machine-learning analysis of gene essentiality data from the DEPMAP database, translated to data from TCGA. This application can be used to visualize other gene essentiality data.
- Christos Gekas
- Zoltan Dezso

Tumor evolution metrics predict recurrence beyond 10 years in locally advanced prostate cancer
Sottoriva and colleagues combine next-generation sequencing and AI-aided histopathology to assess tumor evolvability in patient samples with long-term follow-up and find that it can be a strong predictor of recurrence in high-risk prostate cancer.
- Javier Fernandez-Mateos
- George D. Cresswell
- Andrea Sottoriva
A first-in-class selective inhibitor of EGFR and PI3K offers a single-molecule approach to targeting adaptive resistance
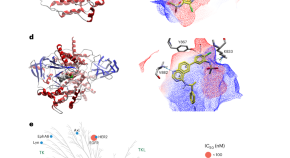
Sebolt-Leopold and colleagues design and develop a small-molecule inhibitor that can target both epidermal growth factor receptor and phosphatidylinositol 3-OH kinase, which can be leveraged to overcome resistance to targeted therapies in vivo.
- Christopher E. Whitehead
- Elizabeth K. Ziemke
- Judith S. Sebolt-Leopold
A deep-learning framework to predict cancer treatment response from histopathology images through imputed transcriptomics
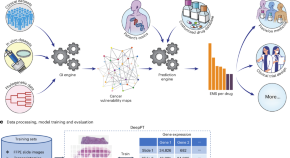
Hoang et al. developed a deep-learning framework called ENLIGHT–DeepPT that predicts therapy response based on imputed transcriptomics and shows predictive power across patient cohorts and cancer types.
- Danh-Tai Hoang
- Gal Dinstag
- Eytan Ruppin

The proteogenomic landscape of multiple myeloma reveals insights into disease biology and therapeutic opportunities
Krönke and colleagues present a multiomic resource of plasma cell malignancies, including multiple myeloma, that comprises phosphoproteomics, RNA and DNA sequencing and provides insights into cancer type biology and candidate therapeutic targets.
- Evelyn Ramberger
- Valeriia Sapozhnikova
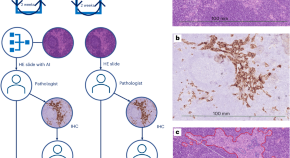
Clinical implementation of artificial-intelligence-assisted detection of breast cancer metastases in sentinel lymph nodes: the CONFIDENT-B single-center, non-randomized clinical trial
Van Dooijeweert et al. conducted a prospective study on the clinical implementation of artificial-intelligence-assisted detection of sentinel lymph node metastasis in persons with breast cancer and report on its effects, including on time and cost.
- C. van Dooijeweert
- R. N. Flach
- P. J. van Diest
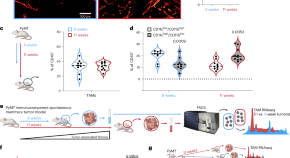
Nucleotide metabolism in cancer cells fuels a UDP-driven macrophage cross-talk, promoting immunosuppression and immunotherapy resistance
Scolaro et al. identify the enzyme cytidine deaminase (CDA) as upregulated in immunotherapy-resistant tumors and find it contributes to the UDP pool, which in turn modulates tumor-associated macrophages to instruct an immune-evasive TME.
- Tommaso Scolaro
- Marta Manco
- Massimiliano Mazzone
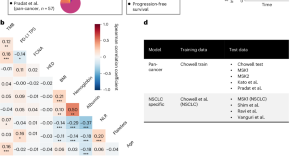
LORIS robustly predicts patient outcomes with immune checkpoint blockade therapy using common clinical, pathologic and genomic features
Chang et al. performed a pan-cancer multimodal data integration analysis and devised a model, LORIS, that can predict objective responses to immunotherapy and patient survival across many cancer types and allow for patient stratification.
- Tian-Gen Chang
- Yingying Cao
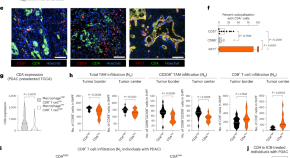
Tumor-associated macrophages restrict CD8 + T cell function through collagen deposition and metabolic reprogramming of the breast cancer microenvironment
Weaver and colleagues show that TGFβ-induced collagen deposition and metabolic reprogramming of the breast cancer microenvironment by tumor-associated macrophages restrict the antitumor activity of CD8 + T cells in female breast cancer.
- Kevin M. Tharp
- Kelly Kersten
- Valerie M. Weaver
Targeting a lineage-specific PI3Kɣ–Akt signaling module in acute myeloid leukemia using a heterobifunctional degrader molecule
Puissant and colleagues identify a myeloid-restricted PIK3CG/p110γ – PIK3R5/p101 axis as a therapeutic vulnerability in acute myeloid leukemia and develop a proteolysis-targeting chimera to potently degrade PIK3CG and suppress AML progression.
- Lois M. Kelly
- Justine C. Rutter
- Alexandre Puissant
Identification of a clinically efficacious CAR T cell subset in diffuse large B cell lymphoma by dynamic multidimensional single-cell profiling
Rezvan and colleagues profile the infusion product from individuals with DLBCL treated with CAR T cells and integrate functional profiling by timelapse imaging microscopy and scRNA-seq to identify a signature of migratory CD8 + T cells associated with response.
- Gabrielle Romain
- Navin Varadarajan
Targeting TGFβ-activated kinase-1 activation in microglia reduces CAR T immune effector cell-associated neurotoxicity syndrome
Zeiser and colleagues show that CAR T cell therapy results in upregulation of the TGFβ-activated kinase-1 (TAK1)–NF-κB–p38 MAPK pathway in microglia, causing neurocognitive defects, and find that TAK1 inhibition can reduce immune effector cell-associated neurotoxicity syndrome.
- Janaki Manoja Vinnakota
- Francesca Biavasco
- Robert Zeiser

Autologous HER2-specific CAR T cells after lymphodepletion for advanced sarcoma: a phase 1 trial
In this phase 1 trial, Hegde et al. treat 13 individuals with advanced sarcoma with lymphodepletion followed by HER2-specific CAR T cells, which were found to be safe and showed antitumor activity.
- Meenakshi Hegde
- Shoba Navai
- Nabil Ahmed

Single-cell multiomic dissection of response and resistance to chimeric antigen receptor T cells against BCMA in relapsed multiple myeloma
Merz and colleagues perform single-cell multiomic analysis of mononuclear cells isolated from individuals receiving BCMA-directed CAR T cell therapy for myeloma and show that nonresponders are characterized by an immune-suppressive microenvironment.
- Michael Rade
- Maximilian Merz
PERCEPTION predicts patient response and resistance to treatment using single-cell transcriptomics of their tumors
Sinha and colleagues present PERCEPTION, a precision oncology computational pipeline that can predict the response and resistance of patients by analyzing single-cell transcriptomic data from their tumor samples.
- Sanju Sinha
- Rahulsimham Vegesna
The selective prolyl hydroxylase inhibitor IOX5 stabilizes HIF-1α and compromises development and progression of acute myeloid leukemia
Lawson et al. show that genetic inactivation of Phd1 or Phd2 hinders progression of AML and compromises leukemic stem cells. They develop a selective PHD inhibitor IOX5 and show therapeutic efficacy in AML, which can be potentiated with venetoclax.
- Hannah Lawson
- James P. Holt-Martyn
- Kamil R. Kranc
An oncolytic virus delivering tumor-irrelevant bystander T cell epitopes induces anti-tumor immunity and potentiates cancer immunotherapy
Ye and colleagues show that an oncolytic virus that delivers tumor-irrelevant bystander T cell epitopes to tumor cells can exploit the abundant population of bystander T cells in the tumor for anti-tumor immunity and potentiate cancer immunotherapy.
- Xiangyu Chen
Direct and selective pharmacological disruption of the YAP–TEAD interface by IAG933 inhibits Hippo-dependent and RAS–MAPK-altered cancers
Chapeau et al. develop a nonallosteric inhibitor of the interaction between YAP and all four TEAD proteins. Treatment with the inhibitor, either as monotherapy or in combination with other treatment modalities, leads to induction of cell death in several in vivo cancer models.
- Emilie A. Chapeau
- Laurent Sansregret
- Tobias Schmelzle
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies

IMAGES
COMMENTS
Organoid biology will further develop with a goal of translating the research into personalized therapy. These research areas may result in the creation of new cancer treatments in the future. Keywords: exosomes, immunotherapy, microbiome, organoid. Cancer research has made remarkable progress and new discoveries are beginning to be made.
The challenge ahead lies in how to explore the data to accelerate new discoveries in cancer biology while advancing precision oncology. You have full access to this article via your institution.
The Cancer Currents blog provides news and research updates from the National Cancer Institute.
Nature Cancer aims to publish the most significant advances across the full spectrum of cancer research in the life, physical, applied and social sciences, ...
MIT researchers have discovered a new way to jump-start the immune system to attack tumors, which could allow cancer immunotherapy to be used against more types of cancer.
Cancer biology is the cornerstone on which much of modern cancer research is based. Continuing to explore the intricacies of this multilayered foundational scientific area is essential.
Since the second half of the 20th century, our knowledge about the biology of cancer has made extraordinary progress. Today, we understand cancer at the genomic and epigenomic levels, and we have identified the cell that starts neoplastic transformation ...
The Cancer Systems Biology Consortium (CSBC), which includes cancer biologists, engineers, mathematicians, physicists, and oncologists, aims to tackle the most perplexing issues in cancer to increase our understanding of tumor biology, treatment options, and patient outcomes. The initiative takes an integrative approach to cancer research, with ...
Research projects funded by the Division of Cancer Biology have led to many new discoveries and advances in basic cancer research that continue to lay the groundwork for future clinical breakthroughs and cancer prevention strategies. Learn about these important cancer biology findings.
eLife reviews research into cancer spanning from oncogenes and tumor suppressors to metastasis and anticancer treatments. Learn more about what we review and sign up for the latest research.
The latest edition of Hallmarks of Cancer adds a new chapter to the holy book of cancer biology, reflecting recent advances.
This Special Issue on "recent developments in cancer systems biology" has compiled several novel approaches that use cutting-edge technologies to build a strong foundation of systems biology in cancer research. The issue includes eight original research articles and four literature reviews on recent efforts that use a variety of in silico ...
A core mission of cancer genomics is to comprehensively chart molecular underpinnings of cancer-driving events and to provide personalized therapeutic strategies. Primarily focused on cancer cells, cancer genomics studies have successfully uncovered many drivers for major cancer types. Since the emergence of cancer immune evasion as a critical ...
Nature Reviews Cancer publishes a dynamic and accessible mix of Reviews, Perspectives, Progress and Highlight articles on the most important primary research ...
Cancer researchers have made an important new connection between a person's cancer risk and the activity of circular RNAs, a poorly understood family of genetic fragments present in the body. A ...
Our understanding of tumorigenesis and cancer progression as well as clinical therapies for different cancer types have evolved dramatically in recent years. However, even with this progress, there are big challenges for scientists and oncologists to tackle, ranging from unpacking the molecular and cellular mechanisms involved to therapeutics and biomarker development to quality of life in the ...
Cancer Biology. cancer gene discovery • tumorigenesis • cancer therapy and resistance • oncogenes • tumor suppressor genes • cancer models • growth control and cell proliferation • metastasis • cell proliferation • cell death • cell-cell and cell-matrix interactions • microenvironment •DNA repair and replication ...
New treatments and technologies offer exciting prospects for cancer research and care, but their global impact rests on widespread implementation and accessibility.
Read the latest medical research on risk factors for cancer, cancer symptoms, treatments and more. Updated daily.
NCI is the nation's leader in cancer research. Learn about key research areas, initiatives, progress, and research tools, including specimens and data.
UVA's Center for Systems Analysis of Stress-adapted Cancer Organelles (SASCO) recently hosted its first annual symposium, sharing updates on major projects and work to improve research inclusivity as the program enters its third year.
The Nature Portfolio editors who handle cancer primary research, methods, protocols and reviews bring you the latest articles, covering all aspects from disease mechanisms to therapeutic ...
University of Queensland research could lead to a more effective immunotherapy treatment against bladder cancer.
The latest cancer news from the US government's principal agency for cancer research, plus resources designed for science writers and reporters.
Nucleotide metabolism in cancer cells fuels a UDP-driven macrophage cross-talk, promoting immunosuppression and immunotherapy resistance. Scolaro et al. identify the enzyme cytidine deaminase (CDA ...
The University of Texas MD Anderson Cancer Center today was awarded nine grants totaling over $21.4 million from the Cancer Prevention and Research Institute of Texas (CPRIT) in support of two new core facilities, faculty recruitment and groundbreaking cancer research across all areas of the institution.