
- My presentations

Auth with social network:
Download presentation
We think you have liked this presentation. If you wish to download it, please recommend it to your friends in any social system. Share buttons are a little bit lower. Thank you!
Presentation is loading. Please wait.
Gene Therapy: Past, Present, and Future
Published by Ζηνόβιος Μαυρίδης Modified over 5 years ago
Similar presentations
Presentation on theme: "Gene Therapy: Past, Present, and Future"— Presentation transcript:

This program will include a discussion of off-label treatment and investigational agents not approved by the FDA for use in the US, and data that were.

Immunotherapies: Key Considerations in the Identification and Management of irAEs.

Systemic Lupus Erythematosus

A New Path Forward: Immune Checkpoint Inhibitors in Bladder Cancer

Clinical Trials in IBD.

Why Gene Therapy for Hemophilia?

This program will include a discussion of off-label treatment and investigational agents not approved by the FDA for use in the United States.

Unraveling Clinical Developments in NASH

Advances in Managing Inhibitors in Patients With Hemophilia A

Pseudomonas Lung Infections in Cystic Fibrosis

Dual Antiretroviral Therapy

Progression After Cancer Immunotherapy in Advanced NSCLC

New Standards of Care in ALK-Translocated Advanced NSCLC

Hemophilia Updates: Incorporating New Concepts Into Practice

Updates in Management of Atopic Dermatitis From Real Patient Cases

Mid-Year Hemophilia Update

Induction Chemotherapy for Patients With High-Risk or Secondary AML

Determining the Best Treatment Algorithm for Patients With Head and Neck Cancer.

The Resurgence of Topical Treatments Across a Spectrum of Psoriasis

Chronic Idiopathic Urticaria
About project
© 2024 SlidePlayer.com Inc. All rights reserved.

Gene Therapy PPT | Free Download Presentation
Gene Therapy PPT | Free Download Presentation : Hello friends, today we are going to share a complete presentation on gene therapy. gene therapy is a technique that is used to cure or treat disease by modifying a person’s genes. There are several mechanisms by which gene therapy works.
Also See: General Seminar Topics
- Bringing in new genes in a person’s body to treat a disease.
- Making genes inactive that are not functioning properly.
- Completely replacing a gene (that is causing the disease) with a fresh and healthy copy of a gene.
Lots of studies and experiments have been made into this technique and it is growing day by day as it is treating many ill peoples. The very first approved gene therapy on humans was done in 1990 to sure a disease named SCID (Severe Combined Immunodeficiency). So lets us learn everything about gene therapy in the above ppt and at the end of this post, you will get a full gene therapy ppt for free download.
Also See: Non- Technical Seminar Topics
There are basically two types of gene therapies that are discussed below.
- Types of Gene Therapy
In Vivo Gene Therapy : In this kind of gene therapy, genes are directly delivered into the target cell to sure a missing gene.
Ex Vivo Gene Therapy : In this gene therapy, genes are removed from the person’s body, and then they are transplanted back into the person’s body. Blood and immunological diseases are treated with the help of ex vivo gene therapy and it is now widely used therapy.
Also See: Shock PPT
Issues Related to Gene Therapy
- Who decides what is normal and what is disorder? It is questionable.
- Due to its high cost, it is only available for wealthy persons.
Content of Gene Therapy ppt for free download
- What Genes can do
- Why Genetic Disorders
- Law of Inheritance
- What is Gene Therapy
- How It Works
- Gene Therapy is Experimental
- Uses of gene therapy
- Goals of gene therapy
- Future of gene therapy
Also See: Hypertension PPT
Here we are giving you Gene Therapy Seminar and PPT with PDF report. All you need to do is just click on the download link and get it.
Gene Therapy PPT and Seminar Free Download
It was all about the Gene Therapy ppt and seminar with pdf report . If you liked it then please share it or if you want to ask anything then please hit the comment button.

Related Posts
Social media marketing ppt presentation seminar free, biomedical waste management ppt presentation free, monkey and the cap seller story ppt presentation free download, 1210 electrical engineering(eee) seminar topics 2024, 112 iot seminar topics-internet of thing presentation topics 2024.

330 Latest AI (Artificial Intelligence Seminar Topics) 2024
No comments yet, leave a reply cancel reply.
Your email address will not be published. Required fields are marked *
This site uses Akismet to reduce spam. Learn how your comment data is processed .

- Upload Ppt Presentation
- Upload Pdf Presentation
- Upload Infographics
- User Presentation
- Related Presentations

Antiviral Drugs
By: FrankMarco Views: 597

Diabetes Mellitus In Children
By: FrankMarco Views: 474

Knee Injuries
By: FrankMarco Views: 618

Human Inheritance
By: FrankMarco Views: 512

Brain Anatomy
By: FrankMarco Views: 776

Genetic Disorders
By: Teodora Views: 1720

Pharmacogenomics
By: medhelp Views: 939


Non-Mendelian Genetics
By: drdwayn Views: 1681

Natural product and Apoptosis
By: aquiamarine2000 Views: 1428

A brief history of imaging genetics
By: KhushbuSG Views: 626

- About : Foundation Year 1 Doctor
- Occupation : Medical Professional
- Specialty : MD
- Country : United Kingdom
HEALTH A TO Z
- Eye Disease
- Heart Attack
- Medications
Academia.edu no longer supports Internet Explorer.
To browse Academia.edu and the wider internet faster and more securely, please take a few seconds to upgrade your browser .
Enter the email address you signed up with and we'll email you a reset link.
- We're Hiring!
- Help Center

Gene therapy.pptx

Related Papers
Shashi Pratap Singh
Dr. Ravindra B. Malabadi
Recent patents on DNA & gene sequences
Gene therapy concept has been being overcome massive challenges from 1972 in ethical, socio-economical and developmental issues. In this review, we have attempted to go through almost all the arenas and described in a methodical way that reflects not only the initial ethical and scientific thoughts but also adorned a solid depiction of gene therapy related physico-chemical barriers, approaches and strategies till to date.
rachna kaul
Laurie Doering
Circulation
Laura Denby
IJAR Indexing
Gene therapy is an experimental technique that uses genes encoding the expression of proteins that are either endogenous or biological to treat or prevent a disease by inserting it into patient's cells. Previously the challenges faced were the gene titers which then changed to their delivery and now expression of the genes. Initially the trial of gene therapy for adenosine deaminase deficiency had proven to be successful but till date only alipogenetiparvovec has been approved in Europe for lipoprotein lipase deficiency. Recent techniques with adenovirus, r-adeno associated virus (r-aav), retrovirus,herpes simplex virus vectors and non-viral lipoplexes (for transfer of large biological products) have completely revolutionized the future of gene therapy. With the upcoming advances in the field of cancer therapy such as herpes simplex thymidine kinase (hs-tk) and its role in suicidal gene therapy in the treatment of cancers has stretched new horizons in the field of gene therapy. Recently the newer treatment approaches with RNA interference and anti-sense oligonucleotides in muscular dystrophies were certainly encouraging for the researchers to further work on the newer prospects of gene therapy. Gene therapy is currently investigated for cardiac failure and for prevention of brain ageing in Alzheimer's disease.
International Archives of Medicine
Simon N Waddington , Michael Themis , Anna David , C. Coutelle , Richard Harbottle
Parveen Salahuddin
Abeloff's Clinical Oncology
James E Talmadge
Loading Preview
Sorry, preview is currently unavailable. You can download the paper by clicking the button above.
RELATED PAPERS
Laboratory Hematology
Faris Alenzi
magendira mani vinayagam
Muhammad M Ibrahim
Raju Jakkula
Biochemical Society Transactions
Jerry Chan , Anna David
Biology of Blood and Marrow Transplantation
Anton Neschadim
Therapeutic Delivery
Simon N Waddington
Journal of Hepatology
Jayanta Roy-Chowdhury
Drug Discovery Today
Carl Pinkert
Ferhat Ozturk
Critical Reviews in Oncology/Hematology
Systems Biology in Reproductive Medicine
Joaquin Gadea
Liver transplantation : official publication of the American Association for the Study of Liver Diseases and the International Liver Transplantation Society
Rajagopal Aravalli , Clifford Steer
Nature Biotechnology
Hatem Zayed
Molecular therapy : the journal of the American Society of Gene Therapy
Zsuzsanna Izsvák
Clinical Orthopaedics and Related Research
Chisa Hidaka
Bob Perkins
Heba N A G E H Gad EL-Hak , Mariam M. Gerges
Clinical Oncology
Atique Ahmed
Molecular Human Reproduction
C. Coutelle , Michael Themis
European Journal of Endocrinology
Luisa Barzon
Arthritis & Rheumatism
Steven Ghivizzani
Liver Transplantation
Clifford Steer
Diana Chiru
Survey of Ophthalmology
Edward Chaum
Molecular Therapy
Perry Hackett
Human Gene Therapy
Justyna Leja-jarblad , Magnus Essand
Molecular Biotechnology
Drugs of the Future
James Mixson
Crit Rev Oncol Hematol.
Mate Hidvegi
Current Gene Therapy
Emran Bashar
The Journal of Gene Medicine
Joanne Wixon
Journal of Cellular and Molecular Medicine
Raymund Horch
R. Karda , Simon N Waddington
International Review of Cell and Molecular Biology
Gerald Schumann
Hematology-oncology Clinics of North America
Ronald Rodriguez
- We're Hiring!
- Help Center
- Find new research papers in:
- Health Sciences
- Earth Sciences
- Cognitive Science
- Mathematics
- Computer Science
- Academia ©2024
JavaScript seems to be disabled in your browser. For the best experience on our site, be sure to turn on Javascript in your browser.
- My Wish List
- Compare Products
- Presentations
Gene Therapy
You must be logged in to download this file*
item details (6 Editable Slides)
(6 Editable Slides)
Related Products
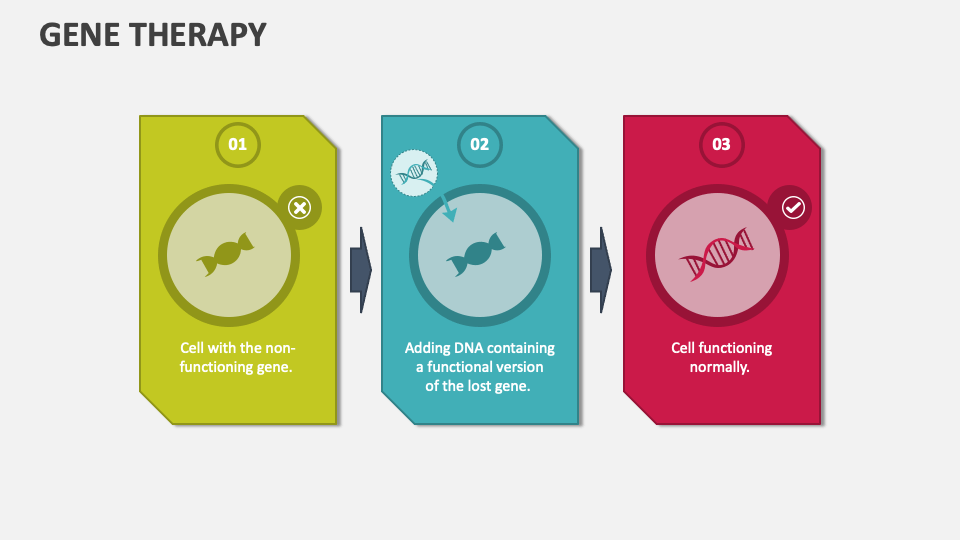
Grab our Gene Therapy presentation template for MS PowerPoint and Google Slides to describe the medical approach that genetically modifies an individual’s cells to treat or prevent diseases. Save loads of time and effort with these pre-designed slides!
Healthcare professionals will find this proficiently designed set handy in depicting the objectives, applications, advantages, and disadvantages of gene therapy in a comprehensible manner. You can also explain the features and key aspects related to somatic and germline gene therapy. Moreover, using our PPT, you can shed light on the problems and issues that arise as a result of gene therapy.
Sizing Charts
| Size | XS | S | S | M | M | L |
|---|---|---|---|---|---|---|
| EU | 32 | 34 | 36 | 38 | 40 | 42 |
| UK | 4 | 6 | 8 | 10 | 12 | 14 |
| US | 0 | 2 | 4 | 6 | 8 | 10 |
| Bust | 79.5cm / 31" | 82cm / 32" | 84.5cm / 33" | 89.5cm / 35" | 94.5cm / 37" | 99.5cm / 39" |
| Waist | 61.5cm / 24" | 64cm / 25" | 66.5cm / 26" | 71.5cm / 28" | 76.5cm / 30" | 81.5cm / 32" |
| Hip | 86.5cm / 34" | 89cm / 35" | 91.5cm / 36" | 96.5cm / 38" | 101.5cm / 40" | 106.5cm / 42" |
| Size | XS | S | M | L | XL | XXL |
|---|---|---|---|---|---|---|
| UK/US | 34 | 36 | 38 | 40 | 42 | 44 |
| Neck | 37cm / 14.5" | 38cm /15" | 39.5cm / 15.5" | 41cm / 16" | 42cm / 16.5" | 43cm / 17" |
| Chest | 86.5cm / 34" | 91.5cm / 36" | 96.5cm / 38" | 101.5cm / 40" | 106.5cm / 42" | 111.5cm / 44" |
| Waist | 71.5cm / 28" | 76.5cm / 30" | 81.5cm / 32" | 86.5cm / 34" | 91.5cm / 36" | 96.5cm / 38" |
| Seat | 90cm / 35.4" | 95cm / 37.4" | 100cm / 39.4" | 105cm / 41.3" | 110cm / 43.3" | 115cm / 45.3" |

Gene Therapy
Oct 23, 2014
850 likes | 2.53k Views
Gene Therapy. Meredith Calhoun. What is Gene Therapy?. - Gene therapy is a form of treatment that changes defective genes to stop a disease. - Gene therapy can be used instead of medicines or surgery. - It uses viruses to genetically modify a cells DNA. How does it work?.
Share Presentation
- gene therapy
- target cells
- curing blindness
- received gene therapy
- somatic cell gene therapy

Presentation Transcript
Gene Therapy • Meredith Calhoun
What is Gene Therapy? - Gene therapy is a form of treatment that changes defective genes to stop a disease. - Gene therapy can be used instead of medicines or surgery. - It uses viruses to genetically modify a cells DNA.
How does it work? • In gene therapy, researchers use viruses to correct the genes in cells by injecting desired genetic material in to the virus. MANY viruses are then put in the body to deliver the correct gene to the cells or take the cells out and let the DNA be corrected. Then corrected cells are put back into the body. These viruses have been genetically engineered so that they can't reproduce. Scientists are researching other ways to accomplish gene therapy. They are looking at a fatty molecule known as a liposome and physically inserting genes into the cell through "gene guns" or micropipettes.
Steps: • 1. Diagnose disease or disorder and then find the gene that causes the disorder. • 2. Engineer virus and insert desired genes. • 3. Inject viruses into body (Vivo) or remove target cells for the disease (Ex Vivo). • 4. Allow viruses to inject genes into cells DNA to fix disorder. • 5. If target cells were removed and placed in culture with the viruses, then the corrected cells will be placed back in the body. Adenovirus
Two types: • Somatic cell gene therapy- is gene therapy, or changing genes to fix a disease, in normal body cells. • This can fix the disease and cause the correct protein to be made, but the gene will not be passed on to your offspring. • Germline Therapy- is just replacing genes in reproductive cells • Because genes are changed in reproductive cells the new gene can be passed on to the offspring. • Affects many generation's DNA make up. The child will have the correct gene instead of the defected one.
History • The 1960's and 1970's was when the idea of gene therapy first arose. • By 1972, Theodore Friedman and Richard Roblin published a paper explaining Stanfield Rogers gene therapy idea for curing humans with diseases. • In 1985 Dr.s W. French Anderson and Michael Blaese from the National Heart, Lung, and Blood Institute and the National Cancer Institute worked together to find that they could cure people,with Adenosine Deaminase Deficiency (ADA), using gene therapy. They conducted the first gene therapy in ADA cells in a tissue culture.
History Continued • In 1986 Anderson and Blaese transferred correct genes into bone marrow of animals. • In 1988 they found gene therapy was more successful in white blood cells. • In 1989 Anderson and Blaese teamed up with Dr. Steven Rosenberg to see if gene therapy would be successful in patients with cancer, particularly melanoma.
In the 1990's the first condition in humans was treated with Gene Therapy by Dr.s W. French Anderson and Michael Blaese. Two little girls, a four and nine year old, with an immune deficiency called ADA were cured through gene therapy.
Obviously it's a positive that gene therapy can eliminate disorders before they begin or get to bad. • It can guarantee a cure for future suffering of some diseases. • Gene Therapy is considered a "medicine for the future" because it can control or alter Germ cells. Resulting in cured hereditary diseases and changing DNA for future generations. • Possibilities are endless. Since gene therapy is such a new technology, researchers are still finding gene therapy can cure many diseases. • Can possibly cure cancer, heart and lung disease, muscular dystrophy, blindness, AIDS, or even Alzheimer's.
The Cons • With all new technologies there are possible consequences.
Before gene therapy begins,your immune system must be weakened, so that the viruses can correctly inject the gene. The immune system can't tell the difference between a bad virus and a helping virus. • Since it is so new, it can be very expensive. • It's a new technology so not much is known about it. There could be consequences we don't know about yet. • The virus may change DNA in cells that don't need fixings or insert genes into the wrong part of the genome, this can lead to more serious problems such as cancer.
Ethical and Religious Considerations • The first ethical issue is, Gene Therapy would put human fate in our hands by giving us the ability to affect genes of future generations. • Some are concerned it could give people the ability to make a superior race(eugenics), however this is not the goal of geneticists. • Some predict it would be hard to reinforce laws on gene therapy. And it could be offered on the black market. • Religion also plays a large role in an individual's considerations, some consider gene therapy is sinful. People believed gene therapy would be like "playing God".
Current Applications • Gene therapy can be used to correct single gene mutation diseases like: • Cystic Fibrosis • Sickle Cell Anemia • Hemophilia has been treated in the U.S. Corrected the gene that allows the blood to correctly clot. • X-SCID is a immune deficiency where people lack B and T cells. Gene therapy corrects mutation on the X-gene and this causes the cells to be produced. • Type I Diabetes has been corrected in rats and dogs. • Paget's Diseases is a rare bone metabolism disorder caused by a gene being missing.
Current Applications Continued • Chronic Granulomatus Disorder (CDG) has been cured in two patients. This disorder causes people to not be able to fight off bacterial and fungal infections. • Some cancer cells have been cured in a lab with many different types of gene therapy. • Neurodegenerative diseases like Parkinson's and Huntington's Disease have been cured in animals through gene therapy. • Diseases that are caused by more then one gene are being researched but are not yet used because of how complex it is to correct more than one gene at a time.
Case Study • Using Gene Therapy to reverse blindness in dogs.
Curing Blindness in Dogs • University of Pennsylvania researchers led scientists in using gene therapy to cure three puppies that were born blind. • The dogs in this experiment had a disease known as Leber Congenital Amaurosis (LCA). • The gene that is mutated stops making proteins that allow normal vision. • Retinitis Pigmentosa GTPase Regulator (RPGR) is the gene that contains the error. • The protein that can't be produced is photoreceptor ciliary. • The retina deteriorates and becomes dysfunctional when the dog has LCA.
Curing Blindness Continued • A few weeks after the new genes were injected into a retina of one eye in each puppy, they began to gain vision. They could see and interact with humans and began to maneuver around objects. • A month after the genes had been injected into the retina, the scientists began testing the activity of each eye (with or without the corrected genes). • They used a technique called electro retinography, it measures the levels of electrical activity in the retina. • At the two month mark there was a 35%-40% electrical activity occurring in the retina that received gene therapy. The eye that didn't receive gene therapy had little to no activity.
Since LCA occurs in humans, scientists are now researching to see if the gene therapy used on the puppies would still be successful on humans.
Resources • http://www.esgct.eu/useful-information/introduction-to-gene-therapy.aspx • http://ghr.nlm.nih.gov/handbook/therapy/genetherapy • http://www.ndsu.edu/pubweb/~mcclean/plsc431/students98/fleck.htm • http://www.ama-assn.org//ama/pub/physician-resources/medical-science/genetics-molecular-medicine/current-topics/gene-therapy.page • http://history.nih.gov/exhibits/genetics/sect4.htm#1 • http://abcnews.go.com/GMA/t/story?id=127024&ref=https%3A%2F%2Fwww.google.com%2F • http://www.hc-sc.gc.ca/sr-sr/biotech/about-apropos/gen_therap-eng.php
- More by User

Gene Therapy Maren Cannell Daniel Garzon Mariah Payne Genes Are carried on a chromosome The basic unit of heredity Encode how to make a protein DNA RNA proteins Proteins carry out most of life’s function. When altered causes dysfunction of a protein
2.85k views • 25 slides

Gene Therapy. The Forefront of Medicine. Group Members. Maja Udovcic – Background and Introduction Matthew Lee – Scientific Considerations Vanessa Crawford – Social Considerations Sean Park – Ethical Considerations Emily Edwards – Case Studies. Gene Therapy - Background.
3.66k views • 74 slides

Gene Therapy. Maren Cannell Daniel Garzon Mariah Payne. Genes. Are carried on a chromosome The basic unit of heredity Encode how to make a protein DNA RNA proteins Proteins carry out most of life’s function. When altered causes dysfunction of a protein
1.24k views • 25 slides
GENE THERAPY
GENE THERAPY. In humans. Cancer 69%. The Food and Drug Administration (FDA) has not yet approved any human gene therapy product for sale. 2) Immune response . It reduces gene therapy effectiveness and makes repetitive rounds of gene therapy useless.
1.11k views • 18 slides

Gene Therapy. Presented to : Dr.Leslye Jhonson Presented by: Khazeema Yousaf & Maheen Alam Biot 412: Medical Biotechnology. What is gene therapy? How does gene therapy works? Tools of Trade—Vector What are the types of gene therapy? Advantages and disadvantages of gene therapy History
1.83k views • 17 slides
GENE THERAPY. Agustina Setiawati , M.Sc., Apt. DEFINITION. Gene therapy is a technique for correcting “defective” genes responsible for disease development. CYSTIC FIBRIOSIS. Cystic fibrosis (CF) is one of the most fatal heredity
1.11k views • 42 slides

Gene Therapy. Molecular genetics presentation Made by / Mostafa Ahmed Supervisor Dr / Noha Khalifa. Introduction. Imagine that you accidentally broke one of your neighbor's windows. What would you do? You could:
873 views • 20 slides

GENE THERAPY.
711 views • 16 slides

Gene Therapy. Phung Nguyen. Brief Overview. What Gene Therapy Is The Different Methods of Gene Therapy Ex vivo: indirect injection Types of tissue removed and process Viral and Non-viral In vivo: direct injection Viral and Non-Viral Uses and Costs of Gene Therapy.
520 views • 14 slides

Gene Therapy. Outcomes: - describe and explain the causes and symptoms of c.f. -compare germline and somatic gene therapy and describe the techniques used for each. -construct a flow chart/diagram showing the stages in gene therapy for cystic fibrosis. Cystic fibrosis.
546 views • 15 slides

Gene therapy
Gene therapy. Gene therapy Using gene technology to treat genetic disorders Usually involves adding a functional copy of a gene to cells which only contain a dysfunctional copy. The dysfunctional gene is not removed, which means that this technique works best with recessive disorders (why?)
440 views • 13 slides

Gene Therapy. by Jin Su Kim & Jun Ho Kang. Index. What is Gene Therapy? How is Gene Therapy Done? Disadvantage and Advantage Benefits & Problems Ethical considerations. What is Gene Therapy?. Treatment of disorders by inserting genetic material inside the cell
781 views • 8 slides

Gene Therapy. Kortlynn Johnson. What is Gene Therapy?. A technique for correcting defective genes responsible for disease development 1. Correcting Faulty G enes. Doctors may use 1 of these approaches 1 :.
1.14k views • 20 slides

Gene Therapy. Antisense Technologies. antisense oligonucleotides(ODNs) Ribozymes DNAzymes RNA interference(RNAi). Part of Gene Therapy. Gene Therapy... ----idea is to replace defective genes. Gene therapy is a technique for correcting defective genes responsible for disease development.
609 views • 11 slides

Gene therapy. Class presentation Nanobiotechnology 2004. Gene therapy. Use of genes to treat disease Several approaches:
478 views • 14 slides
Gene therapy. Fabrizia Urbinati 01/12/2010. Outline. Gene therapy introduction: Delivery method Vectors Candidate Diseases ADA-SCID clinical trial b -Thalassemia. What is gene therapy?. Introduction of normal genes into an individual’s cells and tissue to treat a genetic disease.
1.68k views • 32 slides

Gene Therapy. Done By: Abeer Al-Taweel Albatool Bin Ajlan Amal Alblwi Atheer Hakami Rawan Almugbel Shaikha Aldukhail Supervised by: Dr. Amina Alshibani. Definition. What is Gene therapy?.
1.17k views • 14 slides
GENE THERAPY. Diseases for applying gene therapy. Disease Defect Target cell Severe combined Bone marrow cells or immunodeficiency T-lymphocytes Hemophilia Liver, muscle Cystic fibrosis Lung Cells
874 views • 20 slides

Gene Therapy. By: Sofiya Jooma & Gigi Singh. Introduction. Gene Therapy is a scientifically based method of using a person’s genes to treat or cure a disease. Did you know that Gene Therapy is becoming increasingly popular in medicine? (Hunt, Odle , 2006)
1.49k views • 12 slides

GENE THERAPY. GENE THERAPY. Any procedure intended to treat or alleviate disease by genetically modifying the cells of a patient. GENE THERAPY. Delivery mechanism Ex vivo In vivo Type of cells modified Germ-line cells Somatic cells Mechanism of modification
759 views • 25 slides
Gene Therapy. Medical Interventions Harrison. http://www.wellesley.edu/Biology/Courses/219/Gen_news/i3_Gene_Therapy.jpg. Genes. Are carried on a chromosome The basic unit of heredity Encode how to make a protein DNA RNA proteins Proteins carry out most of life’s function.
715 views • 25 slides
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
The PMC website is updating on October 15, 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- eBioMedicine
- v.106; 2024 Aug
- PMC11372377

Realising gene therapy: a biomedical challenge
In recent years, advancements in applications of gene therapy have accelerated. Gene therapy aims to treat and potentially cure people with genetic diseases that have a substantial effect on quality of life and for which no or few therapeutic options are available. As developments continue at a rapid pace, promising outcomes frequently feature prominently in the news. In November, 2023, the UK Medicines and Healthcare products Regulatory Agency gave the first conditional market authorisation of a CRISPR-based genome-editing therapy for the treatment of people with sickle cell disease and transfusion-dependent β-thalassaemia (TDT) for whom haematopoietic stem cell transplantation is appropriate but not available. The US Food and Drug Administration and the European Commission followed shortly thereafter. In the UK and EU, an estimated 10,000 people with sickle cell disease and TDT are potentially eligible for treatment.
These approvals were delivered after encouraging interim efficacy results from one clinical trial for each disease. The results, published in The New England Journal of Medicine , revealed that most people who received treatment remained free from severe vaso-occlusive crises in sickle cell disease or did not need red blood cell transfusion in TDT for at least 12 consecutive months. Sickle cell disease and TDT affect the ability of red blood cells to carry oxygen and are caused by genetic mutations in both copies of the HBB gene encoding haemoglobin. Rather than editing each mutation of individual people, the strategy of this treatment approach (known as exagamglogene autotemcel [exa-cel]) is to reinstate the expression of fetal globin genes , which are postnatally silenced by transcription factors such as BCL11A. More specifically, the enhancer regulating the expression of BCL11A is inactivated by ex-vivo genome-editing in bone-marrow stem cells, allowing them to express fetal haemoglobin. The cells are then transplanted back, where they can proliferate in the bone marrow.
In a study published in The New England Journal of Medicine in January, 2024, Longhurst and colleagues also used an indirect strategy for hereditary angioedema, thanks to which eligible participants could be administrated the same in-vivo CRISPR-based gene-editing therapy. Instead of editing the mutations in SERPING1 , the gene causing the disease, the gene-editing strategy targeted the KLKB1 gene encoding plasma kallikrein, a downstream protease in the dysregulated contact activation pathway. Ten participants received a single dose of the treatment. No severe adverse events were detected, plasma kallikrein concentrations were reduced for at least 24 weeks, and exploratory analyses promisingly showed a reduction in the number of angioedema attacks per month.
As discussed in a Review published in The Lancet in February, 2024, a core advantage of gene or genome-editing therapies over gene-addition therapies is its potential to expand the spectrum of targetable diseases to autosomal dominant genetic disorders, for which adding a non-mutated gene is not an adequate solution. Instead, genome-editing strategies can directly repair disease-causing mutations or modify gene or genome sequences. They also overcome a limitation of gene-addition therapies whereby the expression of the added gene, typically controlled by promoters and enhancers different from those found in nuclei of human cells, have little fine-tuning. Reflecting its versatility, therapeutic applications and ongoing clinical trials based on genome-editing strategies cited in this Review are as diverse as cancer immunotherapy, HIV infection, type 1 diabetes, or organ xenotransplantation.
Yet, the risk of unwanted effects exists and is linked to off-target editing (ie, unintended genetic modifications) and immunogenicity against the engineered nucleases, such as Cas9, or the viral-based vectors delivering the molecular components of this technology. Basic research and preclinical studies continue to provide computational and biological tools that help reduce this risk. For example, in a study published in Molecular Therapy in August, 2023, Stahl and colleagues optimised the transient delivery of Cas9 ribonucleoproteins in the mouse brain, leading to reduced immune responses but similar editing capacity in neurons to a commonly used adeno-associated virus (AAV)-based delivery.
Gene-addition therapies are complementary approaches to genome editing and, despite being used to treat human diseases for more than a decade, clinical trials continue to test their breadth of therapeutic applications. In a single-arm, single-centre trial published in The Lancet in May, 2024, Lv and colleagues used a gene-addition strategy to treat children who, due to biallelic mutations in the OTOF gene, had autosomal recessive deafness 9. A single injection of an AAV carrying a human OTOF transgene into the cochlea of six children aged 1–6 years with severe-to-complete hearing loss resulted in no dose-limiting toxicity or serious adverse events; five children had hearing recovery and improved speech perception.
However, successes in gene therapies should not make us forget disappointing outcomes. In June, 2024, Pfizer announced that boys aged 4–7 years with Duchenne muscular dystrophy treated with fordadistrogene movaparvovec , an AAV carrying a shortened version of the dystrophin gene, had no improvement of motor function 1 year after treatment compared with placebo. Besides no efficacy, safety concerns can also be a reason for poor outcomes. The ASPIRO trial evaluating the safety and efficacy of a single intravenous infusion of resamirigene bilparvovec was stopped after the death of four participants. The investigational AAV-based gene-replacement therapy of MTM1 was administered to boys younger than 5 years who had X-linked myotubular myopathy (XLMTM) and required long-term mechanical ventilation. Exploratory analyses of the trial reported in The Lancet Neurology highlighted that undiagnosed cholestatic liver disease in children with XLMTM can potentially interact with the gene therapy, leading to serious hepatic and hepatobiliary adverse events and progressive liver disease unresponsive to immune suppression. Nevertheless, a promising reduction in ventilator dependence and improvement of motor function were observed in some surviving participants. In a companion Article published in eBioMedicine , Lawlor and colleagues reported a histopathological analysis of muscle biopsies showing improvement of organelle localisation and myofiber size 24 weeks after treatment.
Dowling and colleagues published in Nature Medicine in July, 2024, a study in which a single participant aged 4 years with hereditary spastic paraplegia type 50 was administrated an AAV-based gene-addition therapy of the target gene, AP4M1, intrathecally. After 12 months, no serious adverse events were detected and preliminary outcomes suggested that the progressive disease course, including developmental delay, microcephaly, and loss of motor skills, might be inhibited. Dowling and colleagues discussed time and costs as key aspects to consider when developing a single-gene therapeutic. In this case, development took almost 3 years from diagnosis to dosing and cost more than CA$3,500,000.
Affordability of treatment for all individuals in need remains an unsolved challenge in gene therapy. Although more than 75% of people with sickle cell disease are born in Africa, as reminded by Munung and colleagues in their review published in Gene Therapy , exa-cel trials have not involved any African countries so far. More generally, Munung and colleagues identified ethical, legal, and social issues that might need to be overcome for the implementation of gene therapy for people with sickle cell disease in Africa. Moreover, the scarce availability of genetic testing in many low-income and middle-income countries and the under-representation of diverse ethnic populations in genomic datasets and genetic research, impeding our understanding of genetic variants in many populations, are initial barriers keeping many individuals far from gene therapy.
As gene therapy enters the area of genome editing, a multidisciplinary and collaborative research effort is required to provide an efficient, safe, and affordable solution for all individuals with burdensome genetic disorders. In this field, basic and preclinical research have made and continue to make invaluable contributions to the clinic by deciphering biological underpinnings of diseases and providing the therapeutic tools to treat them. eBioMedicine welcomes translational research that addresses some of the challenges in this fundamentally different medical approach, in which a cure rather than a treatment might be achievable.

IMAGES
VIDEO
COMMENTS
Gene therapy is an experimental treatment that involves introducing genetic material (DNA or RNA) into a person's cells to fight disease. Majority are Trails • Gene therapy is being studied in clinical trials (research studies with people) for many different types of cancer and for other diseases.
Two examples of Gene Therapy for hematologic diseases. • ADA-SCID • b-thalassemia. Replacement of the gene in Hematopoietic Stem Cells (HSC) Blood and Tissues Bone Marrow. Adenosine-Deaminase (ADA) Deficiency • ADA is an enzyme involved in purine metabolism; It is needed for the breakdown of adenosine from food and for the turnover of ...
Free Download Gene Therapy PowerPoint Presentation. Methods of gene delivery (therapeutic constructs) -- Injection of naked DNA into tumor by simple needle and syringe -- DNA transfer by liposomes (delivered by the intravascular, intratracheal, intraperitoneal or intracolonic routes) -- DNA coated on the surface of gold pellets which are air-propelled into the epidermis (gene-gun), mainly non ...
Sep 25, 2011. 1.31k likes | 3.65k Views. Gene Therapy. The Forefront of Medicine. Group Members. Maja Udovcic - Background and Introduction Matthew Lee - Scientific Considerations Vanessa Crawford - Social Considerations Sean Park - Ethical Considerations Emily Edwards - Case Studies. Gene Therapy - Background. Download Presentation.
Download presentation. Presentation on theme: "Gene Therapy."—. Presentation transcript: 1 Gene Therapy. 2 The invention of recombinant DNA technology consequently. led to the immediate inception of engineered gene transfer into human cells, aiming at reversing a cellular dysfunction or creating new cellular function.
Download presentation. Presentation on theme: "Gene Therapy: Past, Present, and Future"— Presentation transcript: 1 Gene Therapy: Past, Present, and Future. 3 This program will include a discussion of off-label treatment and investigational agents not approved by the FDA for use in the United States. 4 Overview of Hemophilia Treatment.
Gene therapy. Lecture Plan. Molecular Medicine and Gene Therapy: An Introduction Vectors in Gene Therapy Viral vector of gene therapy Non viral vector of gene therapy Gene Targeting Cancer genes and the pathways they control The Genetic Basis of Cancer Slideshow 5651035 by haruki.
Here we are giving you Gene Therapy Seminar and PPT with PDF report. All you need to do is just click on the download link and get it. Gene Therapy PPT and Seminar Free Download. It was all about the Gene Therapy ppt and seminar with pdf report. If you liked it then please share it or if you want to ask anything then please hit the comment button.
This medical PowerPoint presentation talks about Human gene therapy is a technique that involves the introduction or modification of genetic material within a persons cells to treat or prevent disease. This is achieved by using various methods, including viral vectors, non-viral vectors, and genome editing tools, to deliver therapeutic genes ...
IJAR Indexing. Gene therapy is an experimental technique that uses genes encoding the expression of proteins that are either endogenous or biological to treat or prevent a disease by inserting it into patient's cells. Previously the challenges faced were the gene titers which then changed to their delivery and now expression of the genes.
Download our editable Gene Therapy presentation template for MS PowerPoint and Google Slides to exhibit the objectives, applications, ... Grab our Gene Therapy presentation template for MS PowerPoint and Google Slides to describe the medical approach that genetically modifies an individual's cells to treat or prevent diseases. Save loads of ...
Steps: • 1. Diagnose disease or disorder and then find the gene that causes the disorder. • 2. Engineer virus and insert desired genes. • 3. Inject viruses into body (Vivo) or remove target cells for the disease (Ex Vivo). • 4. Allow viruses to inject genes into cells DNA to fix disorder. • 5.
Dowling and colleagues published in Nature Medicine in July, 2024, a study in which a single participant aged 4 years with hereditary spastic paraplegia type 50 was administrated an AAV-based gene-addition therapy of the target gene, AP4M1, intrathecally. After 12 months, no serious adverse events were detected and preliminary outcomes ...